Headlines
Clinical Studies & Translational Reports
- Novo Nordisk’s semaglutide misses in closely-watched Alzheimer’s trials
- J&J’s $5B Alzheimer’s hope fades as anti-tau antibody posdinemab flops in phase 2
- Bayer’s asundexian cuts stroke risk in phase 3, reviving hopes for FXIa class
- Kelun’s TROP2 ADC plus Keytruda improves outcomes in first-line lung cancer
- A therapeutic peptide vaccine for fibrolamellar hepatocellular carcinoma: a phase 1 trial
New Research
- Open sourcing of ultrasound resources, datasets and deep learning models
- Engineered acoustic reporters for simultaneous multivariate ultrasonography tracking
- Scalable spatial single-cell transcriptomics and translatomics in 3D thick tissue blocks
- TIRTL-seq: deep, quantitative and affordable paired TCR repertoire sequencing
- Helixer: ab initio prediction of primary eukaryotic gene models using deep learning
Industry News
- Versant’s Dayra dawns with $50M Biogen pact, oral macrocyclic peptide mission
- Craig Crews, Mikael Dolsten raise $32M for Quarry Thera
- Solventum to buy Acera Surgical for up to $850M, repurchase $1B in stock
Clinical Studies & Translational Reports
1. Novo Nordisk’s semaglutide misses in closely-watched Alzheimer’s trials
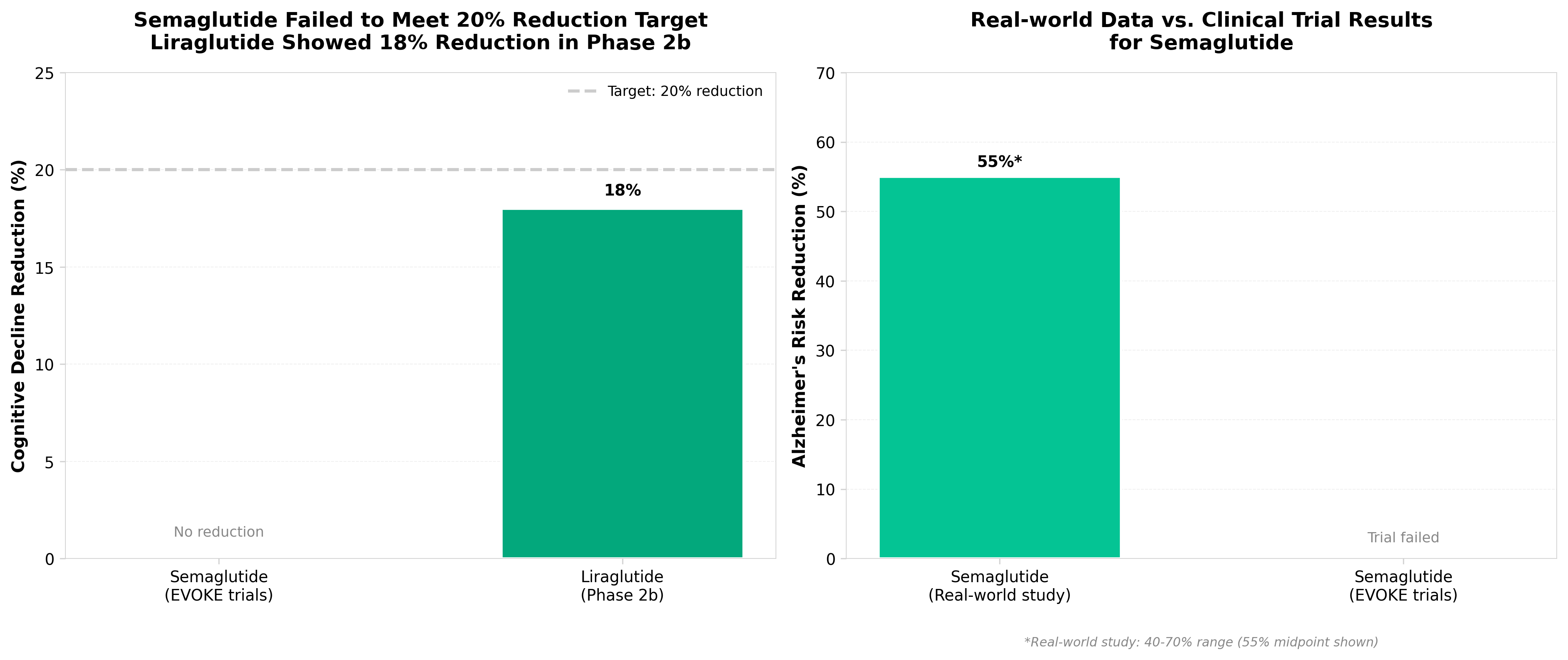
• GLP-1 class expansion into CNS indications faces significant validation challenges. Novo Nordisk’s failure in Alzheimer’s suggests the GLP-1 mechanism may not translate broadly beyond metabolic disorders, requiring industry to reassess pipeline programs targeting neurodegeneration and establish more rigorous early biomarker endpoints before advancing to Phase 3.
• De-risking strategy shifts focus back to core metabolic/obesity markets. This setback reinforces that GLP-1 players should prioritize resource allocation toward obesity, diabetes, and cardiovascular indications where efficacy is proven, rather than pursuing speculative neurology applications that consume capital without clear mechanistic validation.
2. J&J’s $5B Alzheimer’s hope fades as anti-tau antibody posdinemab flops in phase 2
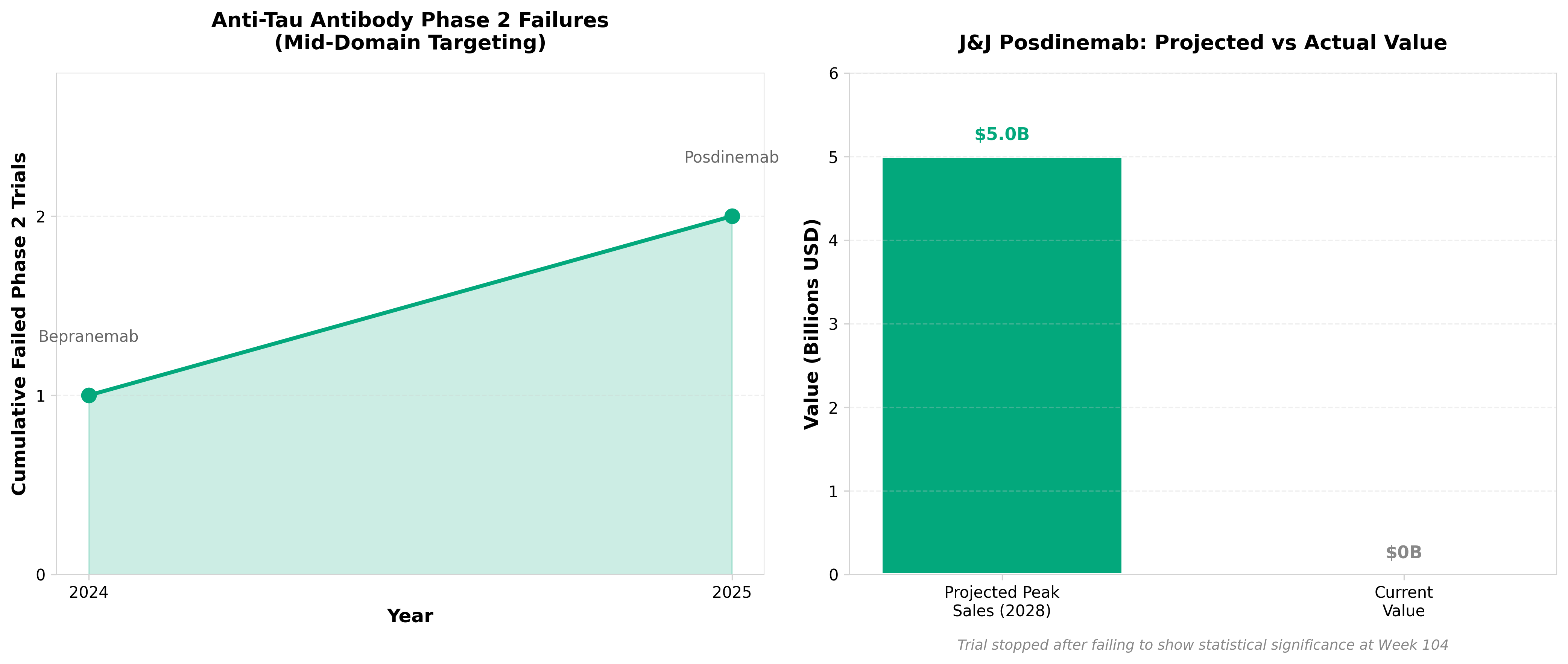
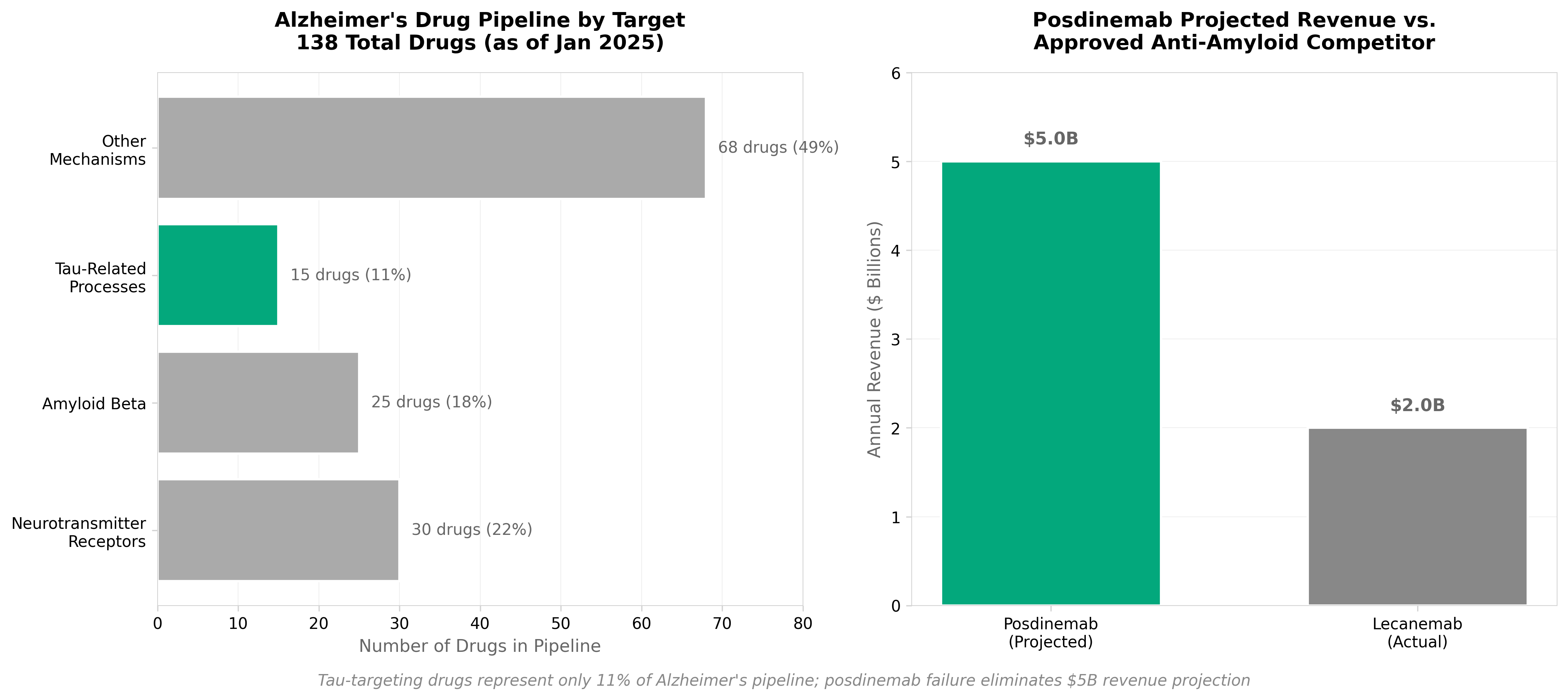
• Anti-tau antibody strategy faces validation challenges: Posdinemab’s phase 2 failure questions the viability of tau-targeting monoclonal antibodies as a standalone Alzheimer’s therapeutic approach, potentially redirecting R&D investment toward combination therapies or alternative tau modulation mechanisms.
