Headlines
Industry News
- “Heavy AI” is taking over biopharma
- Regeneron makes $150M bet on Tessera’s rare disease gene writing prospect
- Protego gets $130M for AL amyloidosis drug from leaders behind Vyndaqel
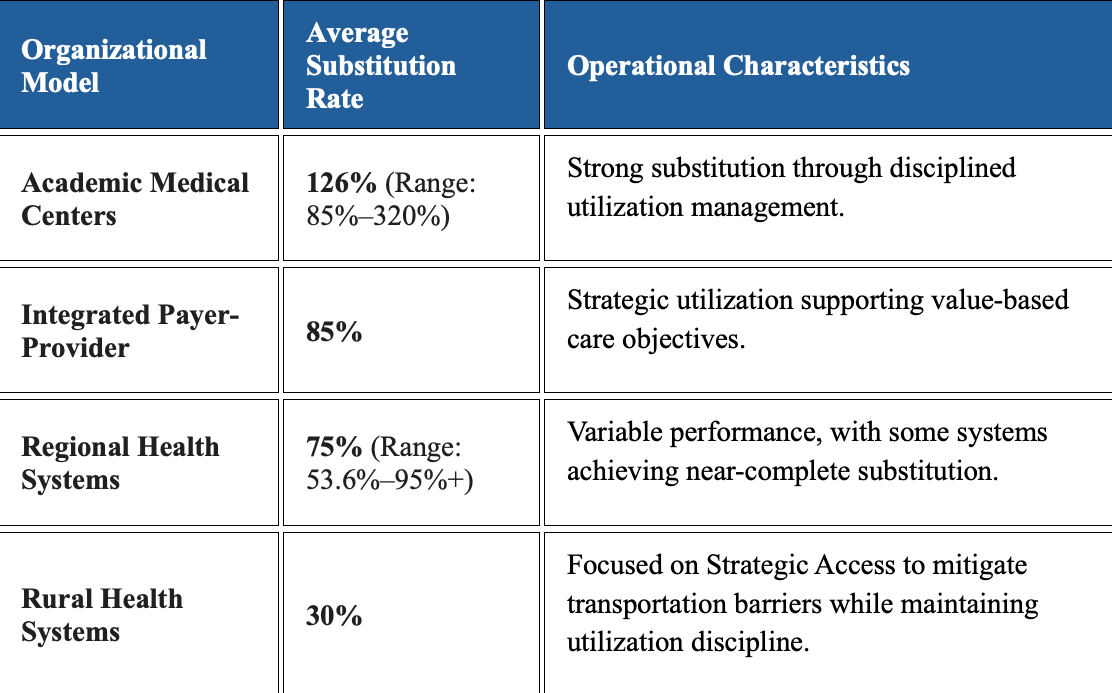
- 74% of Medicare Telehealth Visits Are Substitutive, Not Additive
Clinical Studies & Translational Reports
- Liraglutide in mild to moderate Alzheimer’s disease: a phase 2b clinical trial
- Generate:Biomedicines to start Phase 3 studies for TSLP antibody
- Decentralized Biobanking Pathway to Precision Medicine: Futures Study
New Research
- Knowledge-Practice Performance Gap in Clinical Large Language Models
- Smartphone-based prediction of dopaminergic deficit in Parkinson’s disease
- Next-generation method for preparing plant cells for single-cell RNA sequencing
- ML-driven glycolytic subtyping overcome drug resistance in myeloid leukemia
- Sustained HIV-1 remission after heterozygous CCR5Δ32 stem cell transplantation
Industry News
1. “Heavy AI” is taking over biopharma
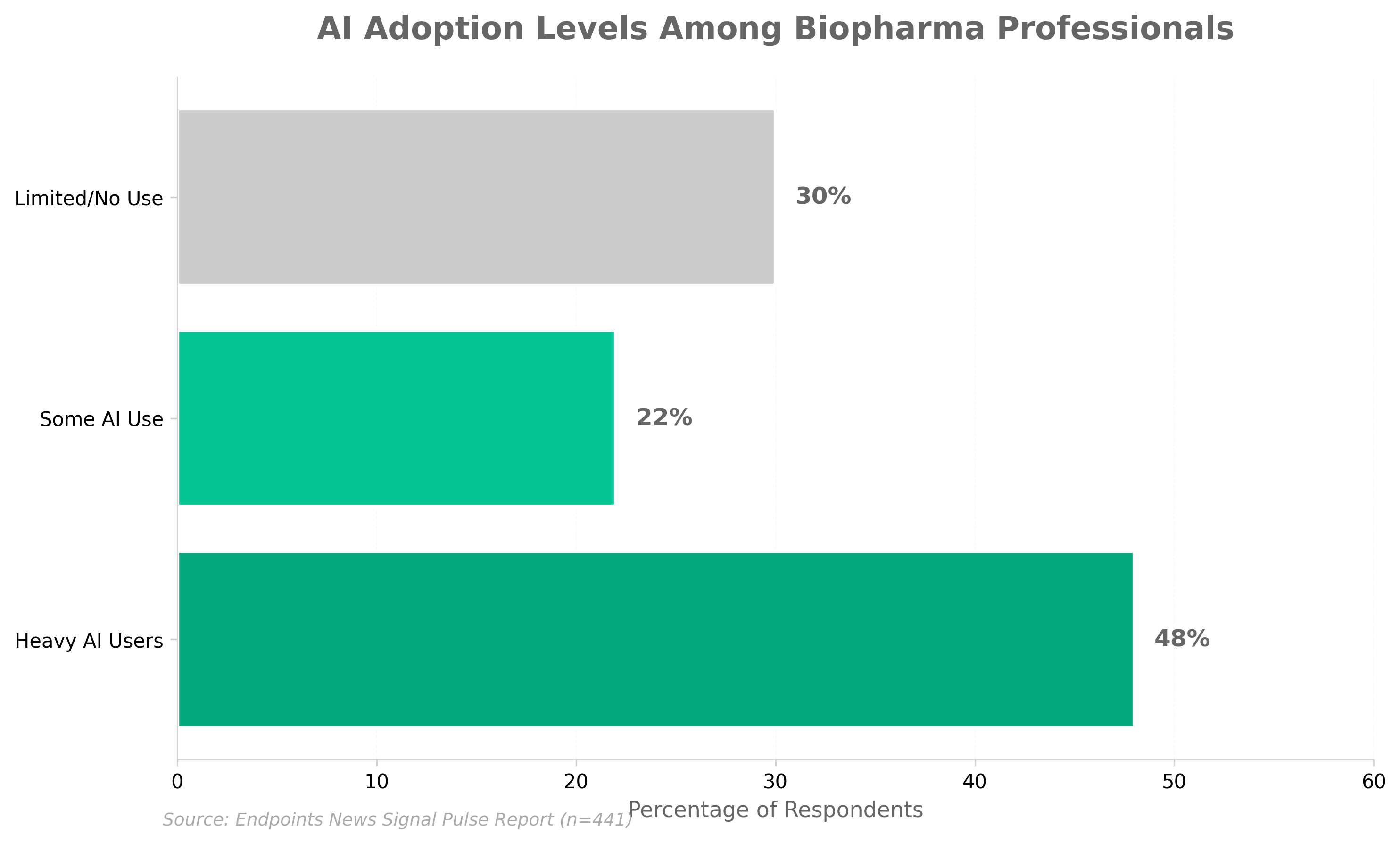
• Nearly half (48%) of biopharma professionals now identify as heavy AI users, signaling rapid mainstream adoption across the industry—organizations should assess their AI competency gaps and workforce readiness to remain competitive.
