Headlines
Industry News
- Clinical trials tech startup Paradigm raises $78M, acquires part of Roche’s Flatiron
- Medtronic’s Hugo surgical robot secures US clearance
- Artera Hits $100M ARR and Secures $65M to Lead Agentic AI in Healthcare
- Sober Sidekick Achieves 88% Accuracy in Identifying High-Cost Risk for SUD
- Circulate Health Secures Investment to Scale Microplastics Removal Technology
Clinical Studies & Translational Reports
- Digital Biometrics in Predicting Risk for Obstructive Sleep Apnea and Hypertension: Decentralized, Prospective Cohort Study
- With Phase 3 Duchenne success, Capricor plots path forward after FDA rejection
- COVID-19 mRNA Vaccination and 4-Year All-Cause Mortality Among Adults in France
New Research
- Deriving novel atrial fibrillation phenotypes using a tree-based artificial intelligence-enhanced electrocardiography approach
- Predicting 1-year successful clinical use of an arteriovenous access for hemodialysis using machine learning
- A bioorthogonal ligation system induces controlled proximity for cancer therapy
- Unlocking the potential of X-rays to scale up tissue ultrastructure mapping
- C-COMPASS: a user-friendly neural network tool profiles cell compartments at protein and lipid levels
Industry News
1. Clinical trials tech startup Paradigm raises $78M, acquires part of Roche’s Flatiron
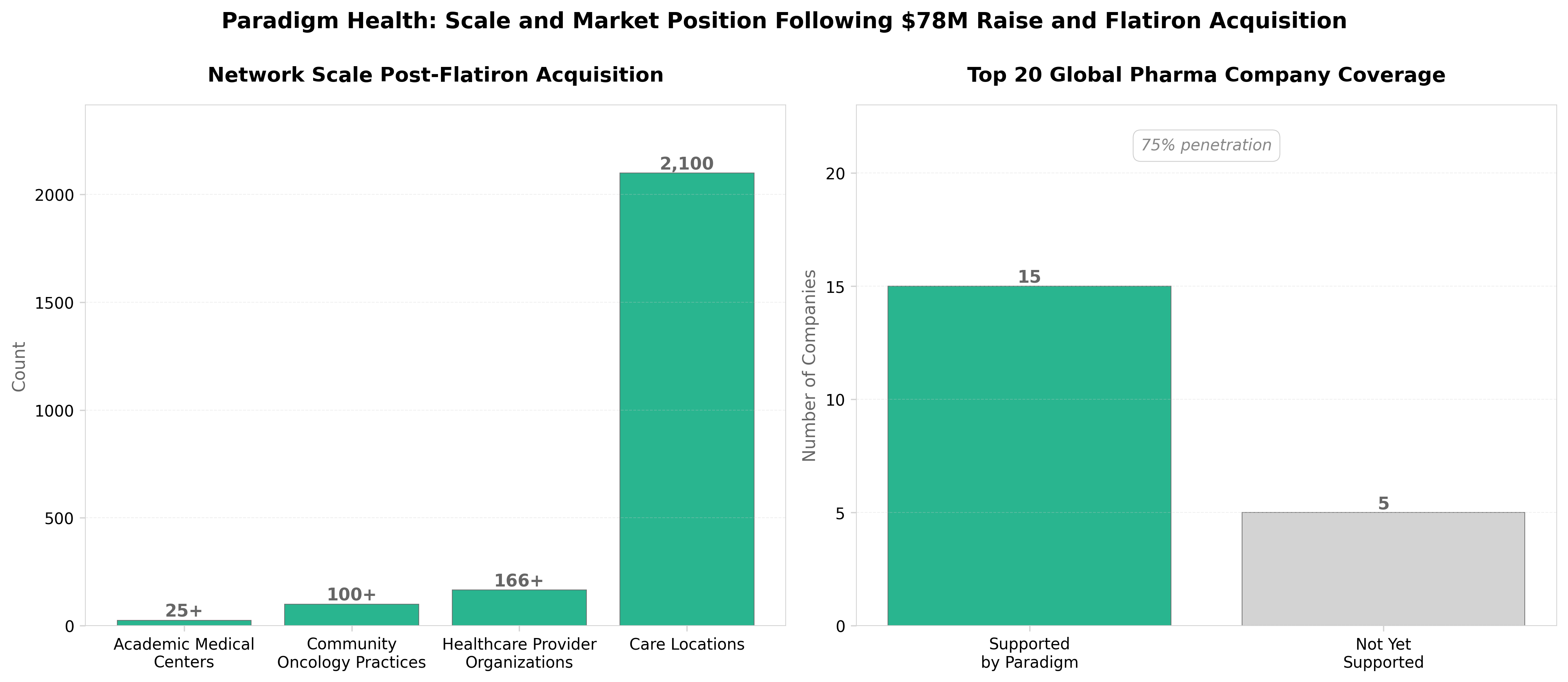
• Market consolidation via strategic M&A: Paradigm’s acquisition of Roche’s Flatiron expands its patient network to 2.4M patients across 2,100 care locations in 45 states, positioning it to capture 75% of top-20 pharma trial activity—demonstrating that clinical trial tech success now requires integrated patient access infrastructure, not just software platforms.
• Accrual bottleneck is addressable at scale: Paradigm’s AI-enabled matching has achieved 4X faster patient accrual, directly tackling the leading cause of trial termination (55% failure rate due to low enrollment). This efficiency gain justifies significant capital deployment and signals a structural shift in how trials are designed and resourced.
• Patient access remains the critical constraint: With U.S. trial participation under 3%, Paradigm’s $78M raise validates that biotech companies will pay premium valuations for technology that solves enrollment rather than pure data analytics—indicating a shift in competitive advantage from trial design optimization to population accessibility.
2. Medtronic’s Hugo surgical robot secures US clearance