• $5B revenue projection eliminated from pipeline: J&J’s decision to halt the program removes a significant near-term commercial asset, signaling biotech companies to reassess valuation models for tau-focused programs and consider earlier-stage biomarker-driven patient selection to de-risk late-stage failures.
• Tau hypothesis validation still incomplete: Despite prior amyloid successes, this setback demonstrates tau pathology may require different mechanistic approaches (e.g., tau aggregation inhibitors, phosphorylation modulators) or biomarker-stratified populations rather than broad monoclonal antibody targeting.
3. Bayer’s asundexian cuts stroke risk in phase 3, reviving hopes for FXIa class
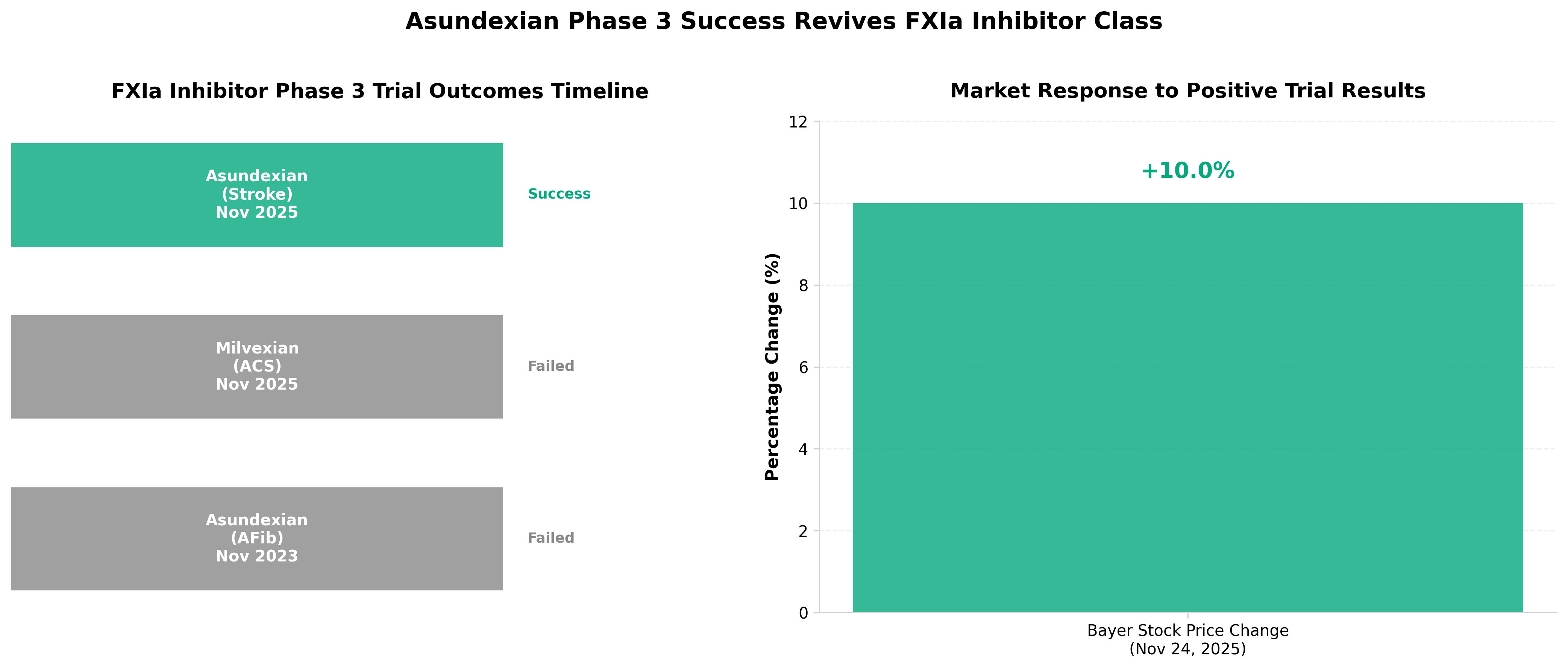
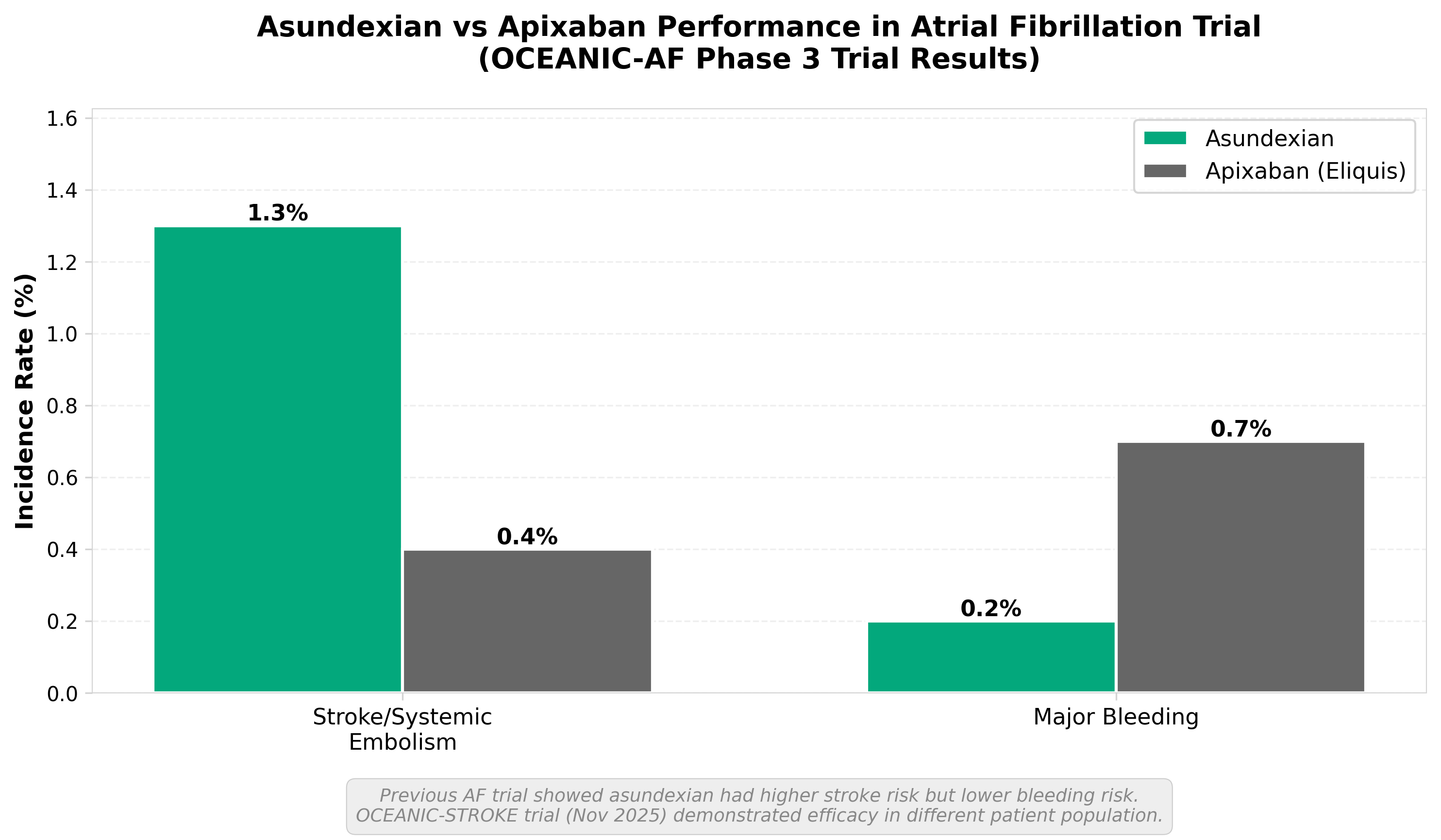
• FXIa inhibitor class validation restores investor confidence: Asundexian’s positive phase 3 results reverse the mechanism’s recent credibility challenges, potentially unblocking development pipelines for competitors (Ionis, Eli Lilly) and signaling renewed commercial viability for anticoagulants targeting this pathway.
• Stroke prevention market opportunity remains substantial: Success in reducing stroke risk positions asundexian as a potential alternative to existing anticoagulants, addressing a large patient population and creating differentiation opportunities if the safety profile proves favorable compared to warfarin/NOACs.