• The shift toward “heavy” AI usage (versus exploratory or minimal use) indicates AI is moving from pilot phase to embedded workflow integration, requiring investment in validation, compliance infrastructure, and data governance protocols.
2. Regeneron makes $150M bet on Tessera’s rare disease gene writing prospect
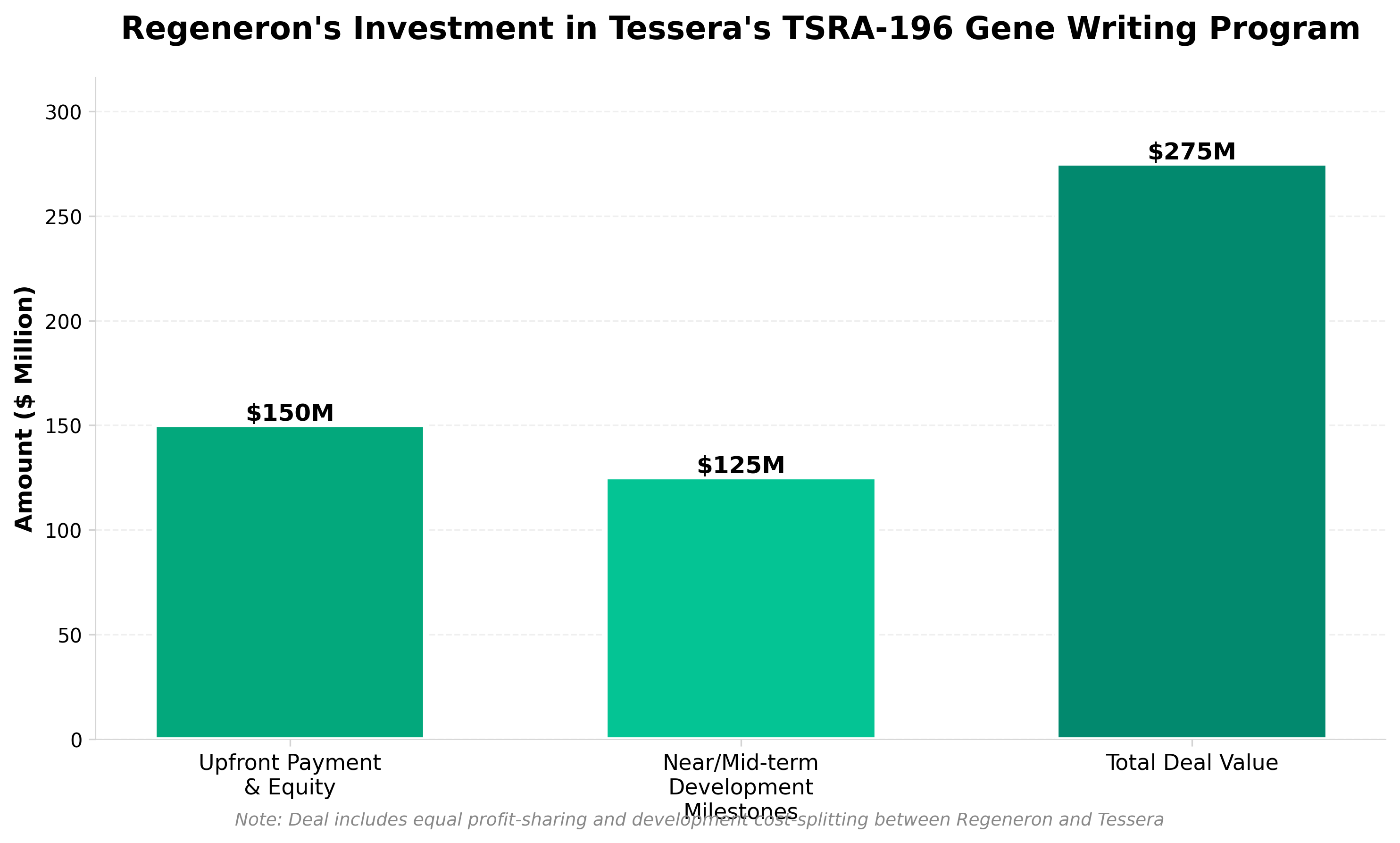
• Regeneron’s $150M investment validates gene writing as a competitive alternative to gene editing for rare genetic diseases, signaling potential market expansion beyond traditional CRISPR approaches and creating a differentiation opportunity for companies with proprietary writing platforms.
• TSRA-196’s advancement to near-clinical stage for AATD—a disease affecting ~100,000 patients in the US with limited treatment options—demonstrates commercial viability of gene writing for monogenic disorders, potentially enabling faster development timelines and reduced off-target risk compared to conventional gene therapies.
• The joint development and commercialization structure indicates confidence in scalability and regulatory pathway clarity, suggesting gene writing candidates may face lower technical and regulatory hurdles than earlier-generation gene therapies, which could accelerate market entry for similar programs in the Tessera pipeline.
