Headlines
New Research
- Prime editing-installed suppressor tRNAs for disease-agnostic genome editing
- A skin-permeable polymer for non-invasive transdermal insulin delivery
- AI-driven discovery of dual antiaging and anti-AD therapeutics
- Deep generative classification of blood cell morphology
- Matrix mechanical remodeled carrier-free nanosystem for reversal of liver fibrosis
Industry News
- GSK picks Flagship Pioneering’s Quotient, ProFound as 1st biotech partners for $7B pact
- Arbiter Emerges from Stealth with $52M Funding
- Philips and Cortechs.ai Expand Partnership to Embed AI Neuroimaging Analytics
- Atropos Health Integrates Evidence™ Agent with Microsoft Teams
Clinical Studies & Translational Reports
- A brain implant that could rival Neuralink’s enters clinical trials
- Factor IX-Padua AAV gene therapy in hemophilia B: phases 1/2 and 3 trials
- Aspen enriches Parkinson’s cell therapy trial with Kite-backed $115M series C
- Efficacy, Immunogenicity, and Safety of Modified mRNA Influenza Vaccine
- Merck KGaA links up with Flagship’s Valo Health in search for Parkinson’s targets
New Research
1. Prime editing-installed suppressor tRNAs for disease-agnostic genome editing
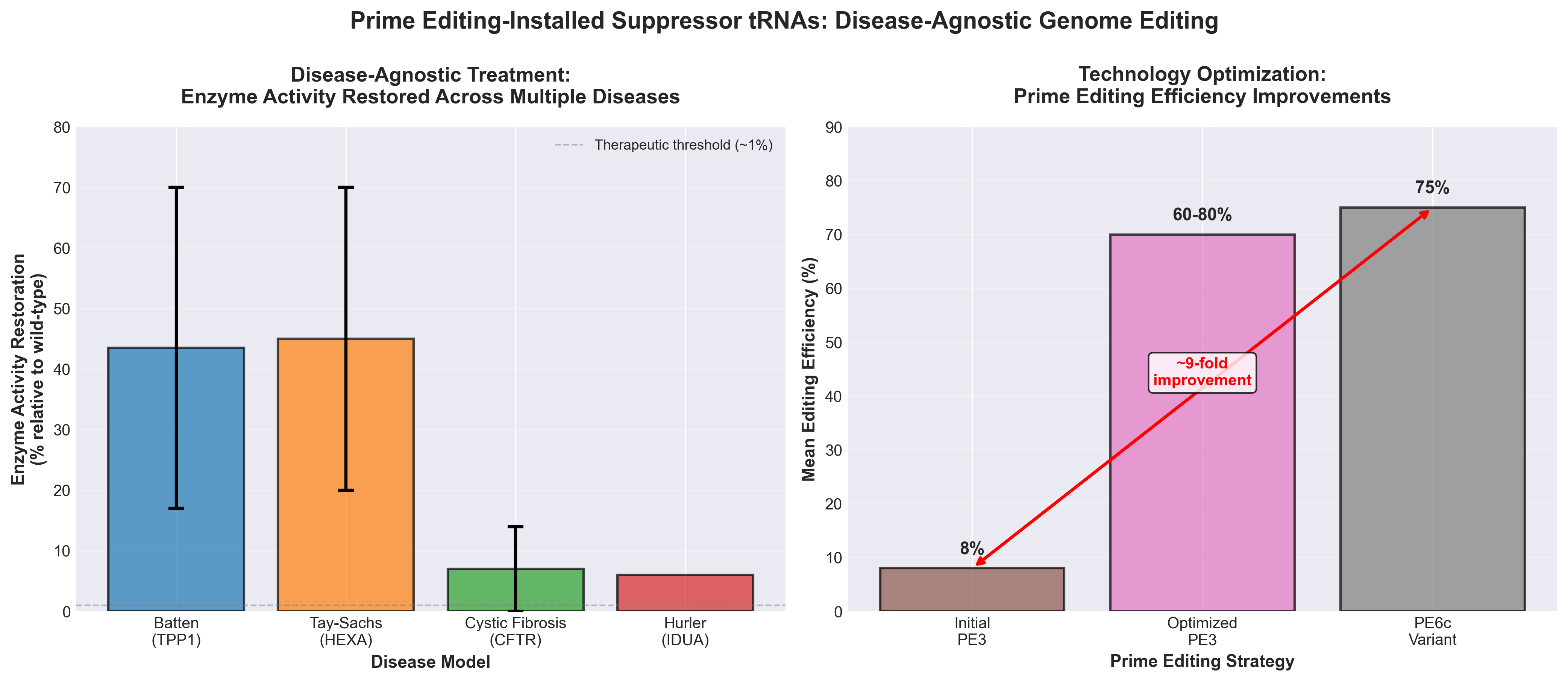
• Disease-agnostic platform reduces development burden: PERT uses a single therapeutic composition to treat multiple genetic diseases caused by premature stop codons (demonstrated in Batten disease, Tay–Sachs, cystic fibrosis, and Hurler syndrome), potentially reducing the need to develop distinct treatments for each of 200,000+ known pathogenic mutations and accelerating time-to-clinic for multiple indications simultaneously.
• Permanent single-locus genomic edit eliminates chronic dosing: Unlike existing suppressor tRNA therapies requiring lifelong administration, PERT achieves durable disease rescue through one-time conversion of an endogenous tRNA, eliminating manufacturing scale-up and patient compliance challenges associated with repeated dosing.
• Screening-optimized tRNA integration ensures safety and efficacy: Iterative screening of all 418 human tRNAs identified optimal candidates that enable efficient readthrough without detectable off-target effects on natural stop codons or significant transcriptomic/proteomic changes, reducing the risk profile compared to overexpression-based approaches.