4. Kelun’s TROP2 ADC plus Keytruda improves outcomes in first-line lung cancer
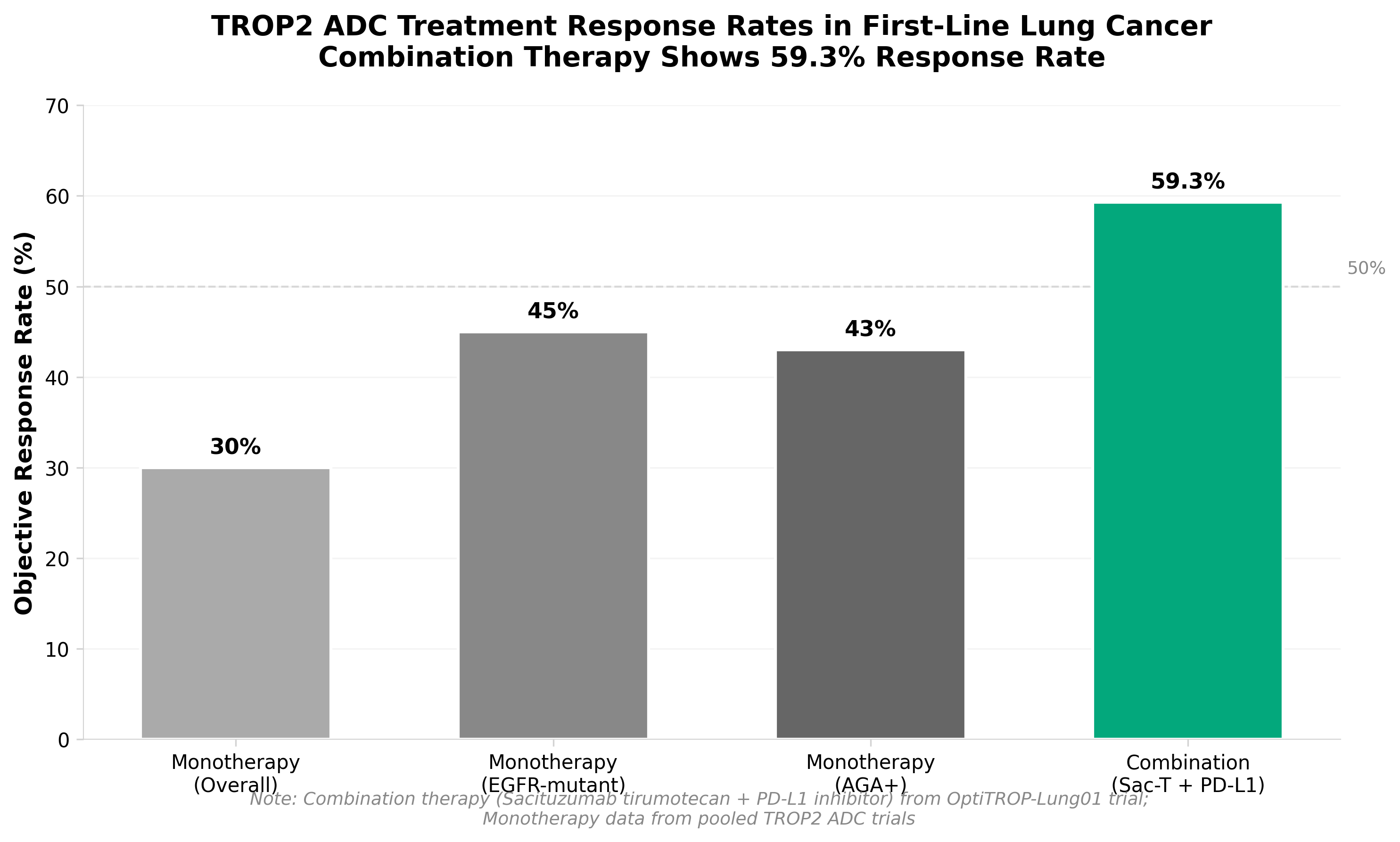
• First-line lung cancer positioning creates competitive advantage: Kelun’s TROP2 ADC combination with Keytruda achieving Phase 3 success in first-line setting positions this therapy ahead of many competitors still in earlier development stages, enabling rapid regulatory filing in China and potential market entry within 1-2 years.
• ADC+checkpoint inhibitor combination validates emerging therapeutic paradigm: This win reinforces the clinical viability of pairing antibody-drug conjugates with PD-1/PD-L1 inhibitors in solid tumors, likely accelerating investment and development timelines for similar combination approaches across oncology pipelines.
• Chinese regulatory pathway acceleration opportunity: Filing with Chinese authorities (NMPA) rather than waiting for global trials may enable faster market access and revenue generation for Kelun, while reducing capital requirements and establishing market precedent in the world’s second-largest pharmaceutical market.
5. A therapeutic peptide vaccine for fibrolamellar hepatocellular carcinoma: a phase 1 trial
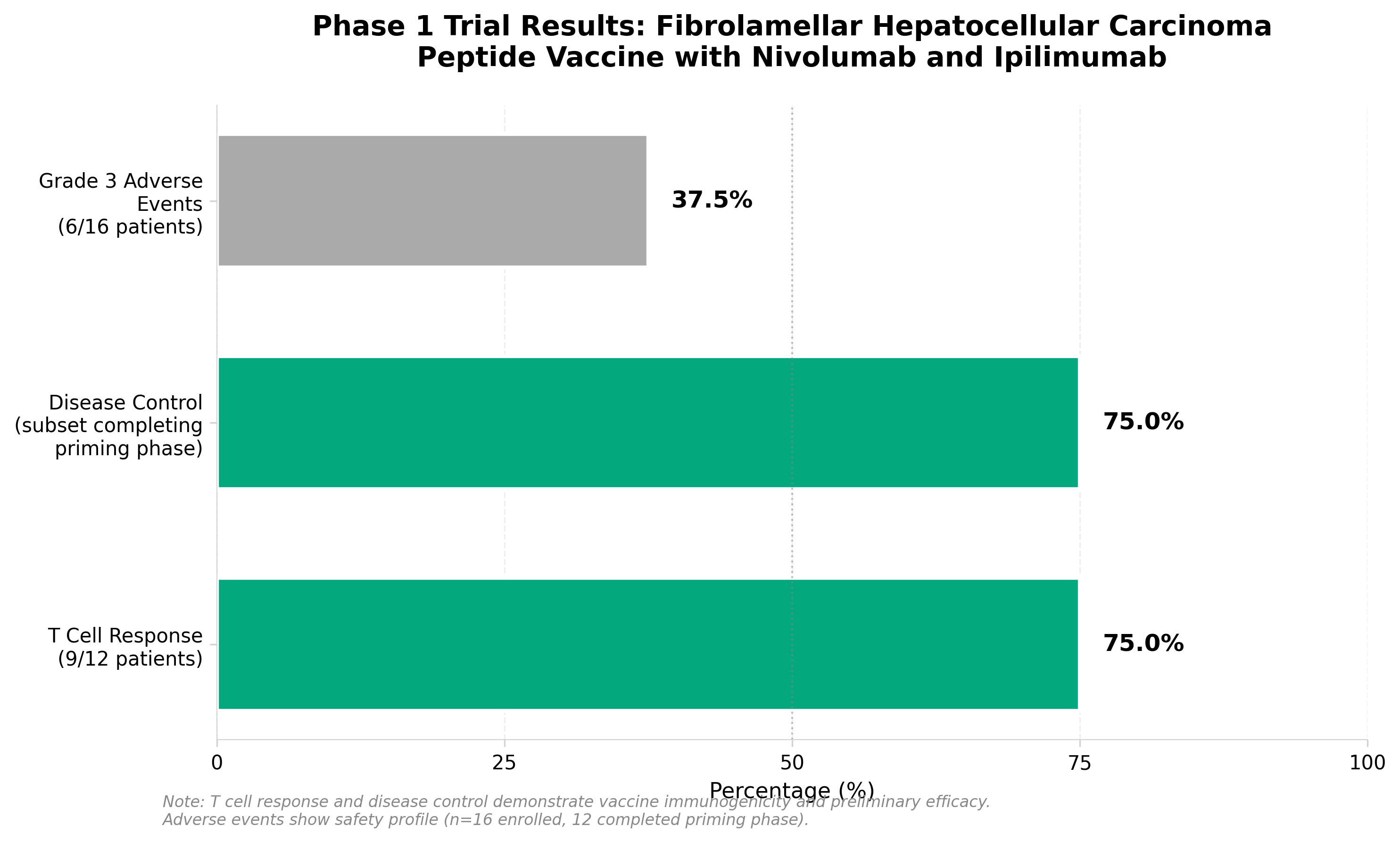
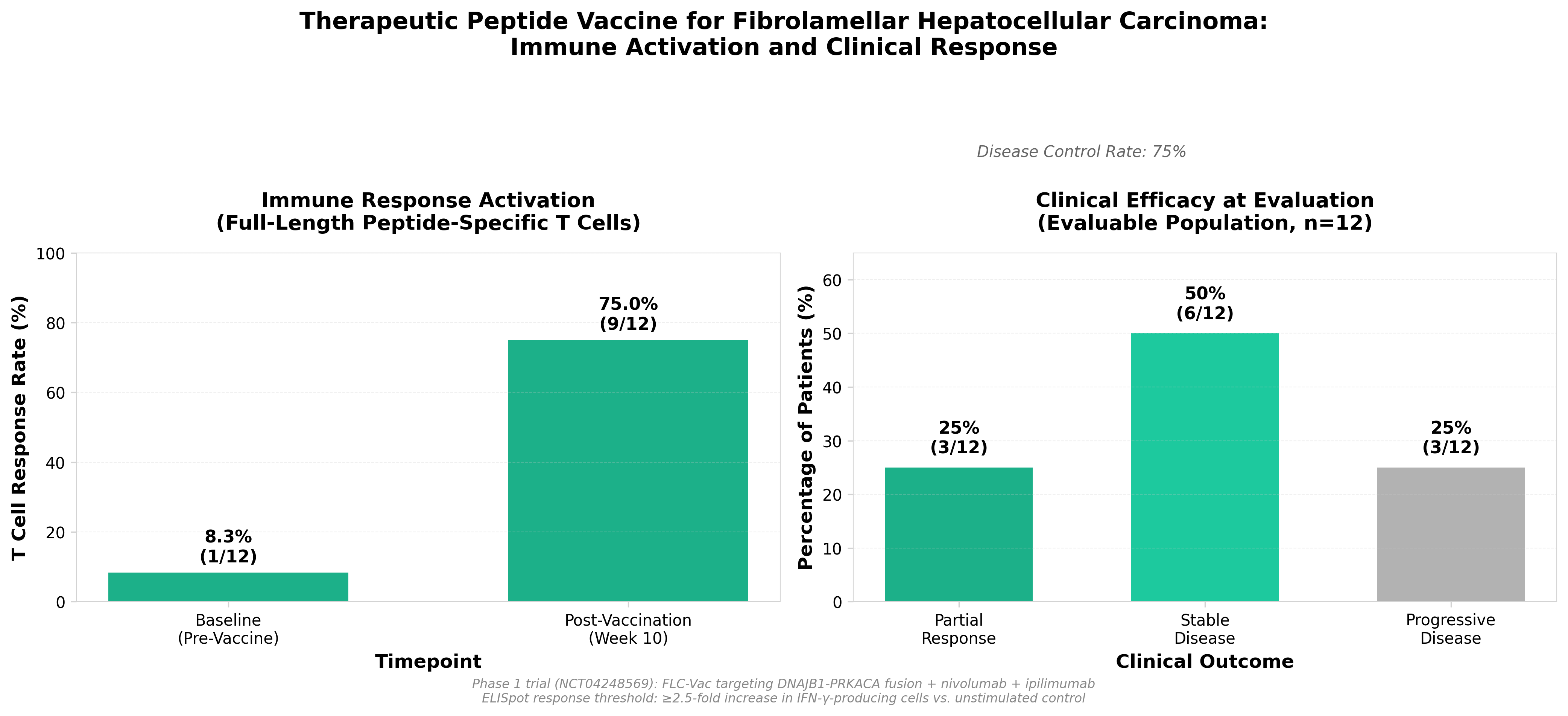
• Disease-specific fusion antigen validation: Targeting the DNAJB1–PRKACA fusion kinase demonstrates viability of driver mutation-specific immunotherapy in rare cancers, establishing a template for therapeutic peptide vaccines against other defined fusion oncoproteins.
• Checkpoint inhibitor combination efficacy: The safety and preliminary clinical responses achieved with the peptide vaccine plus nivolumab/ipilimumab combination validates a synergistic approach worth exploring for other neoantigen-directed therapies, potentially expanding the addressable market beyond monotherapy limitations.