3. Protego gets $130M for AL amyloidosis drug from leaders behind Vyndaqel
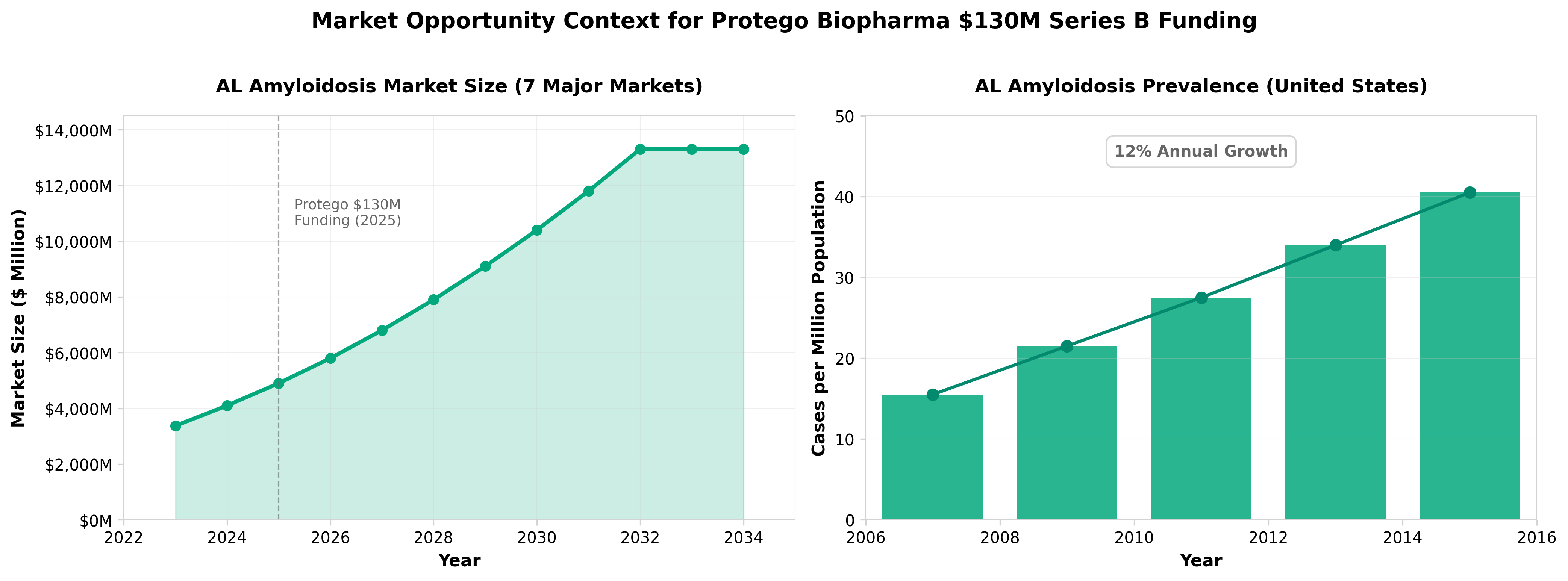
• Protego secured $130M to advance an AL amyloidosis therapeutic toward pivotal trials in 2025, with backing from investors behind Vyndaqel—the $5B+ revenue standard-of-care comparator. This validates market confidence in the AL amyloidosis space and signals potential competitive positioning against an established player.
• The funding scale and investor pedigree suggest substantial commercial expectations for an AL amyloidosis program, indicating the rare protein misfolding disease represents a high-value market opportunity with room for multiple successful therapies despite Vyndaqel’s dominance.
• Timing a pivotal trial launch for 2025 creates a near-term inflection point for clinical data readouts, establishing a competitive window where faster enrollment and efficacy differentiation could determine market share in a disease with growing patient awareness and diagnosis rates.
4. 74% of Medicare Telehealth Visits Are Substitutive, Not Additive (ATA CODE Report)
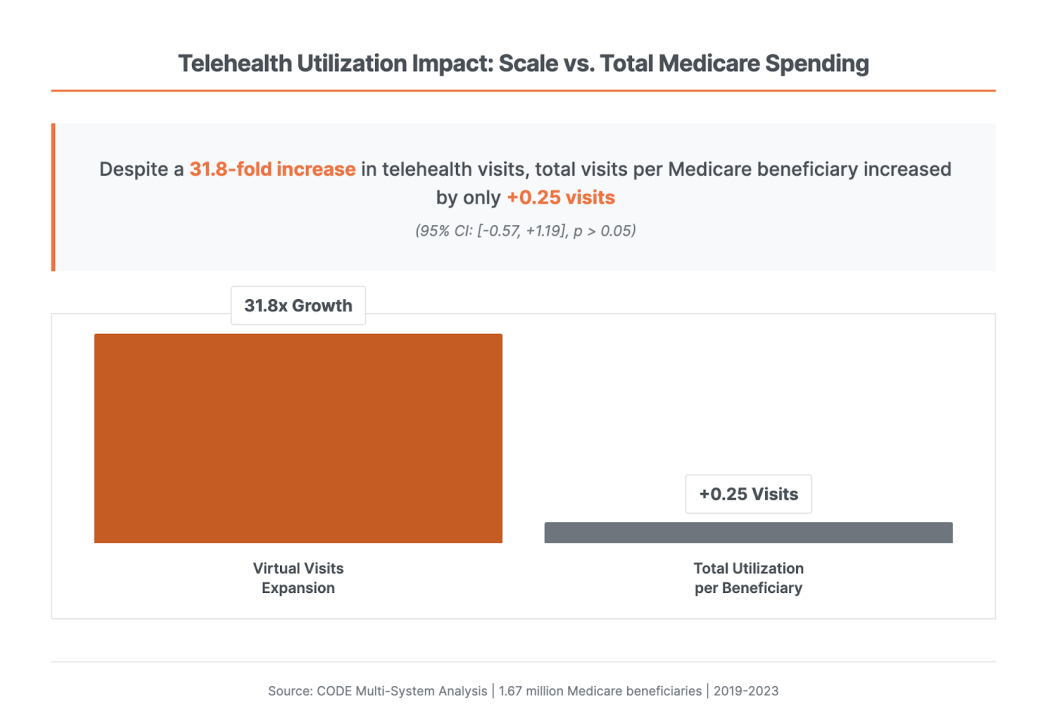
Clinical Studies & Translational Reports
1. Liraglutide in mild to moderate Alzheimer’s disease: a phase 2b clinical trial
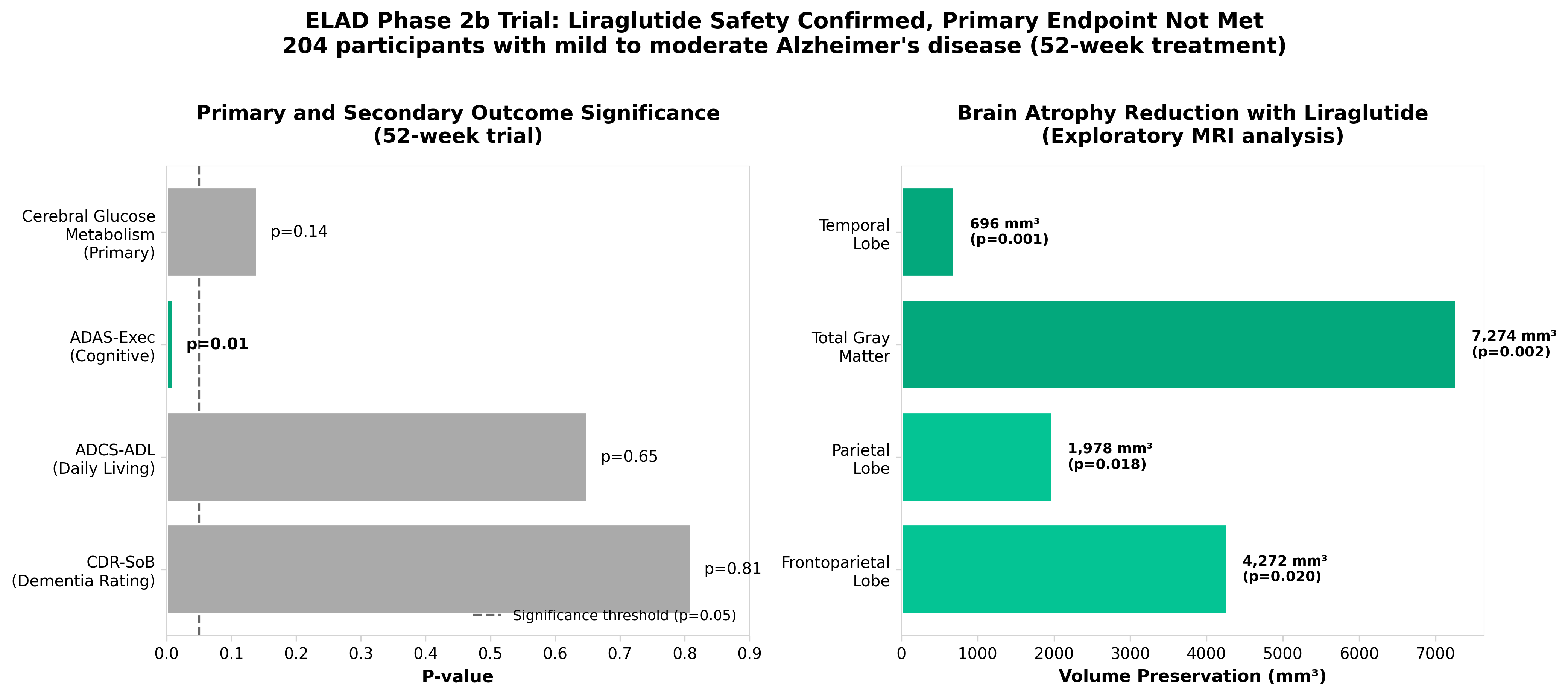
• GLP-1 class repositioning faces efficacy headwinds in neurodegeneration: Liraglutide’s failure to significantly slow brain metabolism decline in mild-to-moderate AD (despite safety/tolerability) suggests GLP-1 agonists may have limited disease-modifying potential in Alzheimer’s, redirecting investment and partnership focus toward alternative mechanisms or earlier disease stages.