2. A skin-permeable polymer for non-invasive transdermal insulin delivery
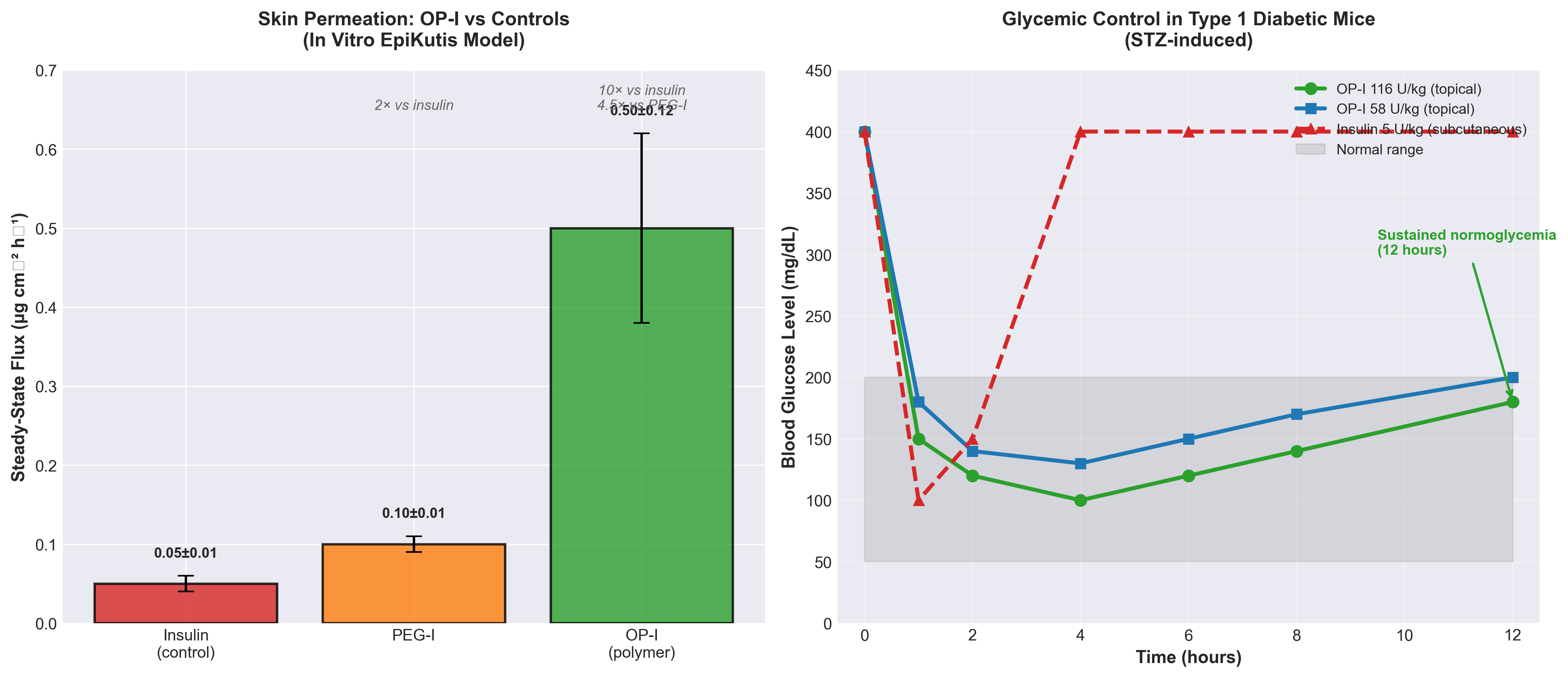
• First non-invasive transdermal insulin delivery achieves therapeutic efficacy: OP-conjugated insulin successfully normalized blood glucose in diabetic minipigs at 29 U/kg via skin application, eliminating the need for subcutaneous injections and addressing a major pain point in diabetes management affecting millions of patients globally.
• Polyzwitterion chemistry enables delivery of large biomolecules previously considered impossible: The OP polymer’s pH-responsive behavior (cationic in acidic sebum, neutral at physiological pH) overcomes the stratum corneum barrier for proteins/peptides, potentially unlocking transdermal delivery for an entire class of therapeutics currently requiring injections or infusions.
• Significant commercial opportunity across multiple therapeutic modalities: Beyond insulin, this platform technology could enable non-invasive delivery of other protein and peptide-based therapeutics, reducing manufacturing complexity, improving patient compliance, and creating new market segments for GLP-1 agonists, monoclonal antibodies, and biologics currently limited to injectable/IV routes.
3. AI-driven discovery of dual antiaging and anti-AD therapeutics
4. Deep generative classification of blood cell morphology
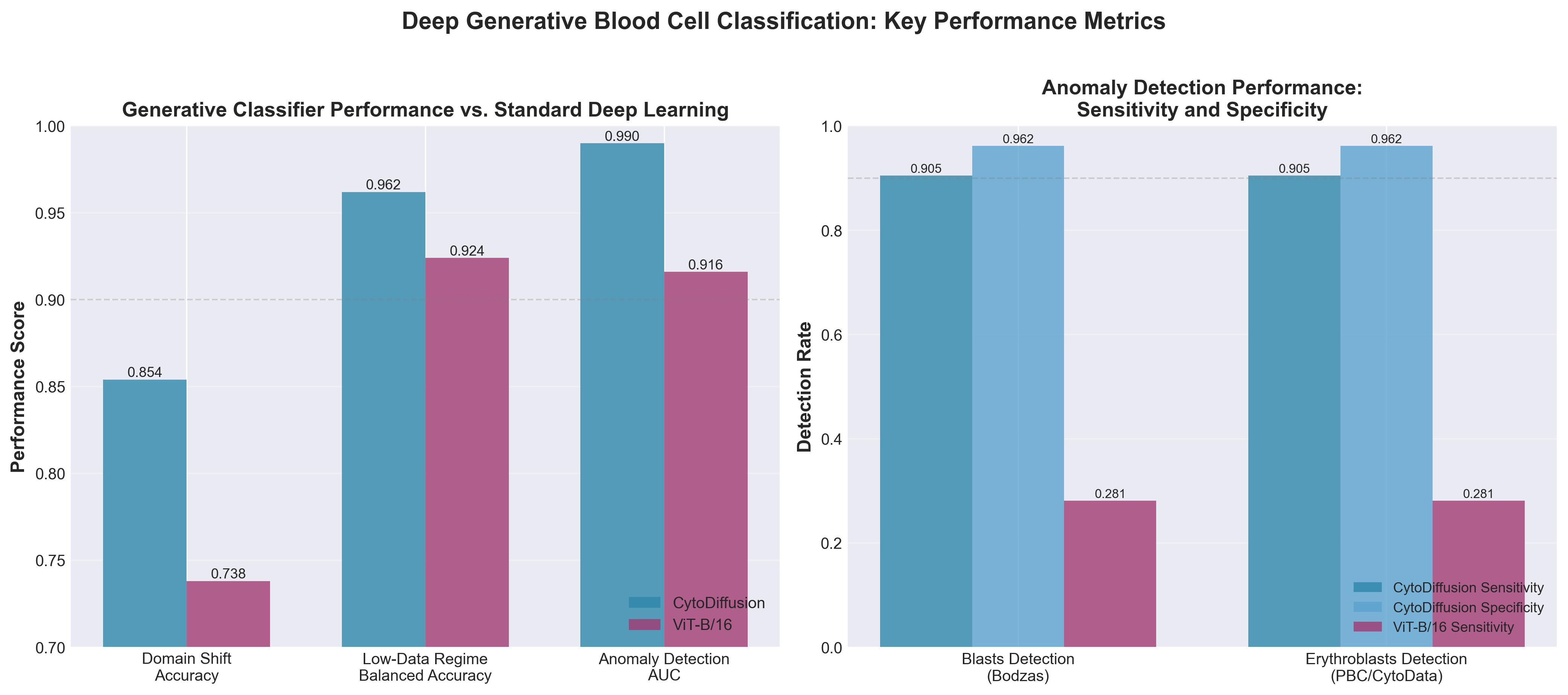
• Generative AI outperforms discriminative models on clinical-critical tasks: CytoDiffusion achieves 0.990 AUC in anomaly detection (vs. 0.916), 0.854 accuracy in domain shift resistance (vs. 0.738), and 0.962 balanced accuracy in low-data scenarios—directly addressing the real-world variability and data scarcity challenges that limit current automated hematology systems’ clinical adoption.
• Synthetic data generation reaches clinical indistinguishability: The model generates blood cell images that expert haematologists cannot reliably distinguish from real images (0.523 accuracy), enabling organizations to overcome data labeling bottlenecks and train downstream applications without extensive manual annotation of rare morphological variants.
• Uncertainty quantification surpasses expert benchmarks: CytoDiffusion provides uncertainty estimates that exceed clinical expert performance, offering actionable confidence levels for diagnostic recommendations and enabling triage of borderline cases—a critical differentiator for clinical laboratory automation and regulatory approval pathways.