• Phase 1 immunogenicity confirmation: Translational evidence of activated immune responses provides mechanistic proof-of-concept necessary for designing Phase 2 efficacy trials and justifies investment in similar personalized/targeted immunotherapy platforms for molecularly-defined malignancies.
New Research
1. On the public dissemination and open sourcing of ultrasound resources, datasets and deep learning models
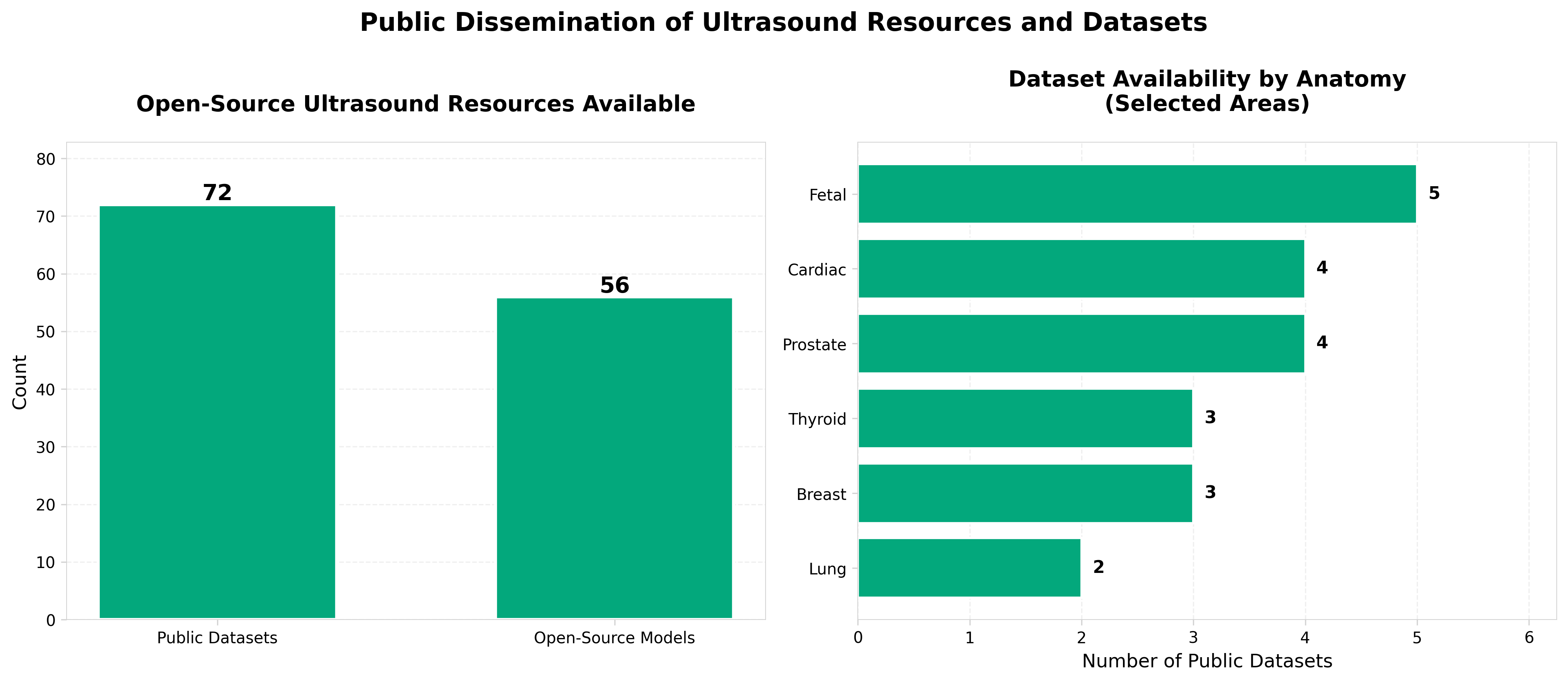
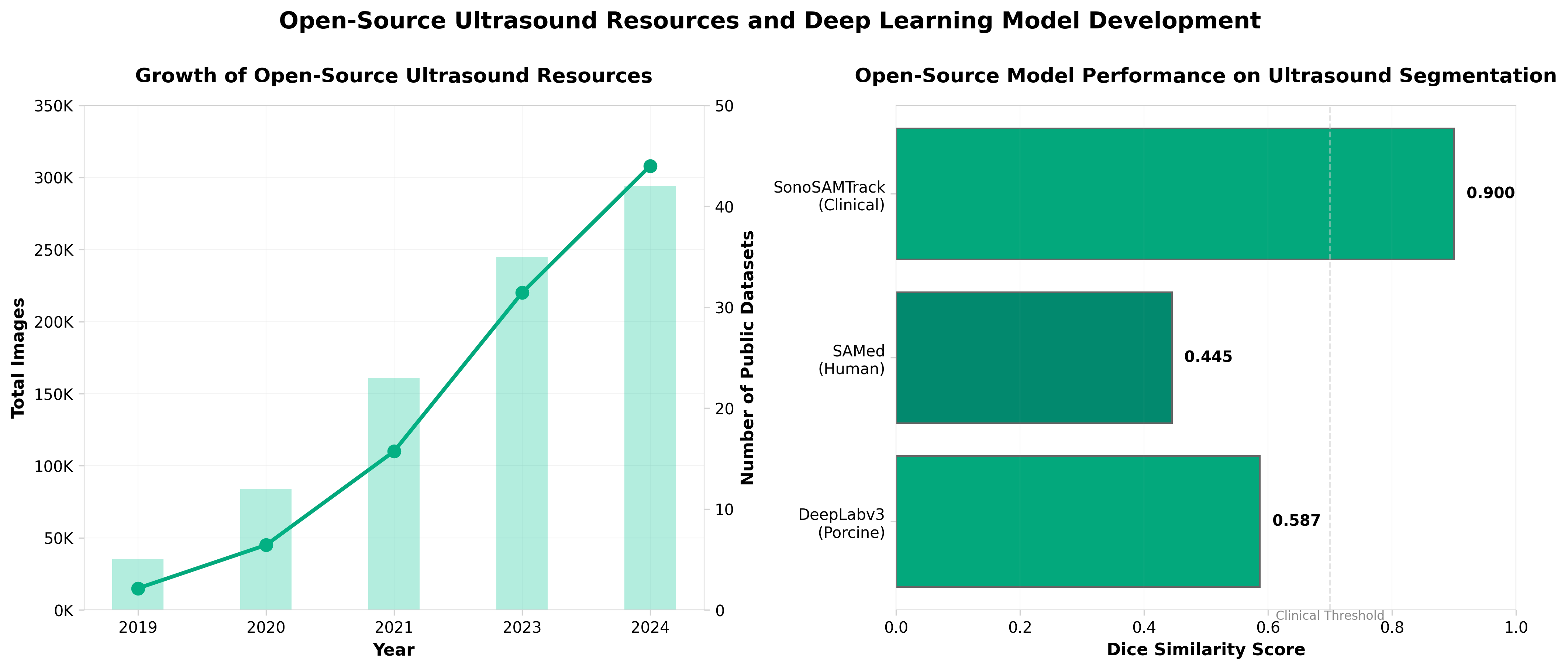
• Open-source ultrasound models reduce development barriers and accelerate clinical adoption. Publicly available datasets and pre-trained deep learning models lower the technical and financial barriers for healthcare organizations to implement AI-assisted ultrasound, potentially expanding diagnostic capabilities across resource-limited settings and smaller clinical facilities.
• Standardized, shareable ultrasound resources enable faster validation and regulatory pathway clarity. Collaborative access to benchmark datasets and model architectures facilitates multi-institutional validation studies, which strengthens evidence for FDA/regulatory submissions and reduces duplicative development efforts across the industry.
• Public dissemination strategies address reproducibility and model generalization challenges in ultrasound AI. Open-sourcing enables the research community to identify and mitigate bias and performance variability across different ultrasound equipment, populations, and clinical contexts—critical for building robust, deployable diagnostic tools.