• Phase 2b negative result impacts pipeline strategy and funding landscape: The ELAD 2 trial outcome will likely reduce biotech enthusiasm for GLP-1 repurposing in neurodegenerative indications, potentially accelerating consolidation around validated targets (amyloid, tau) and creating valuation pressure for companies with concentrated AD portfolios dependent on this hypothesis.
• Safety profile alone insufficient for AD market entry: Establishing tolerability without efficacy in a competitive AD landscape (where amyloid-targeting agents now show modest cognitive benefits) sets a high bar for future GLP-1 AD trials, suggesting companies pursuing this approach must either demonstrate biomarker engagement or shift to prevention/prodromal populations to justify development costs.
2. Generate:Biomedicines to start Phase 3 studies for TSLP antibody
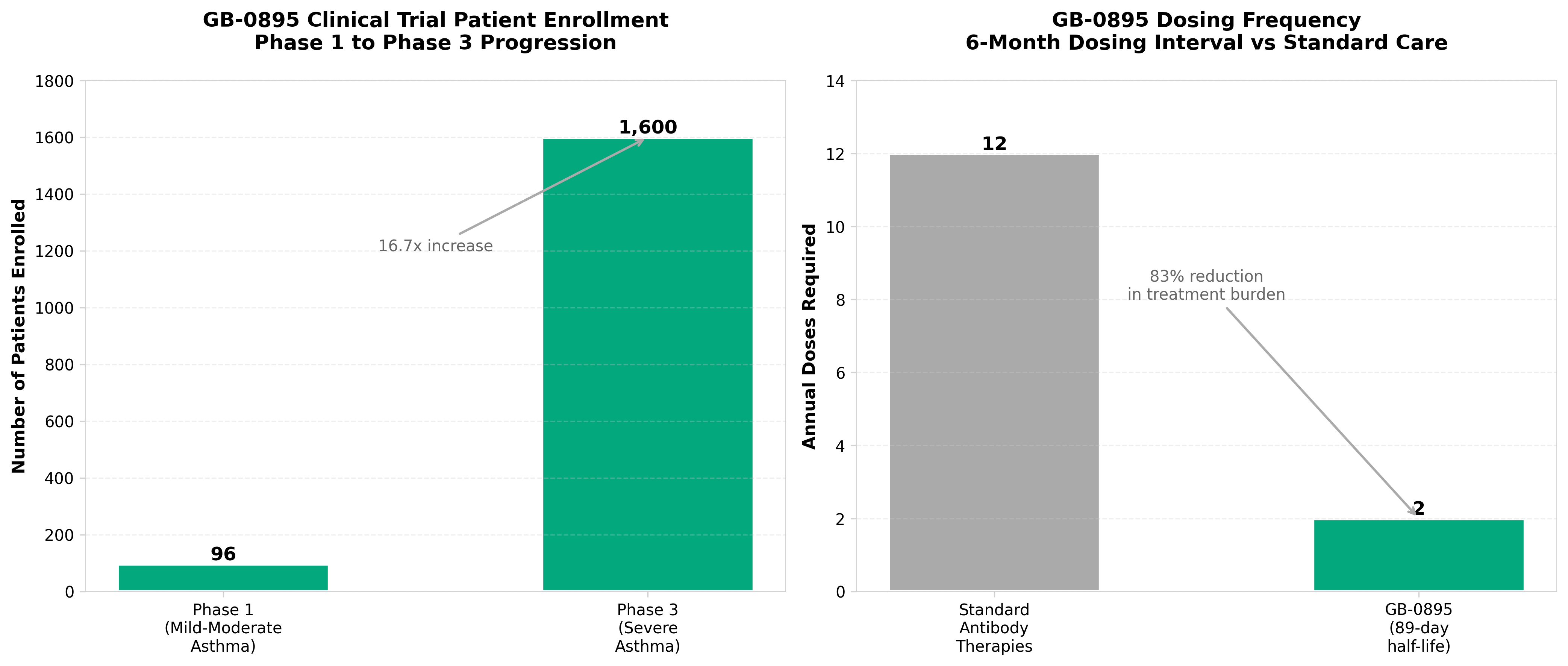
• Generate:Biomedicines is advancing its TSLP antibody candidate to Phase 3 studies, marking a critical inflection point for AI-designed therapeutics moving from preclinical validation into late-stage clinical development and demonstrating commercial viability of computational drug design platforms.
• The dual Phase 3 study launch suggests confidence in the candidate’s efficacy profile and broadens potential market applications, which could validate AI-driven approaches across multiple indications and influence future funding and partnership decisions in the computational biotech sector.
• This milestone represents a near-term catalyst for Generate:Biomedicines’ valuation and could accelerate investor interest in AI-first biotech models, though Phase 3 execution risk remains—clinical data quality and regulatory outcomes will determine whether this validates the broader AI drug discovery trend.