5. Matrix mechanical remodeled carrier-free nanosystem for programmable closed-loop reversal of liver fibrosis via STING alkylation
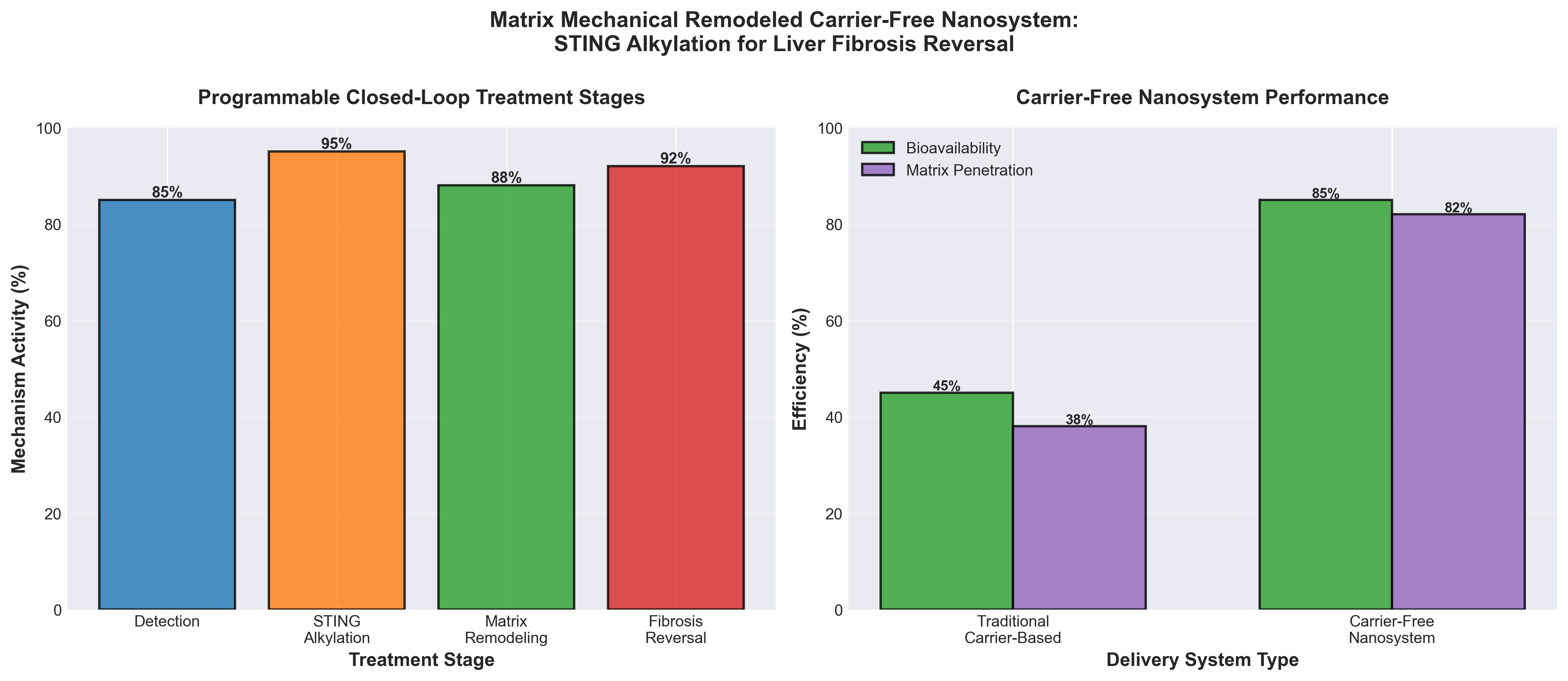
• Novel STING-targeted mechanism for fibrosis reversal: The carrier-free nanosystem uses STING alkylation to achieve programmable, closed-loop reversal of liver fibrosis—a significant departure from conventional approaches that typically focus on halting progression rather than reversing established disease, positioning this as a potential breakthrough therapeutic strategy for advanced liver disease.
• Matrix-remodeling platform technology: The matrix mechanical remodeling approach suggests a multi-functional nanosystem capable of both drug delivery and tissue microenvironment modification, offering potential advantages in biocompatibility and therapeutic efficacy that could be leveraged across other fibrotic diseases (pulmonary, cardiac, renal fibrosis).
• Carrier-free formulation reduces complexity: Eliminating polymer or lipid carriers simplifies manufacturing, reduces potential immunogenicity concerns, and may improve scalability for clinical translation—critical advantages for commercialization timelines and regulatory approval pathways compared to conventional nanoparticle platforms.
Industry News
1. GSK picks Flagship’s Quotient, ProFound as 1st biotech partners for $7B pact
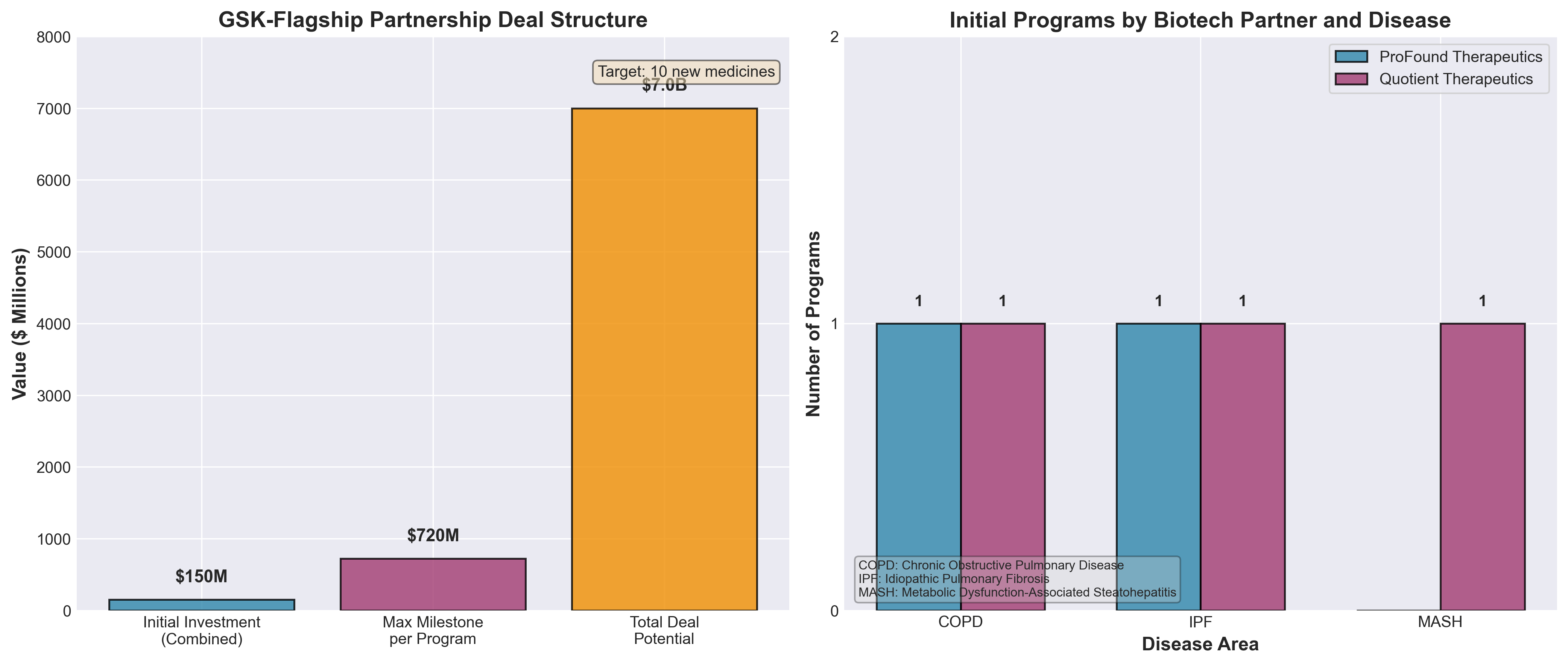
• Milestone-based deal structure de-risks biotech partnerships: GSK’s $7B pact with Flagship allows up to $720M per program (upfront + development + commercial milestones), incentivizing biotechs to hit specific targets rather than relying on upfront capital—a model now standard across Big Pharma-VC collaborations after Pfizer’s 2023 blueprint ($700M per program).