2. Engineered acoustic reporters for simultaneous tracking of two variables on ultrasonography
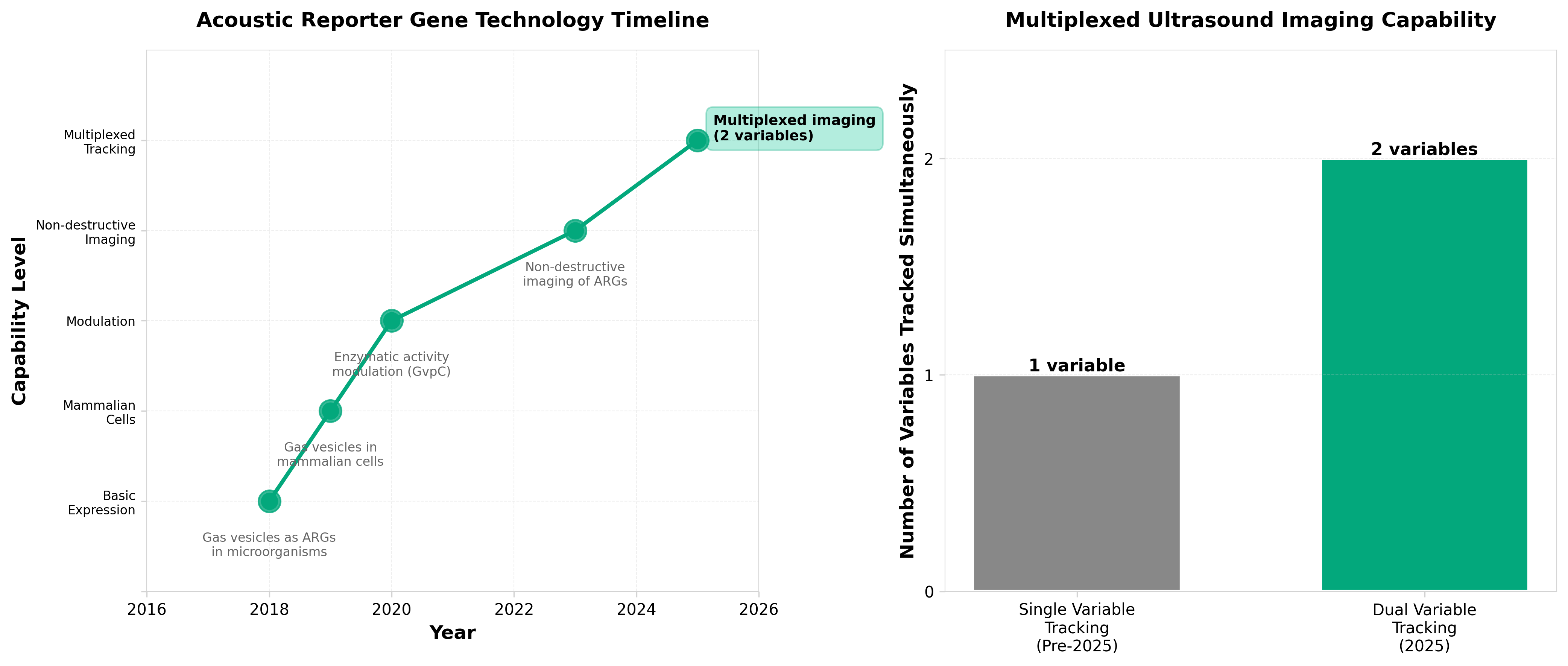
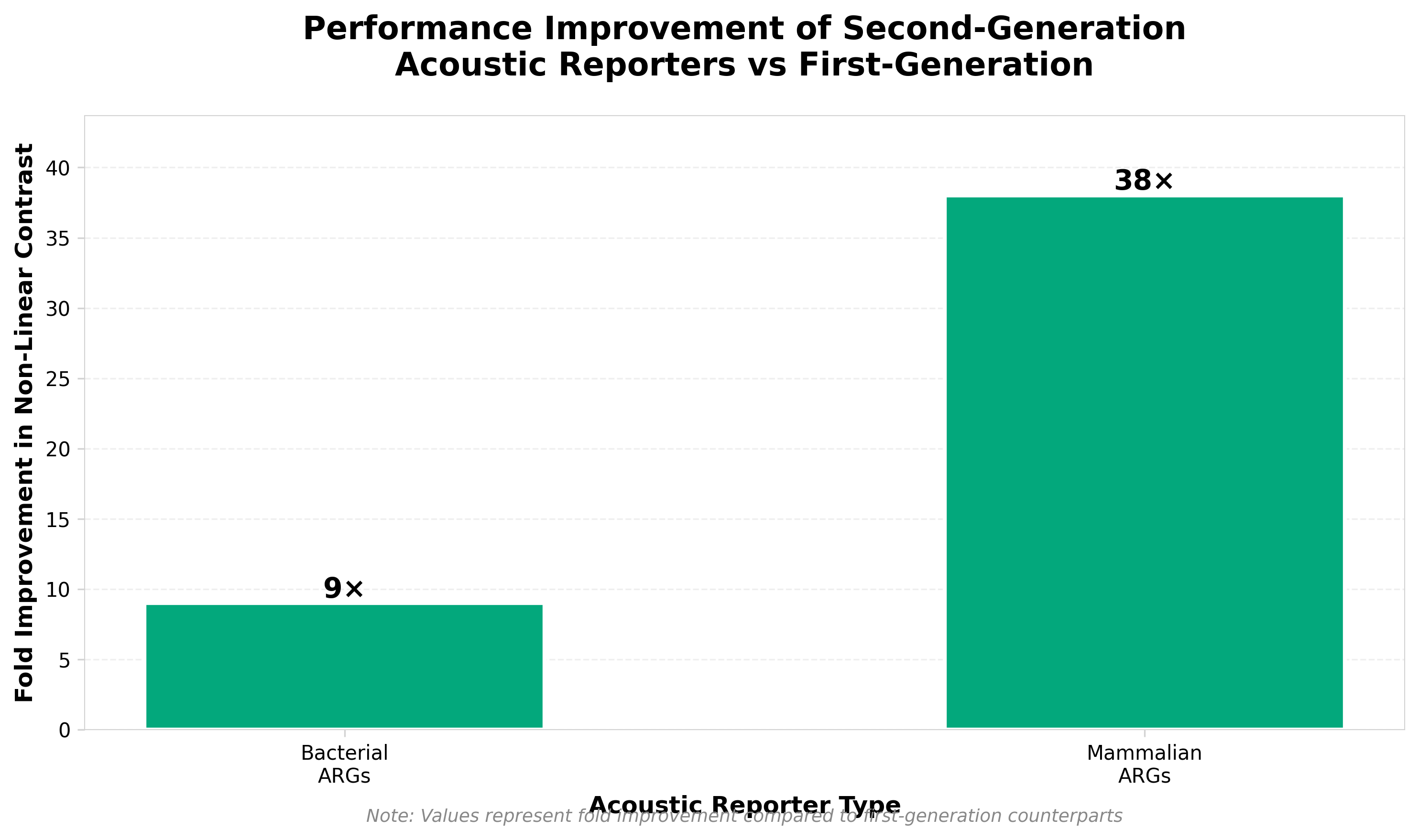
• Multiplexed ultrasound imaging capability: Engineered acoustic reporter genes with differential pressure responses enable simultaneous tracking of two distinct variables in deep tissue, overcoming a key limitation of current ultrasound imaging that typically visualizes only single biological parameters at a time.
• Non-invasive deep-tissue monitoring in opaque tissues: This technology provides real-time visualization of different cell types within optically opaque living tissues without requiring optical access, addressing a significant gap in current imaging modalities for in vivo research and potentially expanding ultrasound applications beyond traditional structural imaging to functional cellular tracking.
• Platform for multiplexed gene expression monitoring: The differential acoustic reporter system establishes a foundation for engineering additional variants with distinct pressure responses, enabling future development of multi-parameter imaging panels that could track complex biological processes (e.g., multiple cell populations, temporal gene expression patterns) in preclinical models and therapeutic applications.
3. Scalable spatial single-cell transcriptomics and translatomics in 3D thick tissue blocks
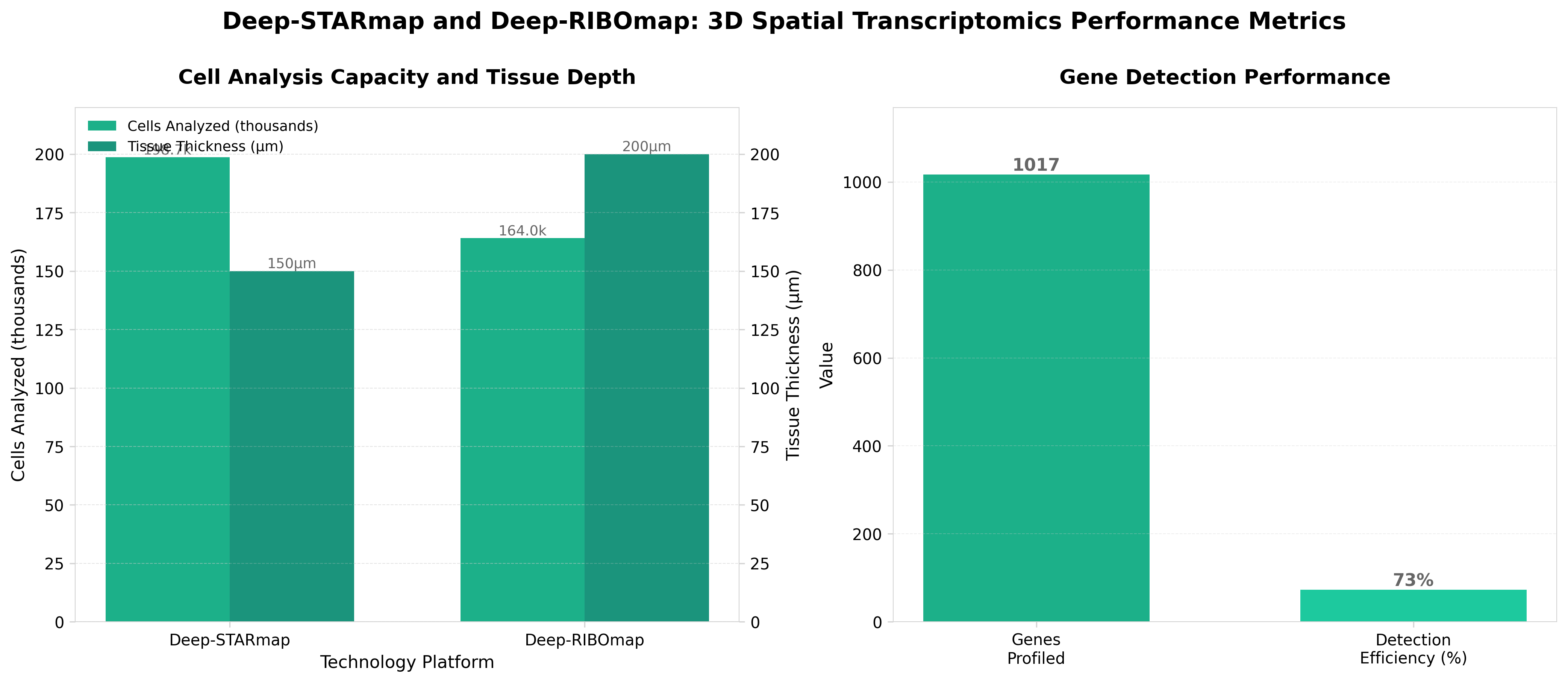
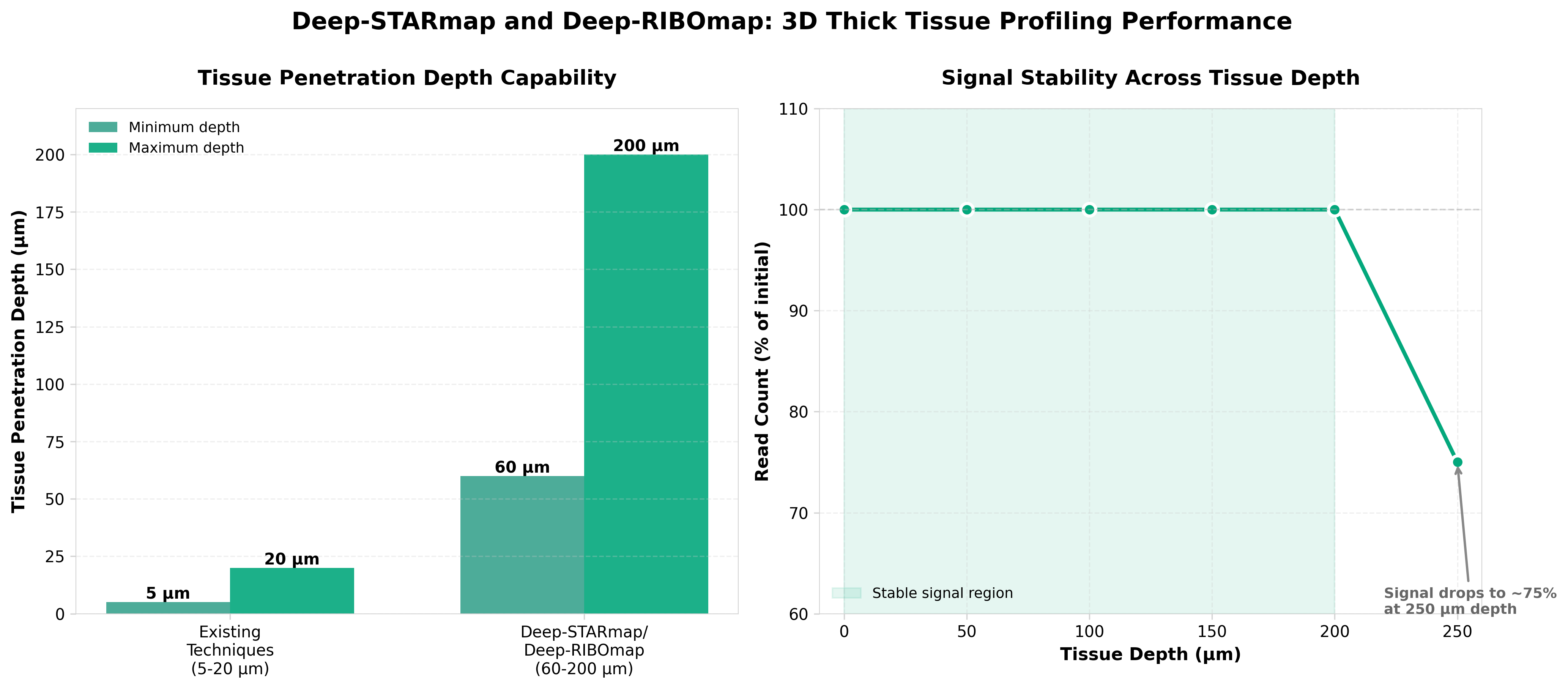
• 3D tissue profiling overcomes previous technical limitations: Deep-STARmap and Deep-RIBOmap enable simultaneous transcriptomic and translatomic analysis in thick tissue blocks, addressing the long-standing challenge of studying gene expression in native 3D tissue architecture rather than thin sections, which should accelerate disease mechanism discovery in complex tissues.