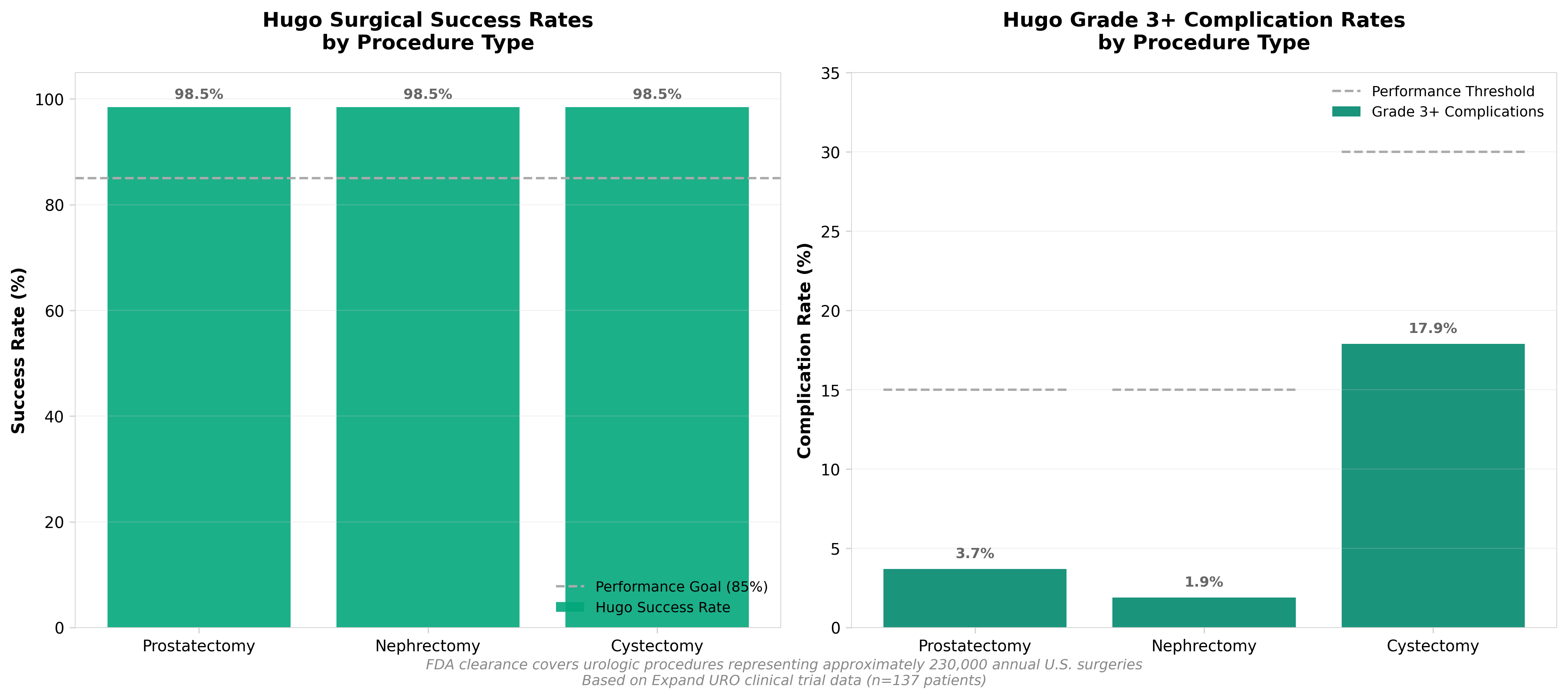
• Immediate Market Expansion Opportunity: Hugo’s FDA clearance covers ~230,000 annual U.S. urologic procedures, with Medtronic positioned at 7% market share in a $11.1B surgical robotics market growing at 13.21% CAGR through 2032; the planned expansion into hernia repairs and gynecology unlocks additional high-volume procedure categories already validated in clinical trials (100% success rate in hernia repair across 193 patients).
• Competitive Differentiation Through Clinical Performance: Hugo achieved 98.5% surgical success in urology (vs. 85% goal) and zero mid-procedure conversions in hernia trials, with complication rates substantially below thresholds; this clinical validation, combined with modular cart-based design deployable across any healthcare setting, positions Hugo to capture share from established players like da Vinci while competing against well-funded entrants (CMR Surgical, Distalmotion) that secured FDA clearance in late 2024.
• Revenue Materialization Timeline Creates Near-Term Catalysts: U.S. sales contribution expected in H2 FY2026 with material impact in FY2027; with tens of thousands of procedures already logged across 30+ countries since 2021 debut, international scale de-risks U.S. launch execution and provides operational