• Proteomics and somatic genomics platforms are becoming core to target discovery: ProFound’s protein detection platform and Quotient’s somatic genomics approach are being deployed across multiple pharma partners (GSK and Pfizer), signaling that these technology platforms have moved beyond exploratory R&D to become validated, productionizable tools for systematic drug target identification.
• Disease focus on respiratory and metabolic dysfunction reflects unmet market needs: GSK’s collaboration targets COPD, IPF, and liver metabolic disease across 5 separate agreements—indicating these indications represent priority pipeline gaps where conventional discovery methods have underperformed, making platform-driven approaches strategically valuable.
2. Arbiter Emerges from Stealth with $52M Funding
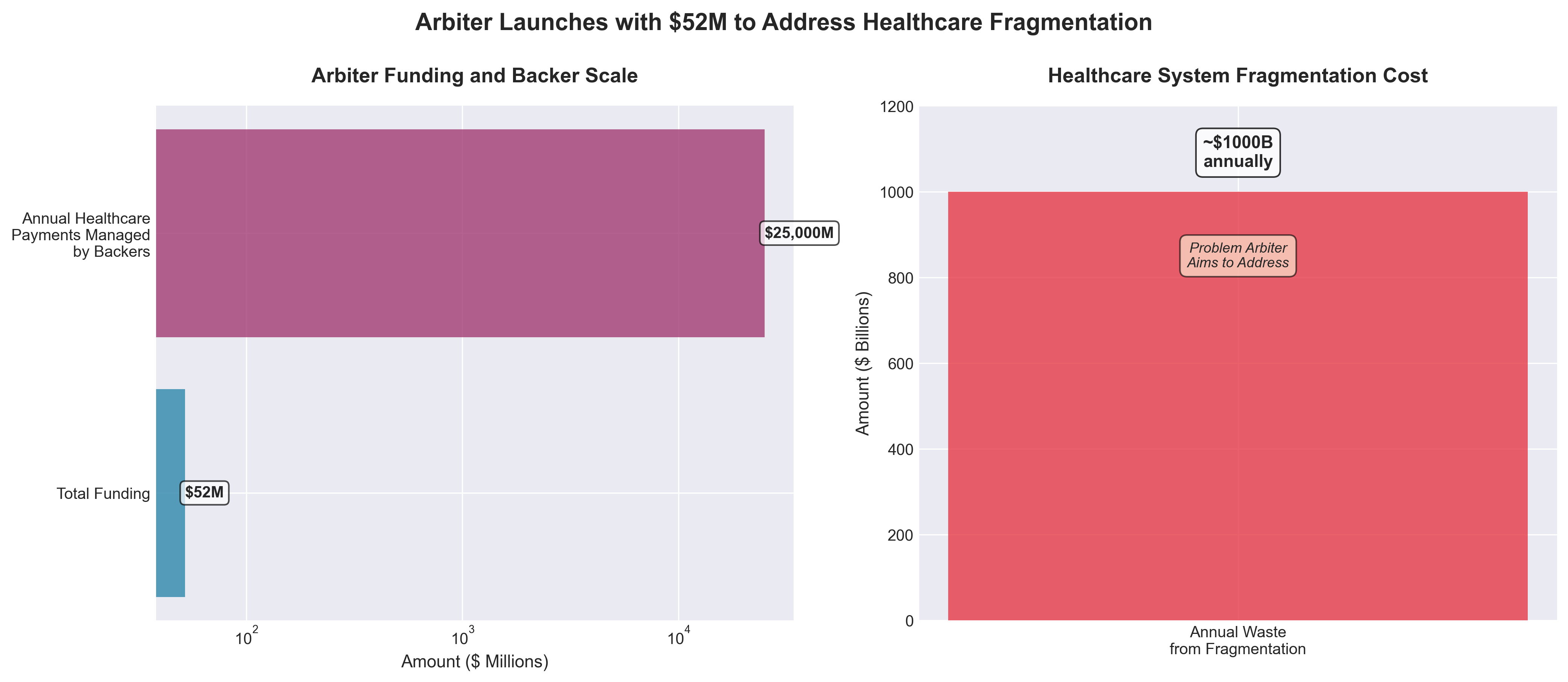
• $1 trillion annual waste opportunity target: Arbiter is addressing healthcare’s costliest inefficiency—fragmentation between payers, providers, and patients—which creates ~$1 trillion in annual waste and delayed care decisions; their Record-Action-Alignment model using AI for real-time site-of-care optimization represents a directly quantifiable ROI lever for health systems and payers.
• Credible insider team with $25B+ payment oversight: Leadership combines C-suite experience from major payers/systems (Cigna, UnitedHealth, Kaiser, VillageMD) with tech talent from Meta, Apple, Google, and Amazon, and collectively oversees $25B+ in annual healthcare spending—reducing execution risk and providing immediate market access and credibility for enterprise adoption.
• Near-term application focus on referral automation: Initial deployment targets real-time referral matching across cost, quality, and availability with automated authorizations and scheduling; this specific use case offers immediate operational efficiency gains and serves as a beachhead for broader care orchestration platform expansion.
3. Philips and Cortechs.ai Expand Partnership to Embed AI Neuroimaging Analytics in MR System
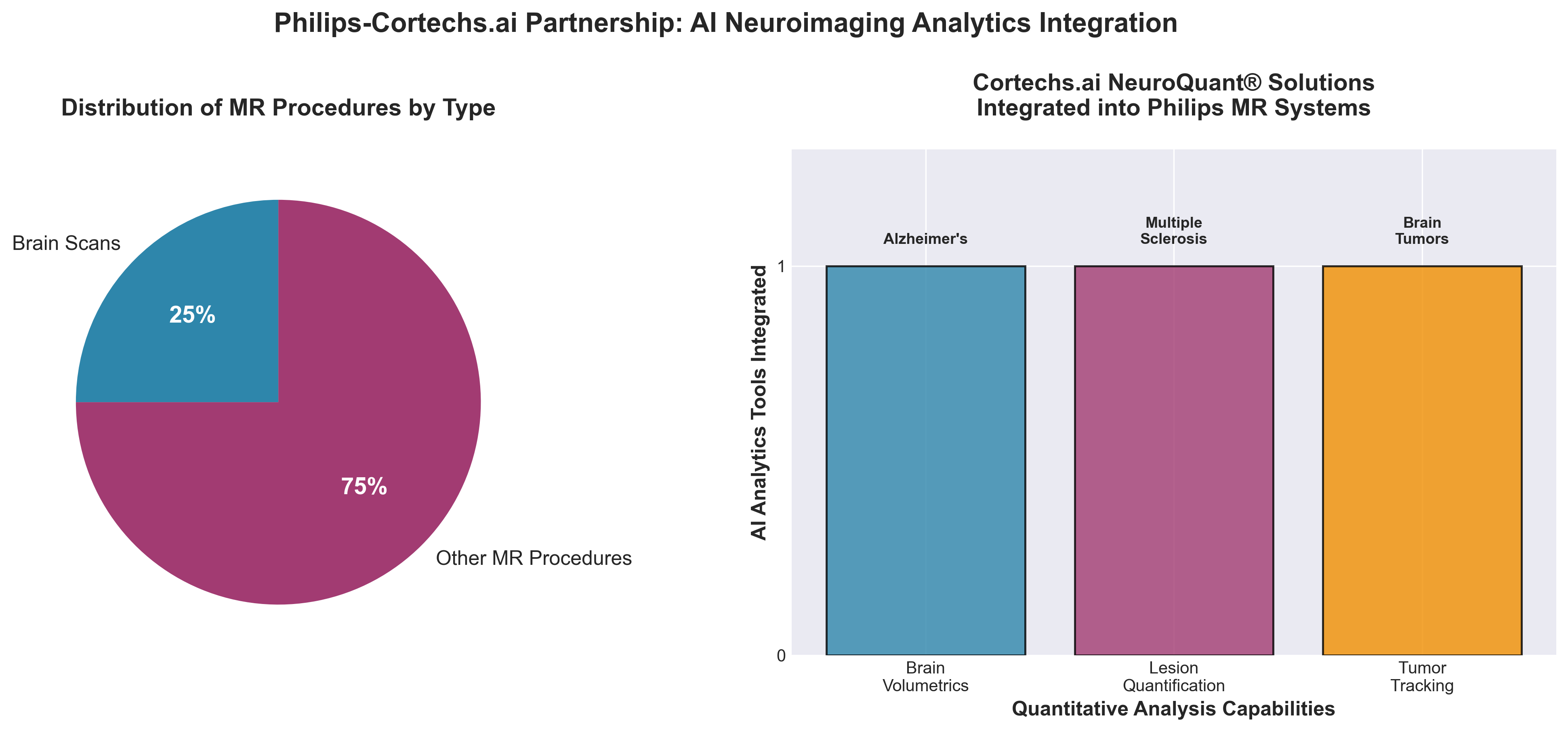
• Workflow Integration Directly on MR Systems Reduces Friction: By embedding Cortechs.ai’s quantitative analytics (brain volumetrics, lesion quantification, tumor tracking) into Philips’ Smart Reading cloud platform with “zero-click” technologist activation, this partnership eliminates post-scan interpretation delays and reduces dependency on expert radiologist time—critical for departments where brain scans represent up to 25% of MR procedure volume.