• Dual imaging-based sequencing approach provides integrated biological insights: By combining transcriptional (Deep-STARmap) and translational (Deep-RIBOmap) data in single experiments, researchers can now directly correlate mRNA presence with protein synthesis rates, revealing post-transcriptional regulation that single-modality approaches miss and enabling more accurate biomarker identification for drug development.
• Scalability positions this as a practical platform for routine research and clinical applications: The imaging-based sequencing design allows processing of entire tissue blocks rather than isolated cells, potentially reducing sample preparation artifacts and enabling high-throughput spatial analysis across multiple samples for translational research and precision medicine workflows.
4. TIRTL-seq: deep, quantitative and affordable paired TCR repertoire sequencing
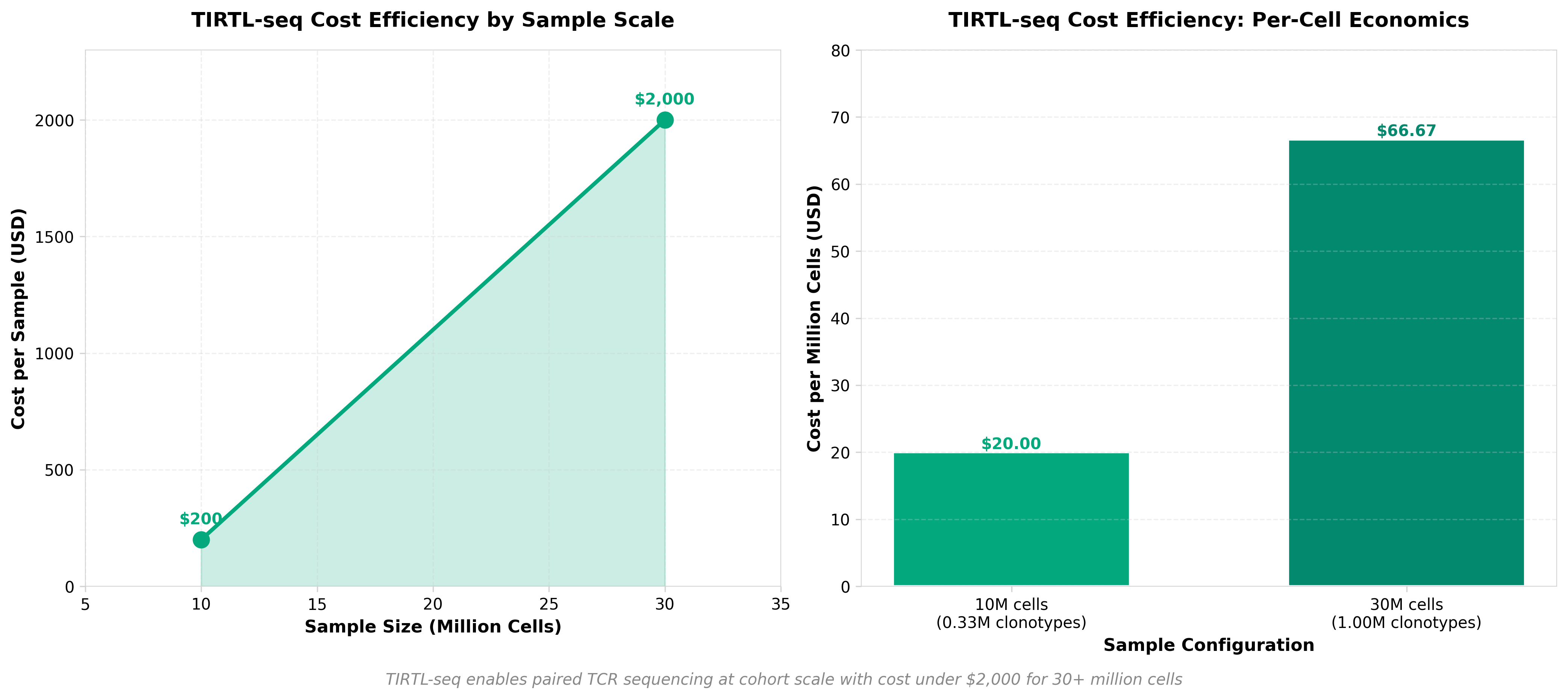
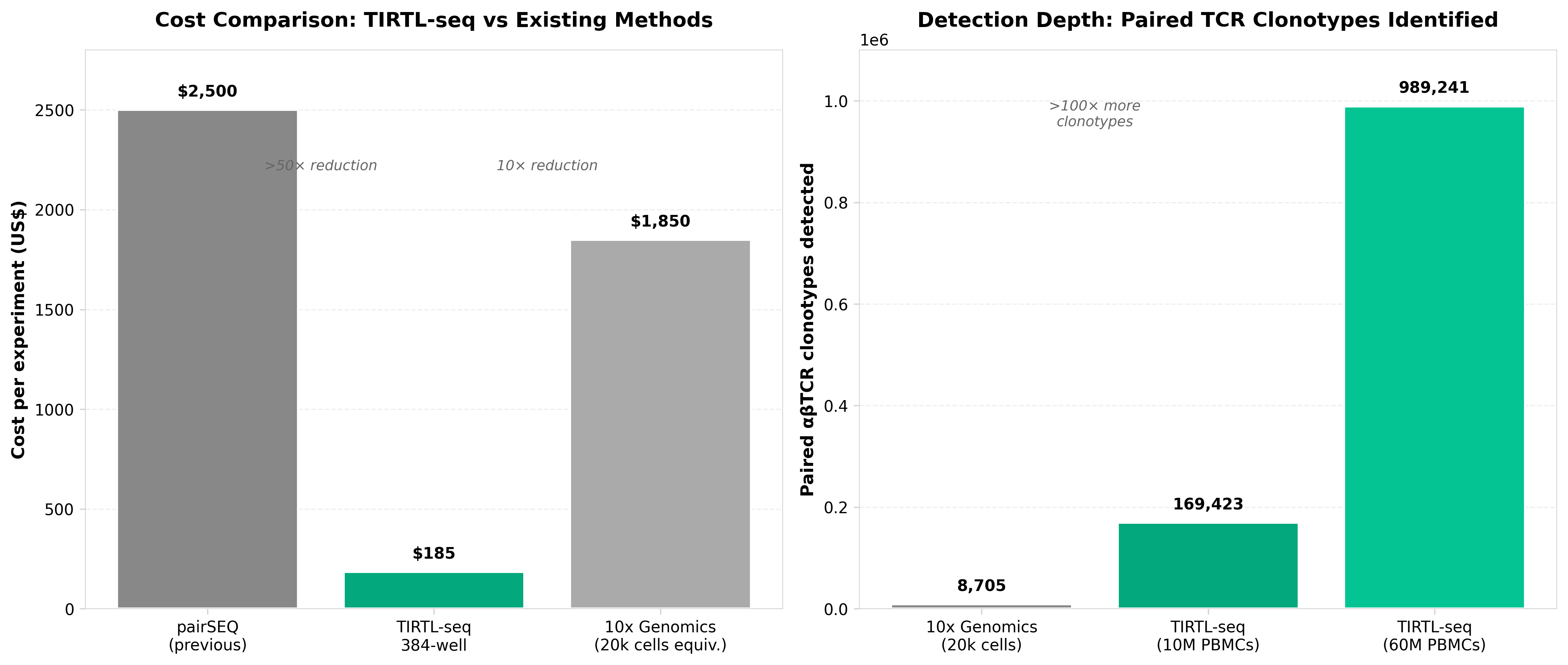
• Cost-effective paired TCR sequencing enables large-scale immunology studies: TIRTL-seq reduces barriers to cohort-level TCR profiling, allowing researchers and biotech companies to conduct deeper immune repertoire analysis at scale without prohibitive sequencing costs—critical for vaccine development, CAR-T optimization, and clinical immunogenomics applications.
• Paired TCR data improves clonotype resolution and therapeutic targeting: Deep quantitative pairing of alpha and beta chains provides more accurate clonotype identification than single-chain approaches, directly enhancing the precision of personalized immunotherapies and T cell selection strategies in clinical development pipelines.