3. Artera Hits $100M ARR and Secures $65M in Funding to Lead Agentic AI Adoption in Healthcare
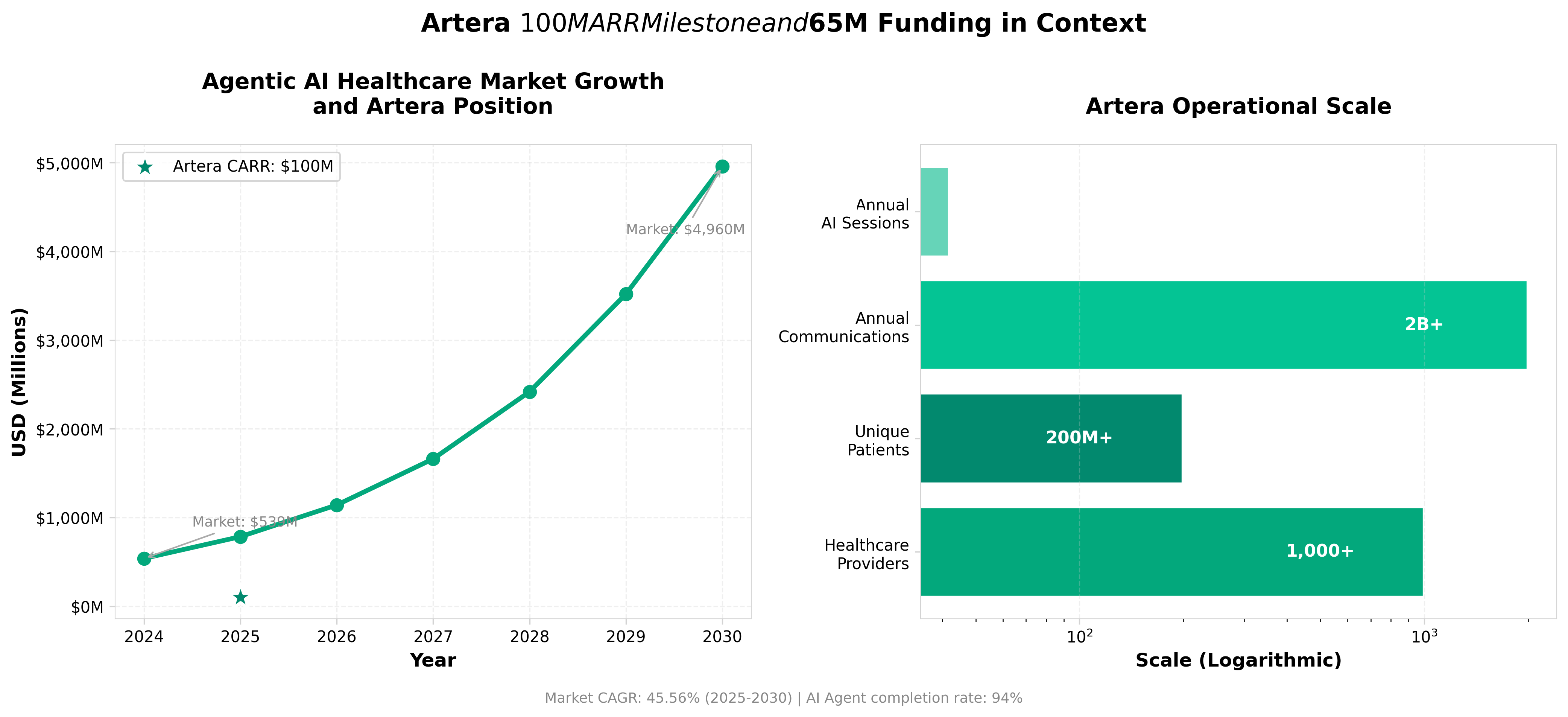
• Agentic AI in healthcare is experiencing explosive market growth (45.6% CAGR through 2030) with Artera commanding ~18% of today’s $538M market. This positions the company as the category leader in a space projected to reach $4.96B by 2030, providing a clear benchmark for investors and competitors assessing market consolidation potential.
• Artera’s 94% autonomous conversation completion rate across 42M annual sessions, paired with a proprietary dataset of 2B+ interactions, creates a defensible moat that newer competitors cannot replicate. This operational efficiency directly addresses the stated market pain point where 47% of patients avoid scheduling appointments due to phone communication friction.
• The $65M funding round validates a decade-long EHR integration strategy and positions agentic AI for enterprise adoption beyond early adopters. With 1,000+ healthcare providers and 200M+ patients already using the platform, the capital deployment will likely accelerate market consolidation and establish Artera as infrastructure rather than point solution.