• Addresses Acute Market Demand Gap: The integration targets a converging crisis—rising neurological conditions (Alzheimer’s, MS, brain tumors) coinciding with radiology staff shortages and subjective visual assessment variability—positioning objective, quantitative AI analytics as a scalable solution to increase diagnostic throughput without proportional staffing increases.
• Standardizes Reproducible Diagnostics Across Institutions: Moving from subjective interpretation to numerical biomarkers within the standard MR workflow enables longitudinal patient monitoring consistency and multi-center clinical trial standardization, particularly valuable for neurodegenerative disease tracking where reproducibility directly impacts treatment decisions and trial outcomes.
4. Atropos Health Integrates Evidence™ Agent with Microsoft Teams
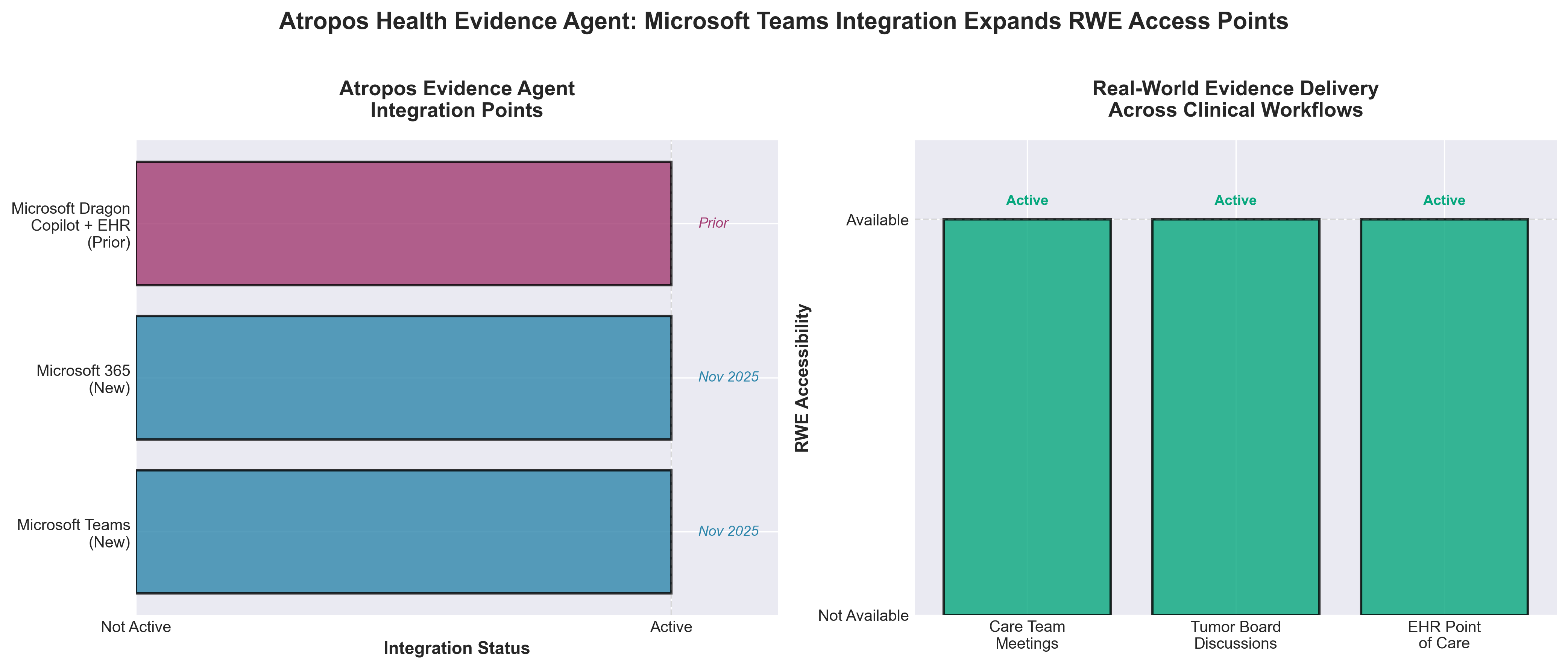
• Workflow Integration as Competitive Advantage: Atropos Health’s direct Microsoft Teams/365 integration eliminates the friction of switching between systems during clinical decision-making—enabling real-time RWE access in multidisciplinary settings (e.g., Tumor Boards) rather than requiring post-meeting research, which directly reduces care delays and decision latency.
• Agentic AI Reshaping Evidence Delivery Model: This represents a shift from passive evidence databases to proactive, on-demand AI agents that generate patient-specific insights at point-of-care; this capability signals a new standard for RWE platforms competing for health system adoption and suggests Microsoft integration will become table-stakes for clinical AI tools.
• Strategic EHR + Meeting Room Convergence: Atropos’s dual strategy—embedding evidence in Dragon Copilot (EHR) and the new Teams agent (care coordination meetings)—creates multiple touchpoints across the clinical workflow; this breadth of integration increases switching costs and positions the company as essential infrastructure rather than a standalone tool.