3. Decentralized Biobanking Pathway to Precision Medicine: Futures Study
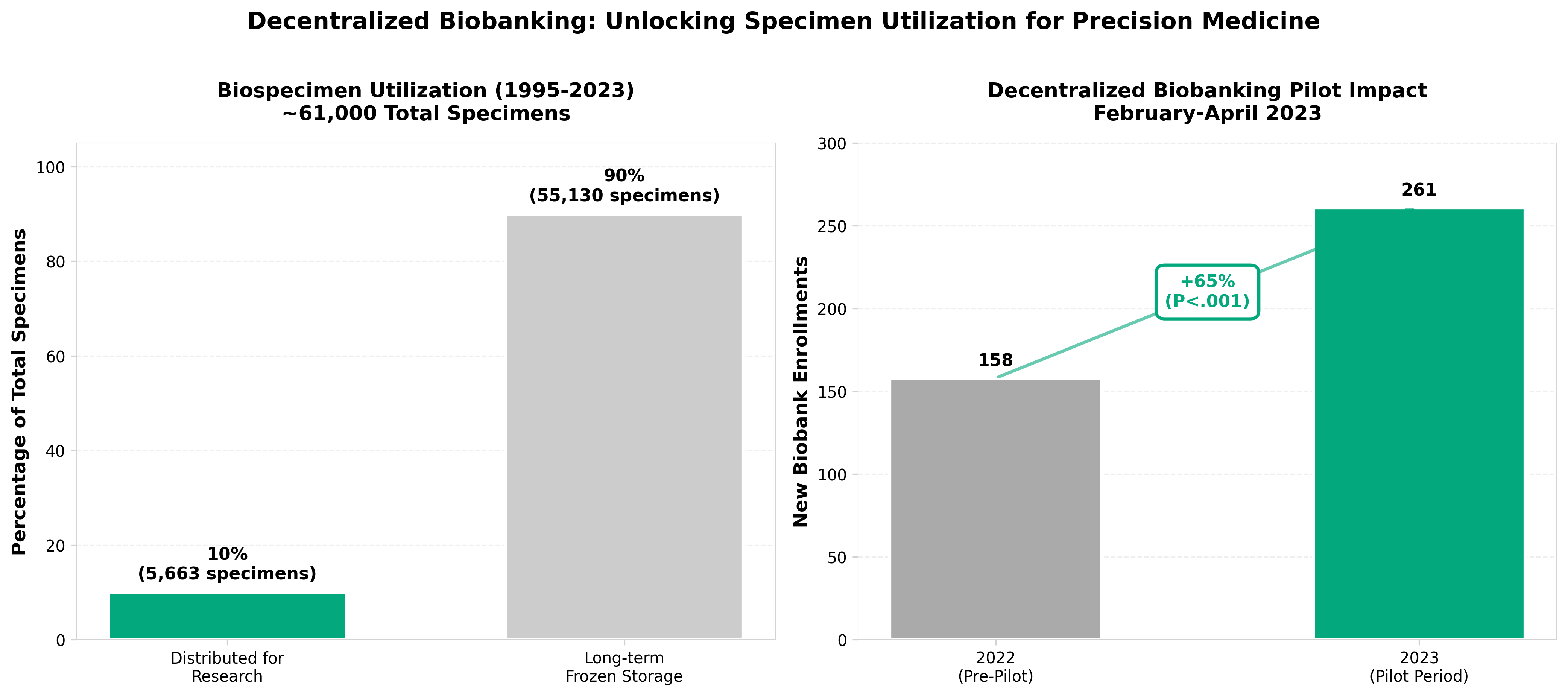
• Patient engagement and biobank enrollment increase through transparency: Pilot data demonstrated robust enrollment and concurrent biobank enrollment increases when patients could track their biospecimens via decentralized apps, suggesting that specimen ownership visibility directly drives participation rates and addresses the current siloing problem that hinders precision medicine research.
• “Rails, rules and tools” framework enables scalable implementation: The study identifies specific governance and ethical oversight mechanisms needed for decentralized biobanking platforms, providing a replicable pathway for institutional adoption rather than theoretical concepts alone—critical for achieving the stated 2030 goal of enabling all U.S. biospecimen donors.
• Reframed incentive alignment unlocks multi-modal data access: By shifting the paradigm from anonymous specimen removal to patient-researcher partnerships with compensation potential and network reciprocity effects, decentralized biobanking removes structural barriers to integrating multi-modal patient data essential for precision medicine applications.
New Research
1. Knowledge-Practice Performance Gap in Clinical Large Language Models: Systematic Review of 39 Benchmarks
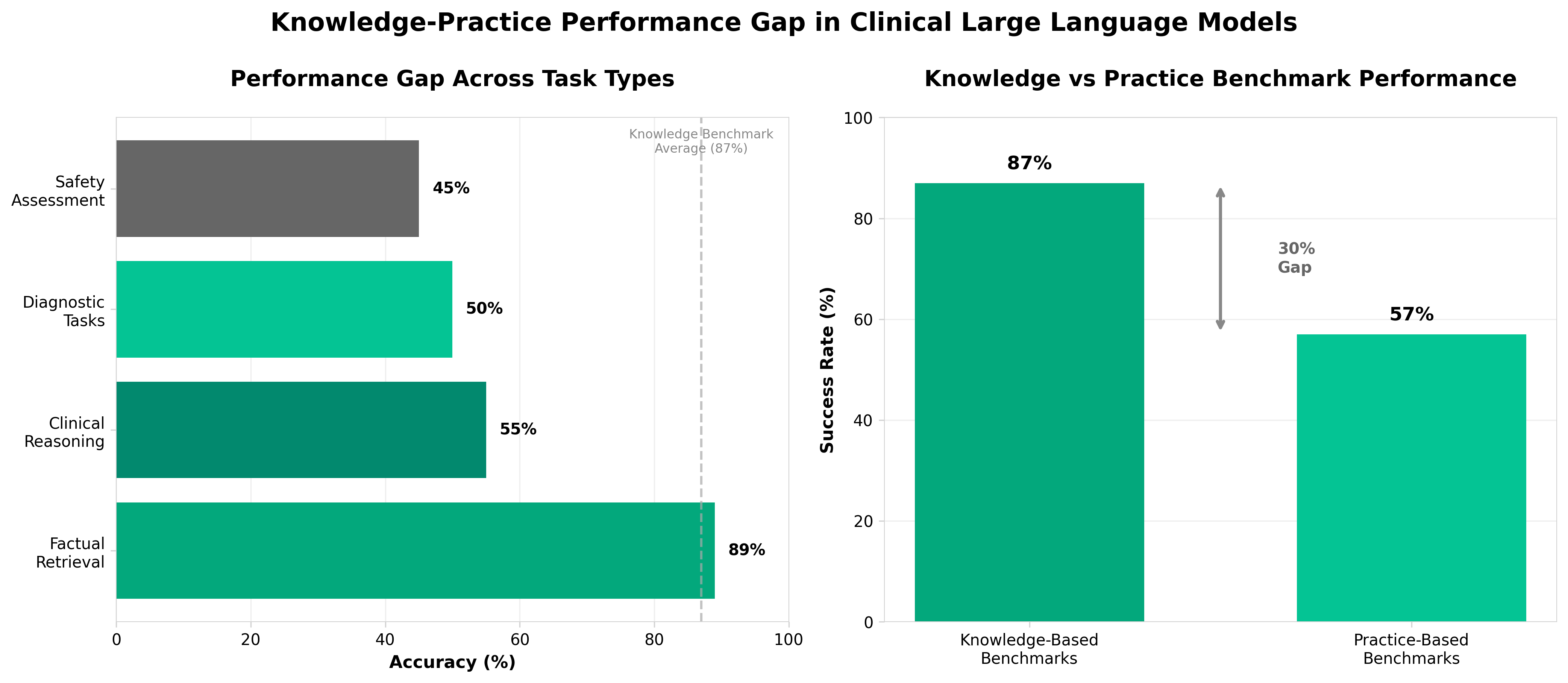
• Knowledge-practice performance gap is substantial and quantifiable: LLMs achieve 84-90% accuracy on knowledge-based exams (USMLE-style) but only 45-69% on practice-based assessments, with safety evaluations critically underperforming at 40-50%—directly undermining claims of clinical readiness and establishing a concrete benchmark for regulatory decision-making.