4. Sober Sidekick’s Empathy Algorithm Achieves 88% Accuracy in Identifying High-Cost Risk, Attracts $7.6M Funding
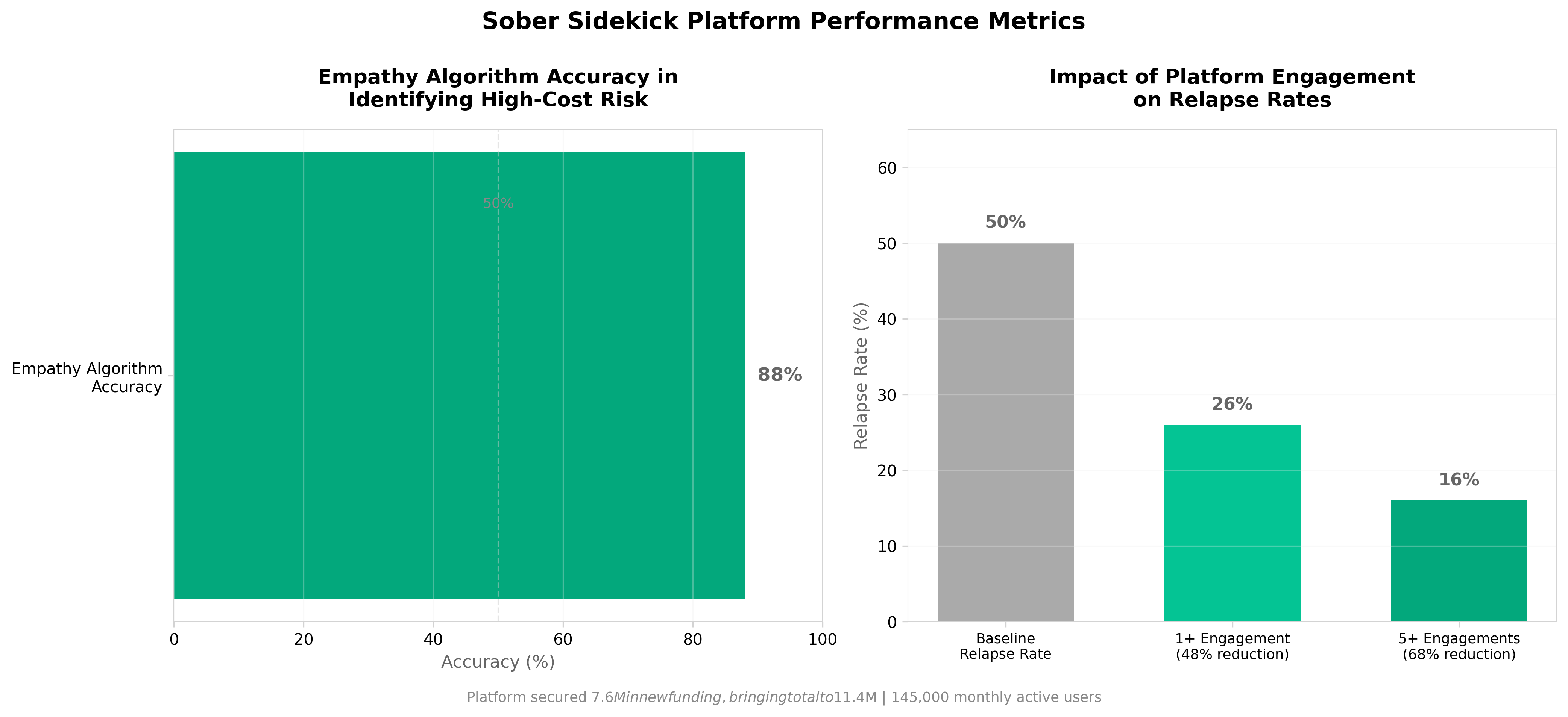
• Predictive accuracy of 86-88% in identifying high-cost Substance Use Disorder (SUD) events creates a quantifiable risk stratification tool for payers. The 100% ROI make-whole guarantee and low PMPM model provide payers with a de-risked entry point to value-based care agreements, directly addressing the $50B annual SUD treatment cost burden and enabling measurable ROI measurement in behavioral health contracting.
• Real-time peer response infrastructure (6.2 responses per post, 100% response rate within minutes) demonstrates a scalable, low-touch intervention model. With 88% of users who self-report relapse re-engaging on the platform and 145,000 monthly active users generating 2M+ peer engagements, this creates an alternative to costly ED visits and rehab readmissions that traditionally drive SUD spend.