Clinical Studies & Translational Reports
1. Paradromics’ brain implant could rival Neuralink’s enters clinical trials
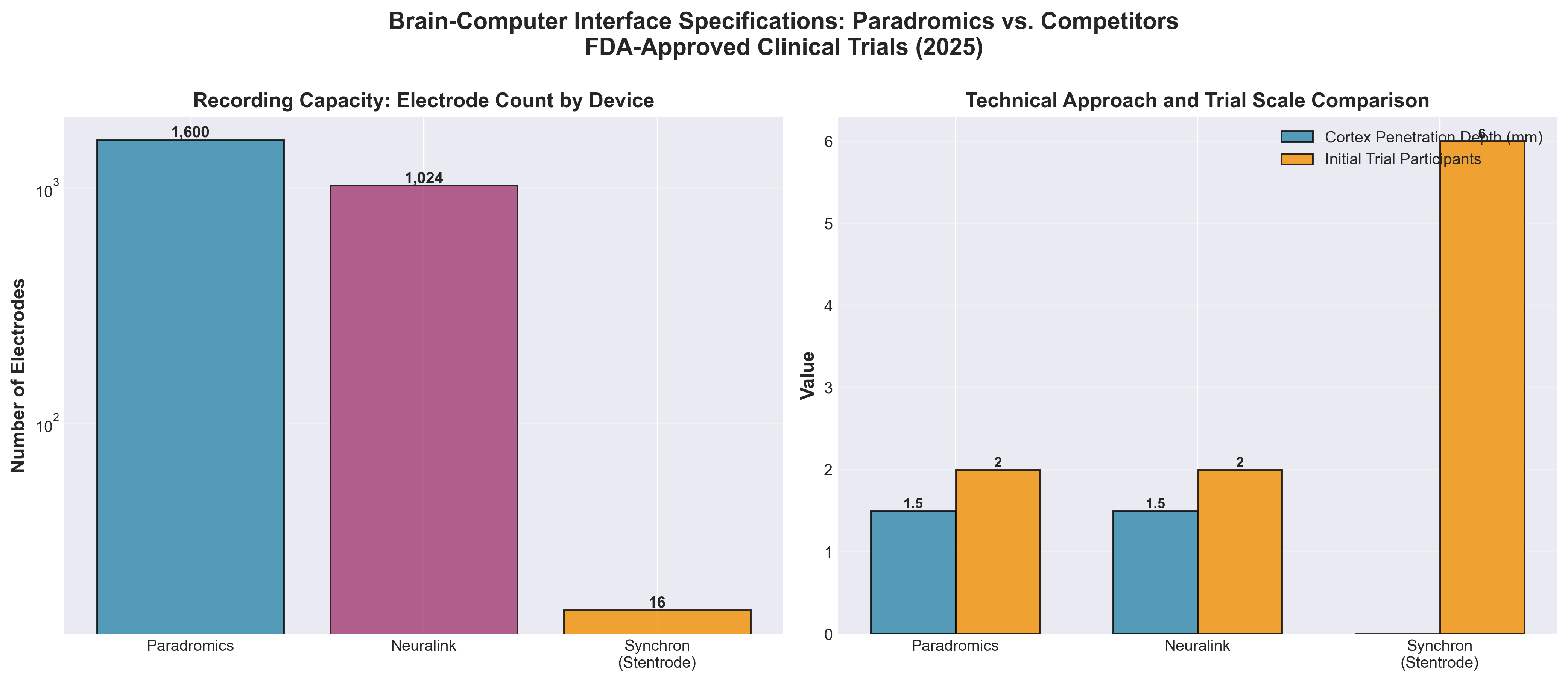
• Direct Competition in Implantable BCI Market: Paradromics’ FDA-approved trial positions it as a credible competitor to Neuralink with a clinically-validated device featuring a 7.5mm electrode array capable of recording from individual neurons at 1.5mm cortical depth—establishing a critical benchmark for the emerging implantable neurotechnology sector.
• Synthetic Speech Generation as Primary Clinical Application: This is the first BCI trial formally targeting real-time voice output using patients’ own voice recordings, addressing the highest immediate quality-of-life need and creating a differentiated value proposition that could accelerate regulatory approval pathways and reimbursement models for speech restoration applications.
• Scalability Path Defined: The trial design explicitly allows expansion from 2 to 10 participants with dual implants to increase signal fidelity, demonstrating a planned route to proof-of-concept that de-risks manufacturing, surgical placement, and signal processing validation before larger-scale commercialization efforts.
2. Factor IX-Padua AAV gene therapy in hemophilia B: phases 1/2 and 3 trials
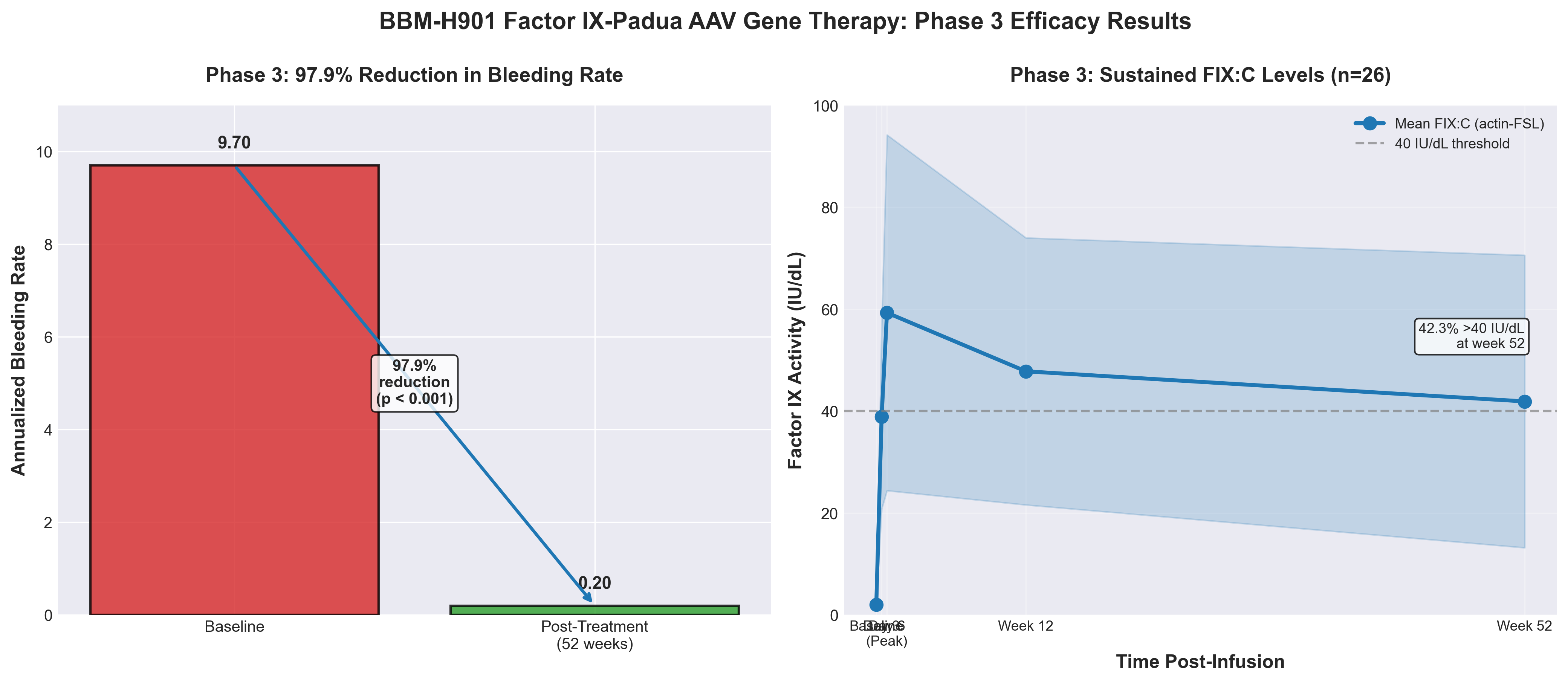
• Significant clinical efficacy with manageable safety profile: Phase 3 data demonstrates 80.8% of hemophilia B patients achieved zero bleeds over 52 weeks with mean ABR of 0.60, meeting superiority margins against prophylactic therapy; transaminitis was the primary adverse event (reversible) with no grade 3+ drug-related AEs, establishing a favorable risk-benefit profile for market differentiation.