5. Helixer: ab initio prediction of primary eukaryotic gene models combining deep learning and a hidden Markov model
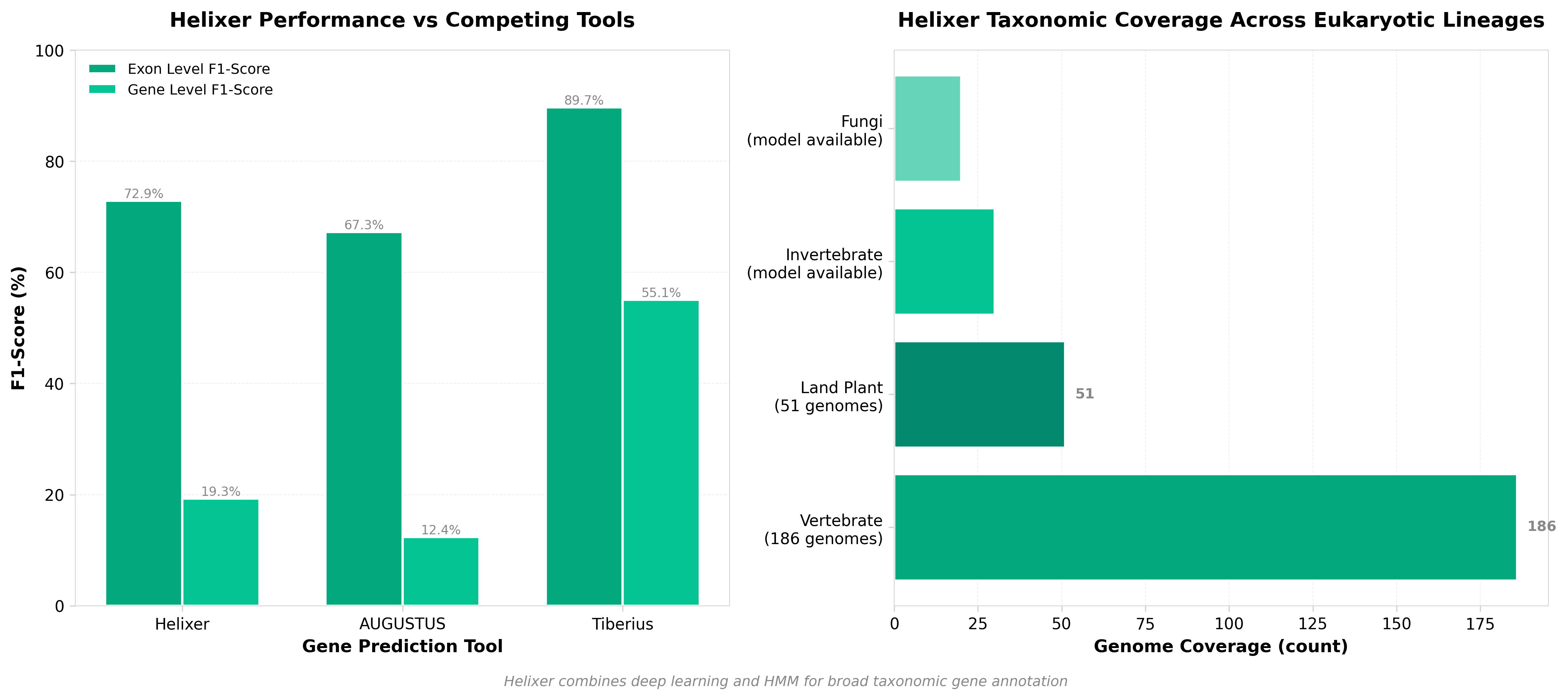
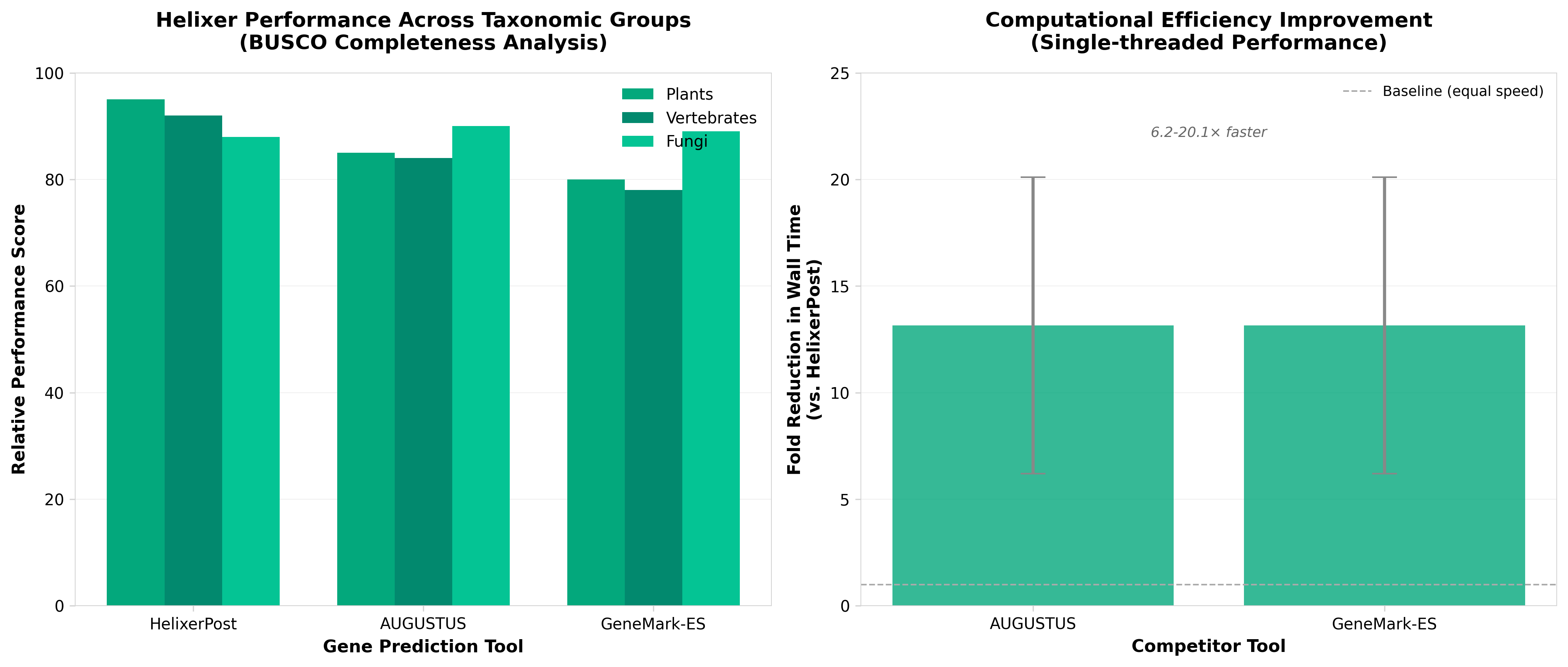
• Unified annotation tool across eukaryotic diversity: Helixer’s broad taxonomic coverage (fungi, plants, vertebrates, invertebrates) addresses a critical bottleneck in genomics—the need for organism-agnostic gene prediction. This eliminates the requirement for species-specific model training, reducing time and computational costs for genome projects across emerging model organisms and non-model species.
• Hybrid ML architecture improves annotation accuracy: The combination of deep learning with hidden Markov models leverages both pattern recognition and biological sequence constraints, likely improving precision over purely statistical or purely neural approaches. This architecture is particularly valuable for complex eukaryotic genomes with alternative splicing and variable gene structure.
• Accelerates functional genomics and agricultural applications: Ab initio gene prediction quality directly impacts downstream analysis (variant interpretation, GWAS, crop improvement). Reliable annotation across diverse taxa enables faster genome-to-phenotype research for both basic biology and crop/livestock breeding programs.
Industry News
1. Versant’s Dayra dawns with $50M Biogen pact, oral macrocyclic peptide mission
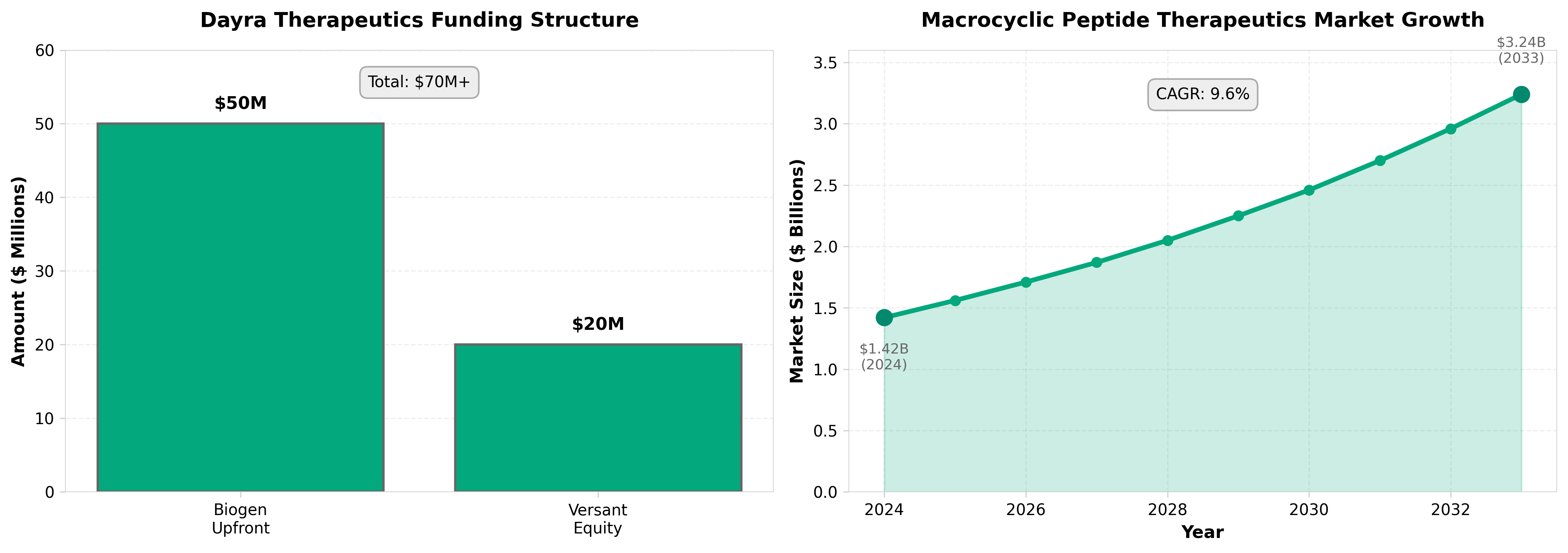
• Oral macrocyclic peptides represent a significant drug delivery advancement: Dayra’s focus on converting macrocyclic peptides to oral formulations addresses a major pharmaceutical bottleneck, as these molecules are typically limited to injectable routes due to poor bioavailability—a solved problem here could unlock a new therapeutic class and expand addressable markets.
• Early-stage validation through a $50M Biogen partnership demonstrates commercial viability: The partnership validates the platform technology at founding and provides substantial capital, suggesting macrocyclic peptide oral delivery has moved beyond theoretical promise to practical application worth major pharma investment and de-risking.
• VC-backed platform companies are increasingly targeting undruggable molecule classes: Versant’s backing of Dayra reflects an industry shift toward solving fundamental chemistry/delivery problems rather than incremental target optimization, positioning enabler platforms as high-value acquisition or partnership targets.