• The 34.3M treatment gap (40M affected vs. 5.7M receiving annual care) represents a significant addressable market for digital-first engagement models. $7.6M in new funding fueling payer partnerships signals investor confidence in SUD as a near-term reimbursement priority, making this relevant for organizations evaluating behavioral health technology adoption or value-based care strategy.
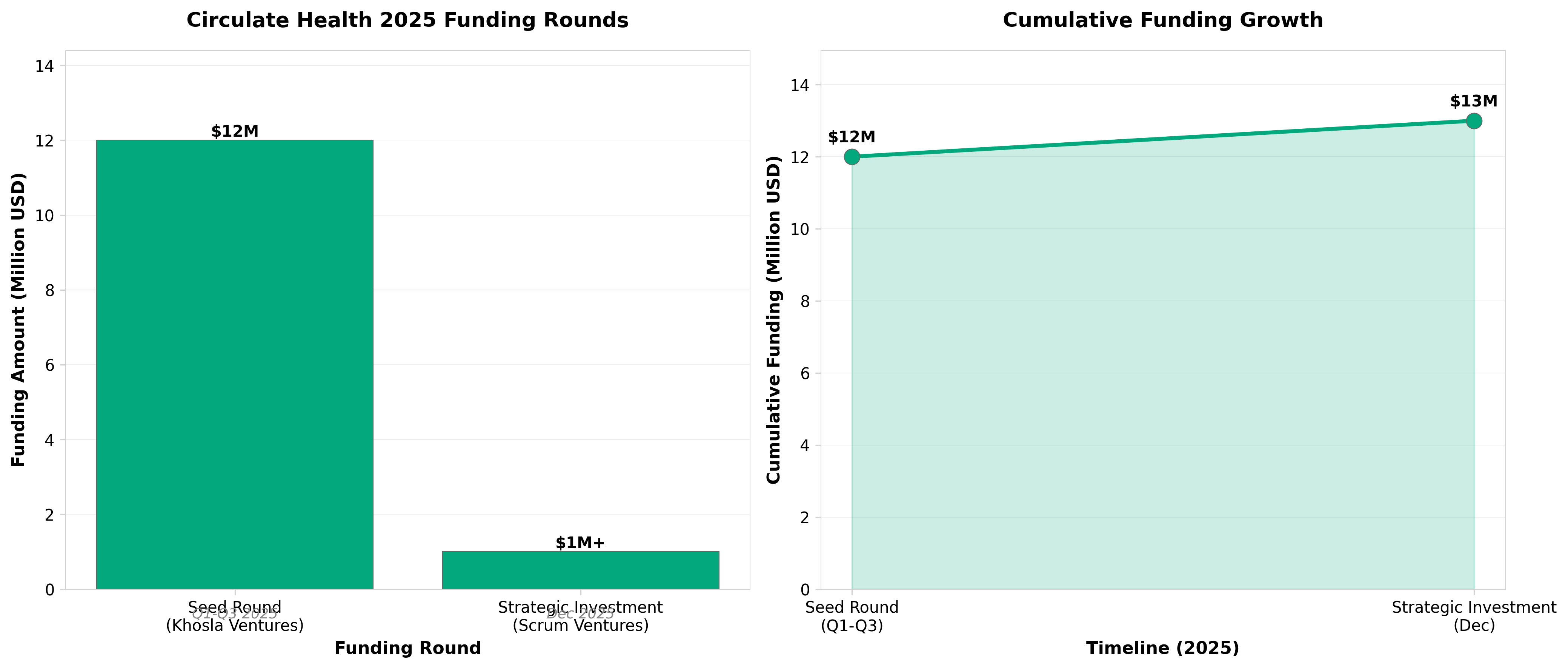
5. Circulate Health Secures Strategic Investment from Scrum Ventures to Scale TPE and Microplastics Removal Technology
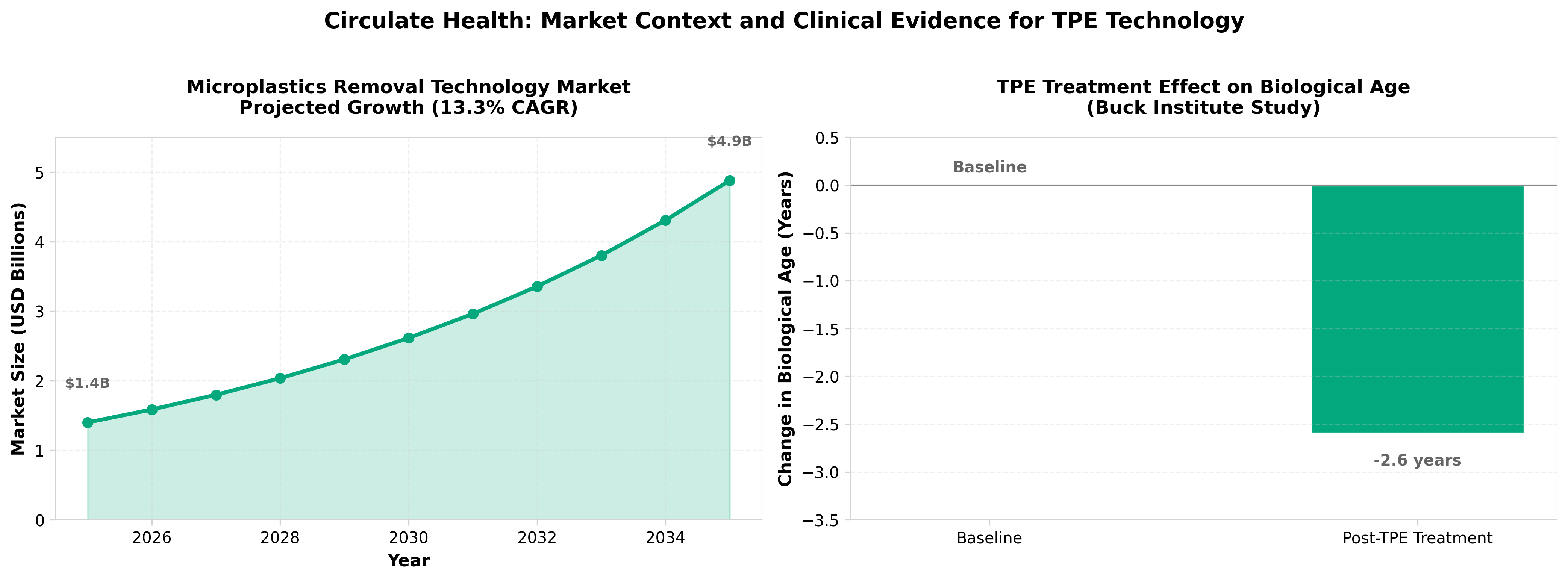
• Market timing is critical: The microplastics removal market is projected to grow from $1.4B (2025) to $4.9B (2035) at 13.3% CAGR, with biological methods expanding fastest at 14.0% CAGR—Circulate’s TPE approach positions it to capture significant share in an emerging category before filtration-based competitors dominate.
• Clinical validation drives commercial runway: A published study in Aging Cell demonstrated 2.6-year biological age reduction using TPE, providing credible evidence for the longevity positioning; this peer-reviewed proof-of-concept is essential for reimbursement discussions and enterprise partnerships as TPE scales beyond approved neurological indications.
• Investor pattern signals category validation: Back-to-back funding rounds ($12M seed from Khosla + seven-figure strategic from Scrum Ventures in 2025) from tier-one VCs focused on human performance indicates institutional confidence in TPE/microplastics as a defensible longevity category—professionals should monitor regulatory pathways and payer coverage decisions as the next validation milestone.