• Single-dose durability addresses treatment burden in underserved markets: BBM-H901’s one-time administration model directly challenges the on-demand/prophylactic therapy paradigm in China where most patients lack access to continuous factor replacement, positioning AAV-FIX Padua as a transformative option in cost-constrained healthcare systems and representing untapped commercial expansion opportunities.
• Dose optimization reduces development risk for manufacturers: Establishment of the 5 × 10¹² vg/kg⁻¹ dose as both safe and efficacious de-risks future Phase 3 programs and accelerates regulatory pathways; the consistent transaminitis signal (manageable, non-dose-limiting) provides predictable safety monitoring protocols for comparable AAV gene therapy programs in development.
3. Aspen enriches Parkinson’s cell therapy trial with Kite-backed $115M series C
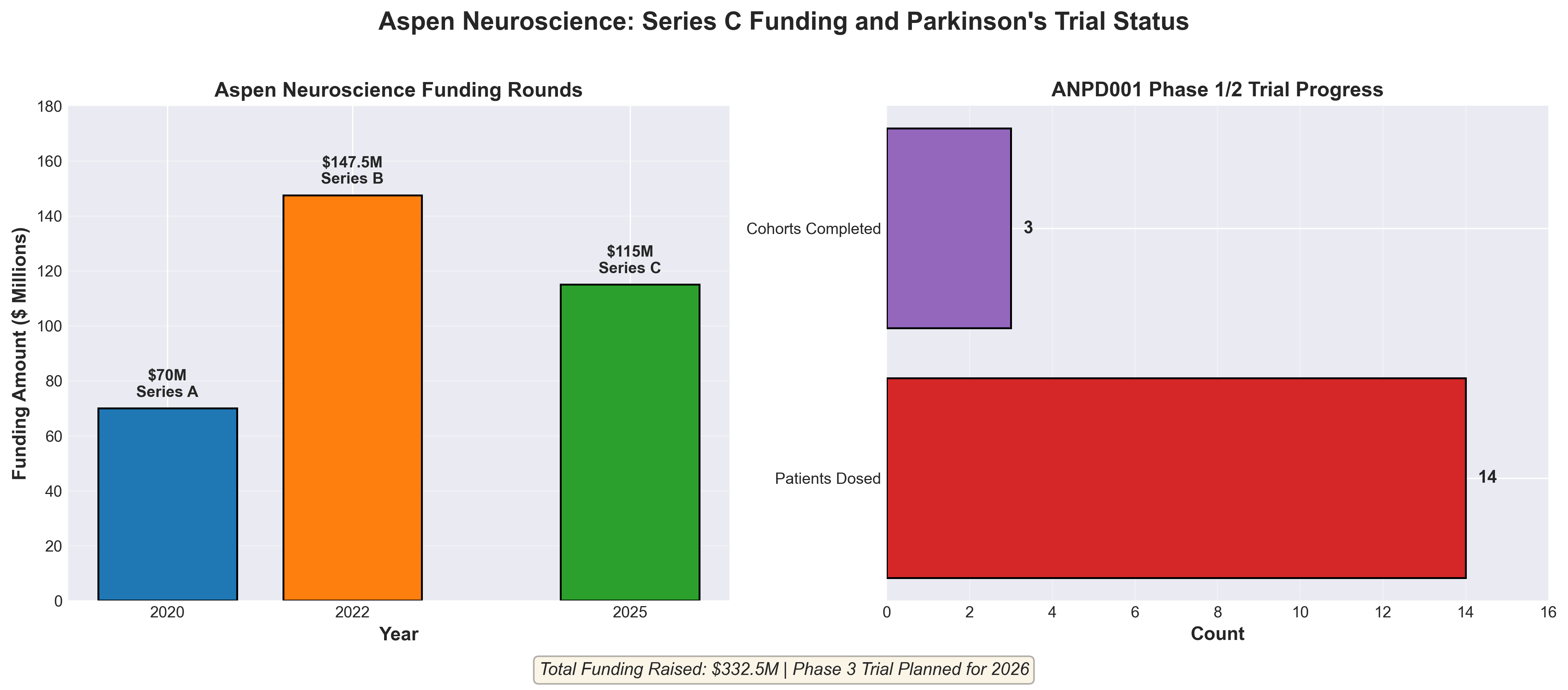
• Accelerated regulatory pathway with manufacturing readiness: Aspen plans to request FDA approval for a direct Phase 3 pivotal trial (targeting H1 2026 conversation), supported by an existing 14,000-sq-ft GMP manufacturing facility already operational since 2022—positioning the company for potential commercialization within 2-3 years if Phase 3 data supports approval.
• Demonstrated clinical efficacy across symptom spectrum: Early Phase 1/2 data (14 dosed patients) shows improvements in both motor and non-motor Parkinson’s symptoms, a rare achievement in the field; upcoming March 2026 data release will be critical for validating this dual-benefit claim and supporting Phase 3 enrollment strategy.
• Strategic manufacturing partnership de-risks scale-up: Kite Pharma’s $115M Series C investment and board seat provides access to Gilead’s autologous cell therapy expertise in manufacturing, clinical development, and commercialization—reducing execution risk and signaling strong M&A acquisition potential given Gilead’s recent cell therapy acquisition track record (Interius BioTherapeutics, August 2024).
4. Efficacy, Immunogenicity, and Safety of Modified mRNA Influenza Vaccine
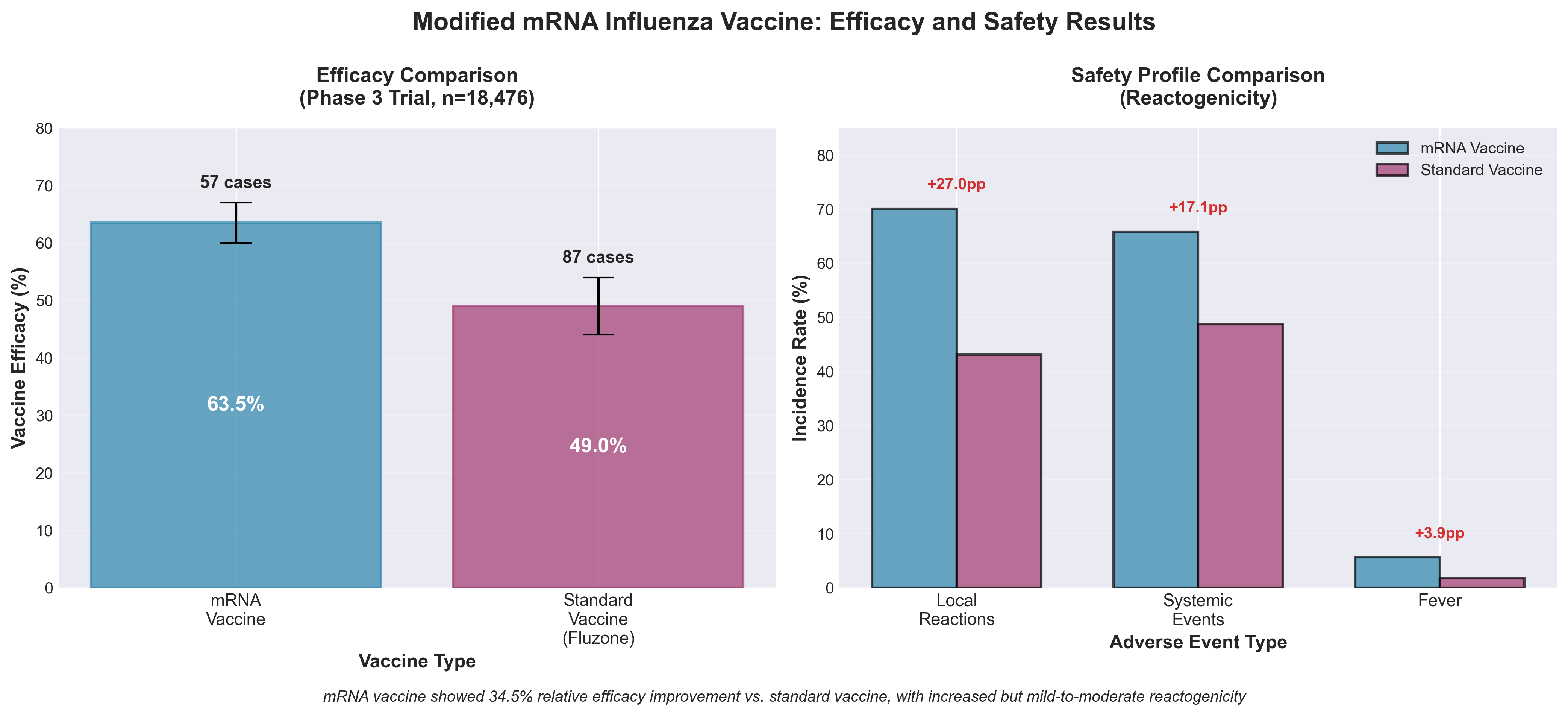
• Pfizer’s mRNA flu vaccine demonstrated 34.5% relative efficacy improvement over standard vaccines (60-67% vs. 44-54% absolute efficacy), representing a meaningful clinical advantage that could shift market positioning and justify premium pricing—critical for biosimilar differentiation in a competitive influenza vaccine market generating $3-4B annually.