2. Craig Crews, Mikael Dolsten raise $32M for Quarry Thera
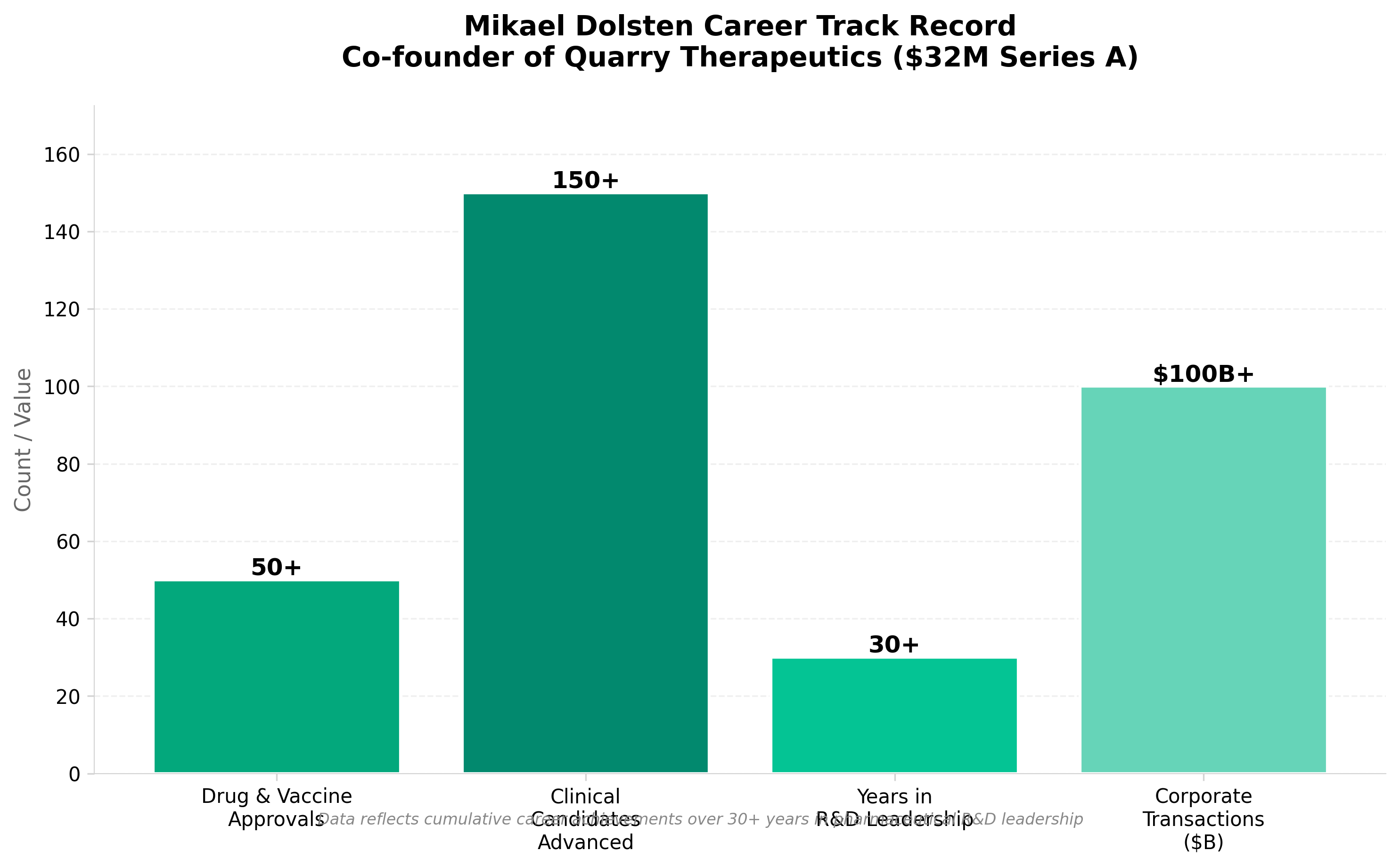
• $32M seed funding validates targeted protein degradation as commercial opportunity. Quarry Thera’s capitalization signals investor confidence in the degradation-focused modality beyond major pharma, creating a potential market segment for specialized biotech focused on this mechanism.
• Leadership pedigree (former Pfizer CSO + established researcher Craig Crews) indicates access to differentiated science and capital networks. The combination of C-suite pharma experience with academic credibility positions the company to potentially attract downstream partnerships and talent more efficiently than typical early-stage ventures.
3. Solventum to buy Acera Surgical for up to $850M, repurchase $1B in stock
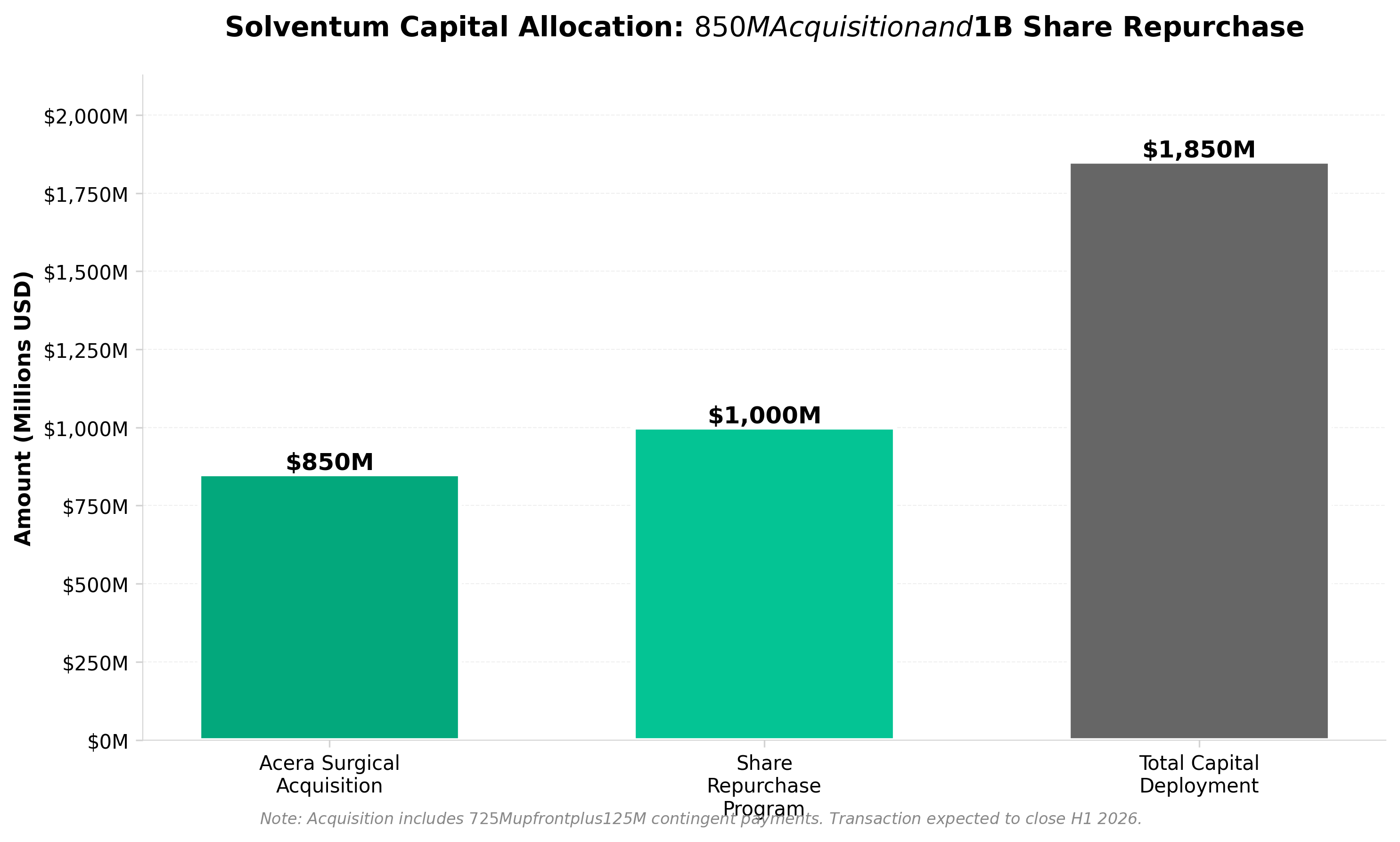
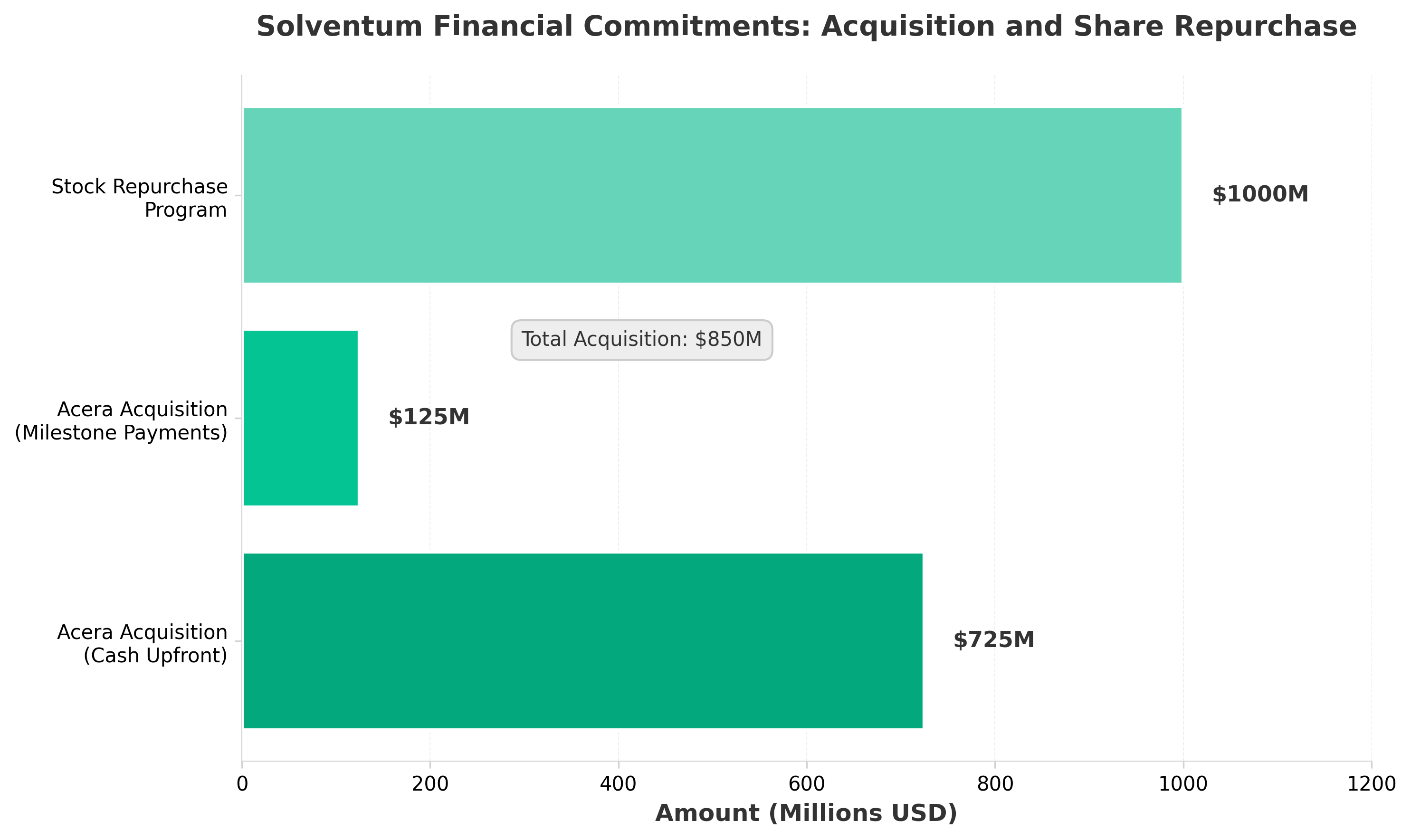
• Solventum’s $850M acquisition of Acera Surgical signals aggressive portfolio consolidation in wound care, a high-margin segment with recurring revenue potential. This represents the company’s first major M&A move post-3M spinoff, indicating strategic focus on specialized healthcare segments over diversified industrial businesses.
• The concurrent $1B stock repurchase demonstrates balanced capital allocation strategy—acquiring growth assets while returning capital to shareholders. This dual approach suggests management confidence in Acera’s integration and Solventum’s standalone financial position approximately 2 years post-separation.
• Wound care acquisition aligns with broader medtech trend toward specialty clinical segments with higher reimbursement predictability and customer lock-in, differentiating Solventum from legacy 3M’s broad-based healthcare portfolio approach.