• Clinical reasoning and diagnostic tasks are performance bottlenecks: Task-specific analysis reveals factual retrieval maintains 85-93% accuracy while clinical reasoning drops to 50-60% and diagnostic tasks achieve only 45-55%—indicating that organizations deploying LLMs must prioritize validation in these specific domains rather than relying on examination scores.
• Current evaluation methodologies are insufficient for deployment decisions: 26% of benchmarks lack adequate methodological reporting, and 54% of existing frameworks remain knowledge-based despite the paradigm shift toward practice-based assessment—requiring health systems and regulators to demand standardized practice-oriented validation protocols and mandate human-in-the-loop oversight before clinical implementation.
2. Smartphone-based prediction of dopaminergic deficit in prodromal and manifest Parkinson’s disease
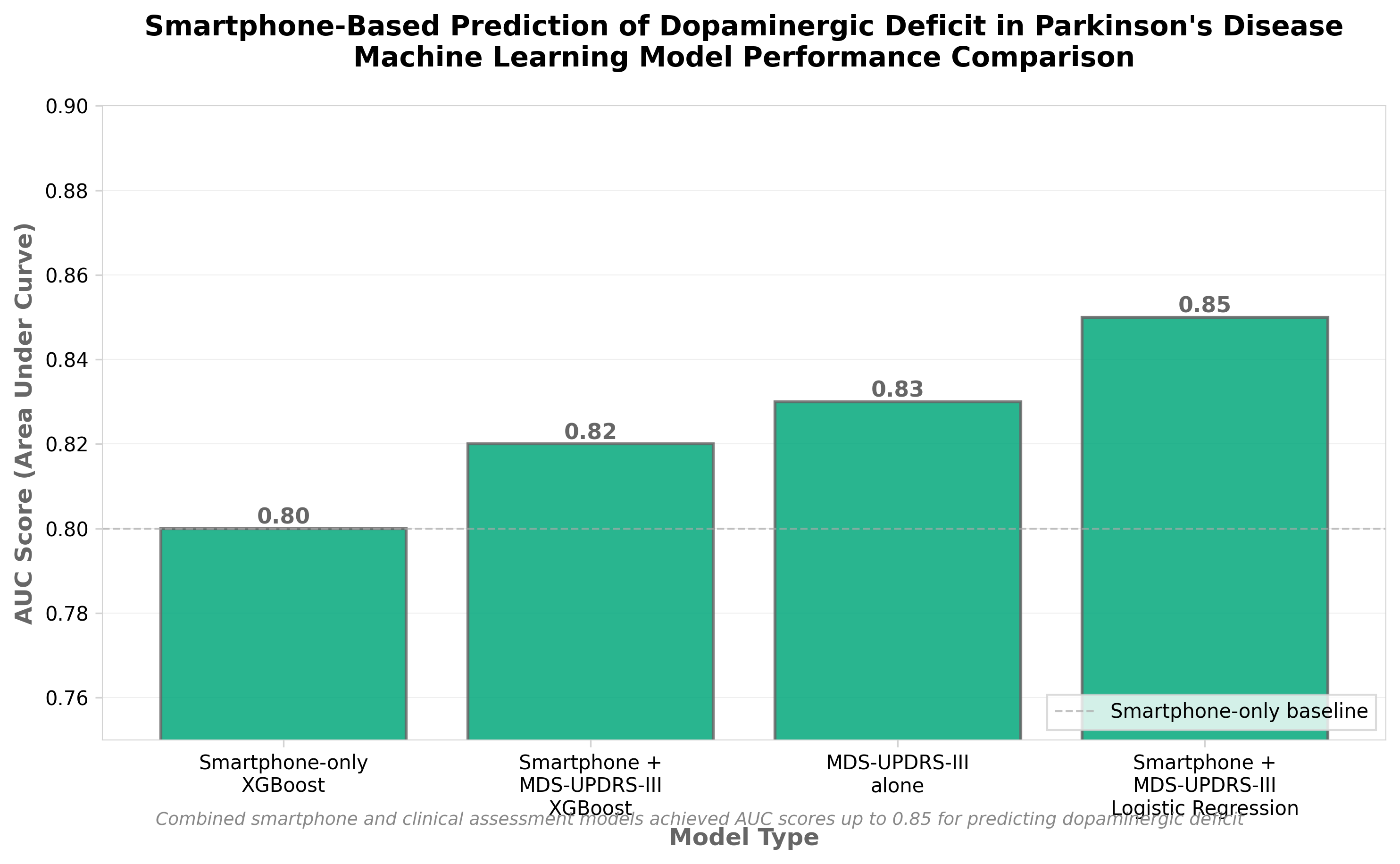
• Early detection capability shifts diagnostic paradigm: Smartphone-based assessment enables identification of dopaminergic deficits in prodromal (pre-symptomatic) Parkinson’s disease, potentially allowing therapeutic intervention before irreversible neurodegeneration occurs—addressing a major unmet need in neurodegenerative disease management.
• Digital biomarker reduces assessment barriers: Mobile phone-based prediction eliminates need for specialized PET imaging or clinical visits, lowering cost and accessibility barriers for large-scale screening programs and enabling remote monitoring in distributed patient populations.