• Increased reactogenicity (70.1% local reactions vs. 43.1% for standard vaccine) creates a commercialization risk requiring targeted safety communication strategies—companies must develop clear patient education materials and risk mitigation protocols to prevent adoption hesitation despite superior efficacy, particularly for annual vaccination campaigns.
• The mRNA influenza platform represents a template for rapid vaccine development against seasonal variants, positioning Pfizer and Moderna to potentially consolidate market share as healthcare systems increasingly favor platforms enabling faster strain updates—regulatory pathways for strain-updated mRNA vaccines should be prioritized given cancelled government funding reducing competitive pipeline programs.
5. Merck KGaA links up with Flagship’s Valo Health in search for Parkinson’s targets, offers $3B in biobucks
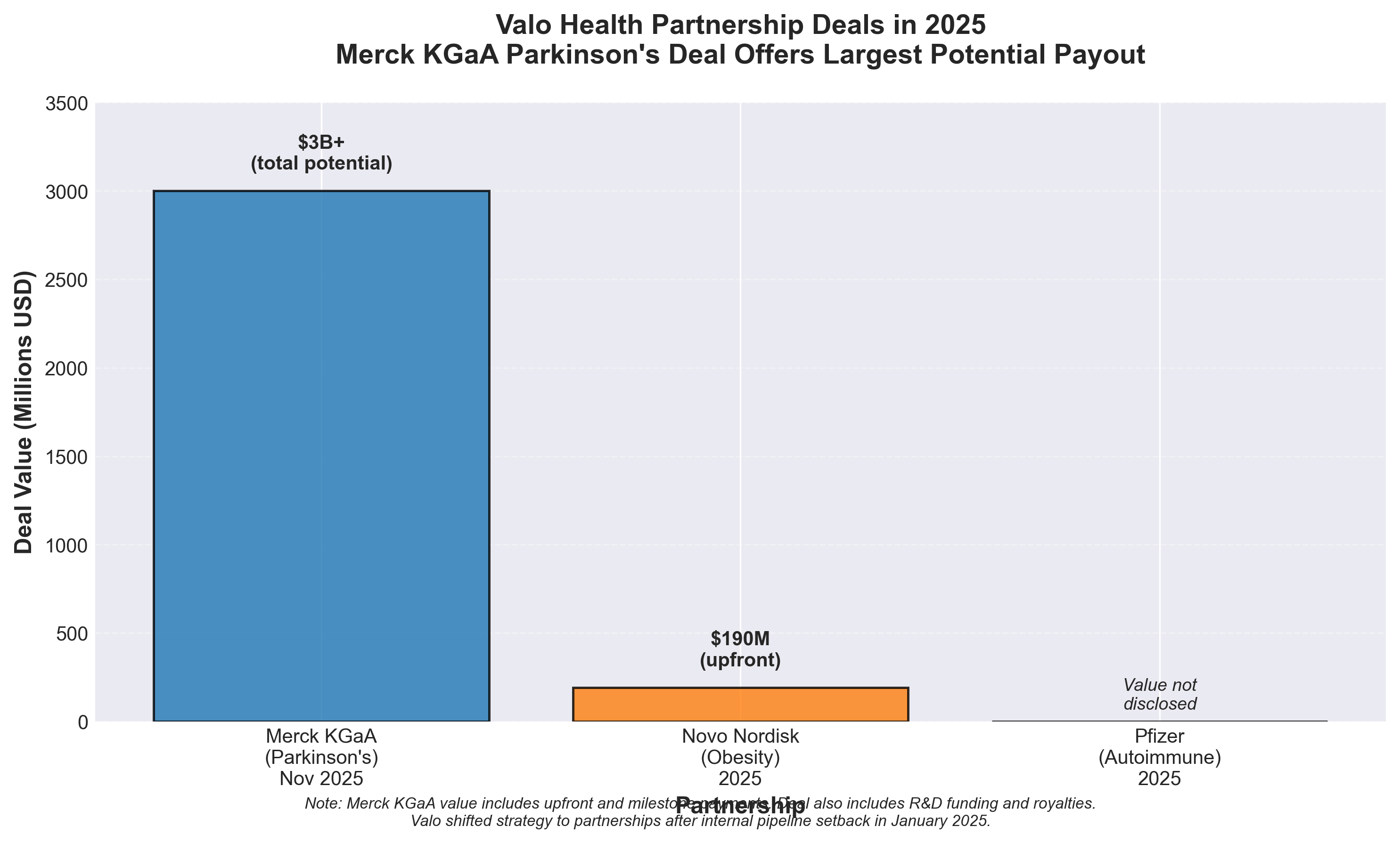
• Data-driven target discovery is becoming a competitive moat: Valo’s ability to leverage real-world human biology data to identify patient populations and unmet needs is attracting multiple $billion+ partnerships (Merck, Novo Nordisk, Pfizer), signaling that Big Pharma now values AI-enabled patient stratification and target validation as highly as traditional drug discovery capabilities.
• Partnership revenue model masks internal pipeline weakness: Valo’s shift to licensing its discovery platform ($3B+ deal values) after its own pipeline stumbled (diabetic retinopathy failure) demonstrates a viable pivot for biotech platforms—outsourcing drug development while monetizing data/AI expertise through upfront payments, milestones, and royalties rather than clinical execution.
• CRO integration accelerates go-to-market for discovery platforms: Charles River’s co-development of Valo’s platform and subsequent adoption by multiple pharma partners (Lundbeck, Flagship portfolio companies) shows that embedding discovery tools within established CRO infrastructure rapidly scales platform adoption and reduces friction for enterprise clients.