• Validates digital health for neurological conditions: Successful dopamine prediction using smartphone data establishes a replicable model for quantifying neurochemical deficits through behavioral/motor tasks, with potential applications across other neurodegenerative diseases requiring early biomarker detection.
3. Next-generation method for preparing plant cells for single-cell RNA sequencing
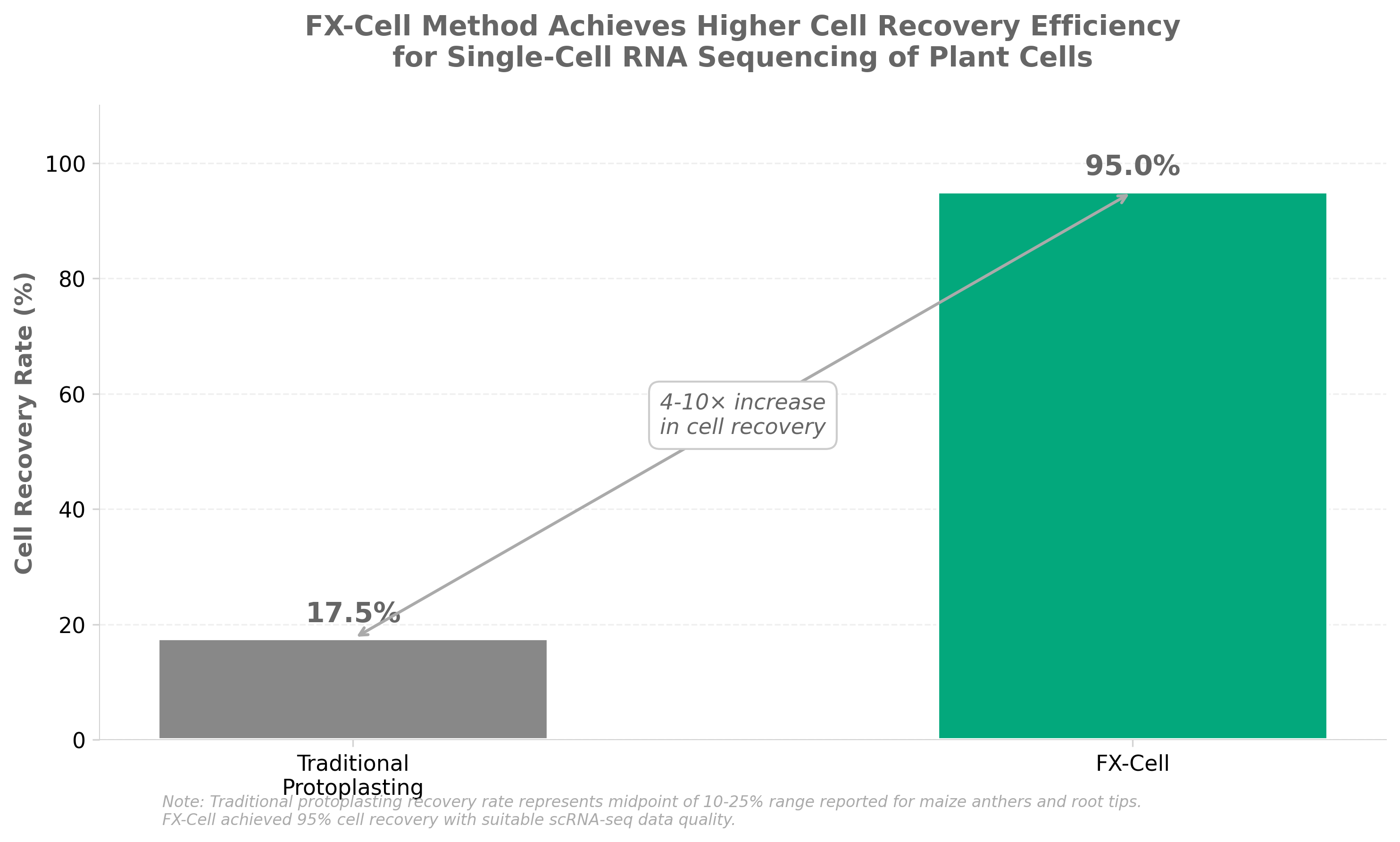
• Addresses a critical technical bottleneck in plant genomics research: The plant cell wall has been a major obstacle for single-cell RNA sequencing applications; FX-Cell’s high-temperature enzymatic digestion approach removes this barrier, enabling wider adoption of scRNA-seq across plant biology and agricultural biotechnology research.
• Expands market opportunities for single-cell analysis in plant breeding and synthetic biology: High-quality plant cell preparation will accelerate crop improvement programs, plant synthetic biology applications, and cell-type specific gene discovery in agronomically important species—markets currently underserved compared to animal cell scRNA-seq tools.
• Likely to drive standardization in plant cell preparation protocols: Publication in Nature Methods signals methodological rigor and reproducibility; industry adoption could establish FX-Cell as a preferred standard, creating downstream opportunities for compatible reagent kits, automation platforms, and integrated analysis workflows targeting the plant research community.
4. Machine learning-driven glycolytic subtyping and exosome-based PKM splicing modulation overcome drug resistance in hyper-glycolytic myeloid leukemia
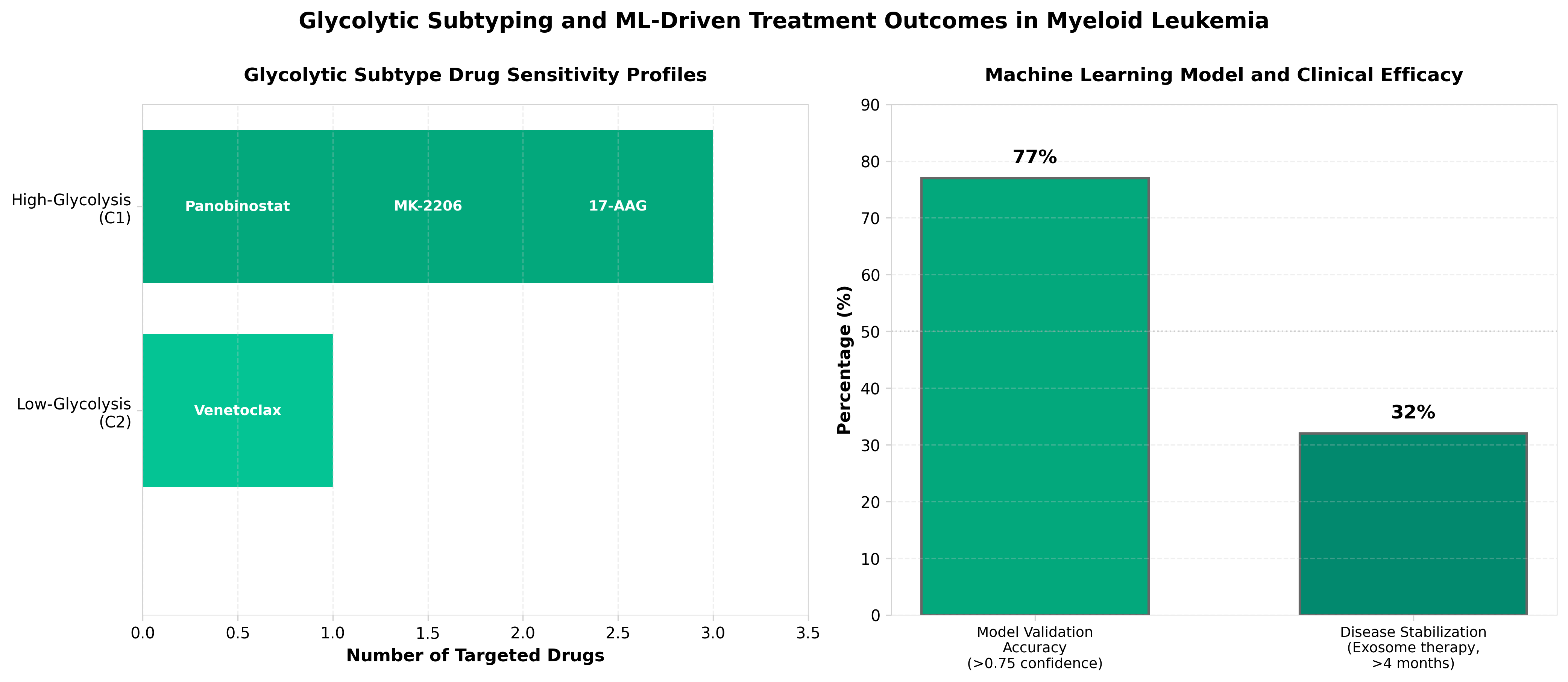
• ML-enabled patient stratification creates precision medicine opportunity: Machine learning-driven glycolytic subtyping enables identification of hyper-glycolytic myeloid leukemia patients who would benefit from targeted therapy, potentially reducing trial failures and improving clinical trial efficiency through better patient selection.
• Exosome-based PKM splicing modulation addresses resistance mechanism: Targeting pyruvate kinase M (PKM) splicing via exosome delivery provides a novel approach to overcome drug resistance in glycolytic-dependent leukemias, opening a new therapeutic angle beyond standard cytotoxic or kinase inhibitor strategies.
• Metabolic biomarkers validate actionable drug resistance pathway: Glycolytic profiling serves as a predictive biomarker for drug response, enabling companion diagnostic development and potential combination therapy strategies that link metabolic phenotyping to treatment selection in acute myeloid leukemia.
5. Sustained HIV-1 remission after heterozygous CCR5Δ32 stem cell transplantation
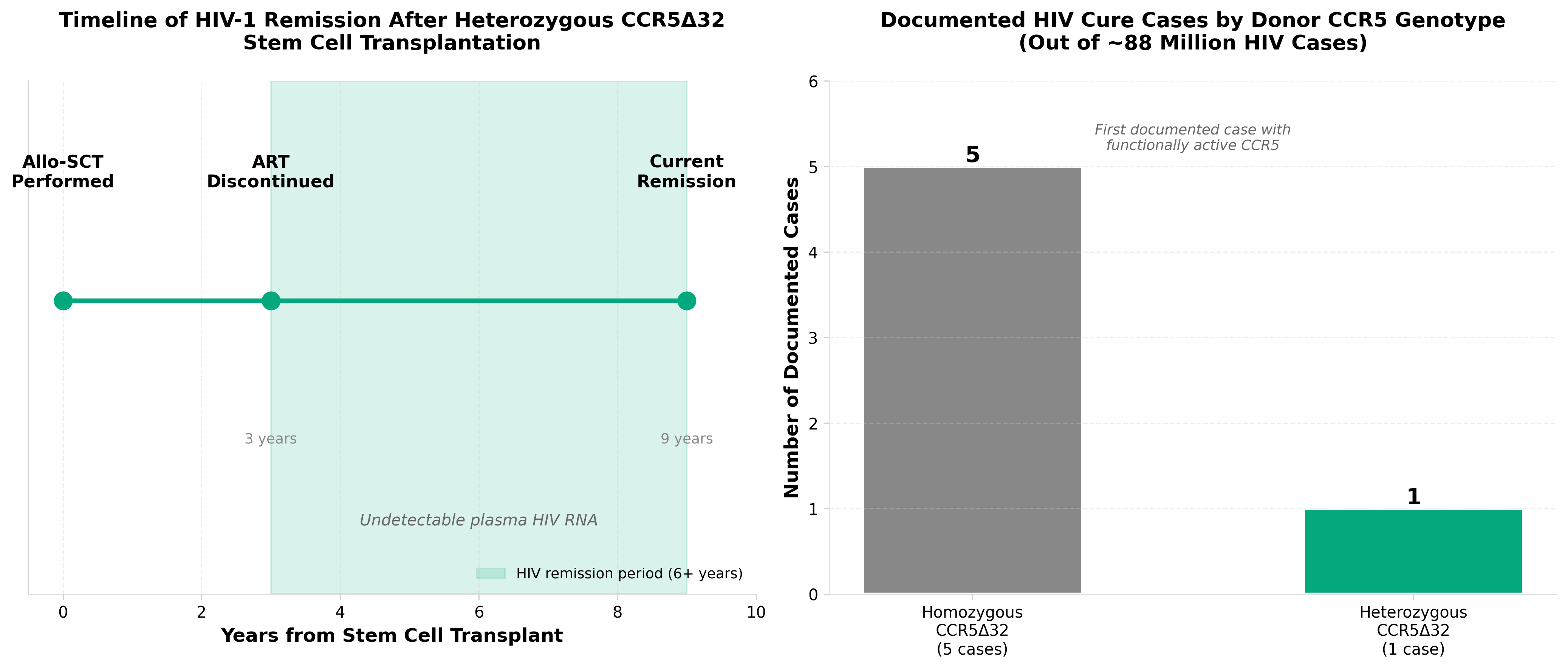
• Heterozygous CCR5Δ32 demonstrates clinical efficacy for HIV remission: This finding expands the therapeutic window beyond previous homozygous-only approaches, potentially increasing the eligible patient population and reducing the technical barriers for stem cell engineering-based HIV treatments in clinical practice.
• Shifts HIV cure strategy from ablative to non-myeloablative transplantation: The sustained remission achieved with heterozygous CCR5Δ32 cells suggests reduced conditioning requirements compared to earlier “Berlin patient” homozygous protocols, lowering transplant-related toxicity and mortality risk—a critical factor for commercial scalability and patient acceptance.
• Validates CCR5 gene editing as a durable HIV functional cure pathway: Long-term remission data supports investment in autologous stem cell engineering platforms and validates CCR5-targeting as a preferred mechanism over other gene therapy approaches, informing pipeline prioritization for biotech companies developing HIV cell therapies.