Clinical Studies & Translational Reports
1. Digital Biometrics in Predicting Risk for Obstructive Sleep Apnea and Hypertension: Decentralized, Prospective Cohort Study
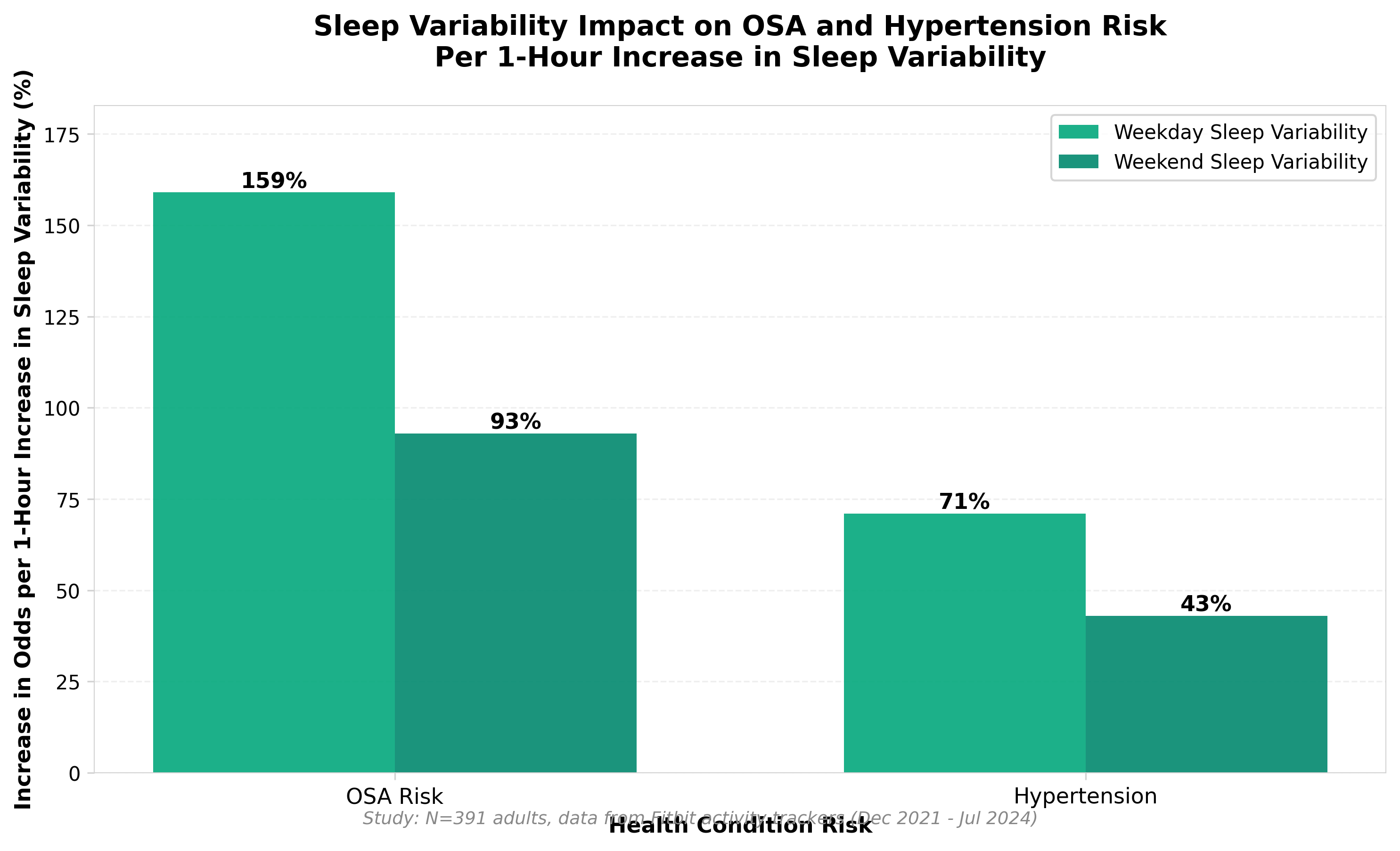
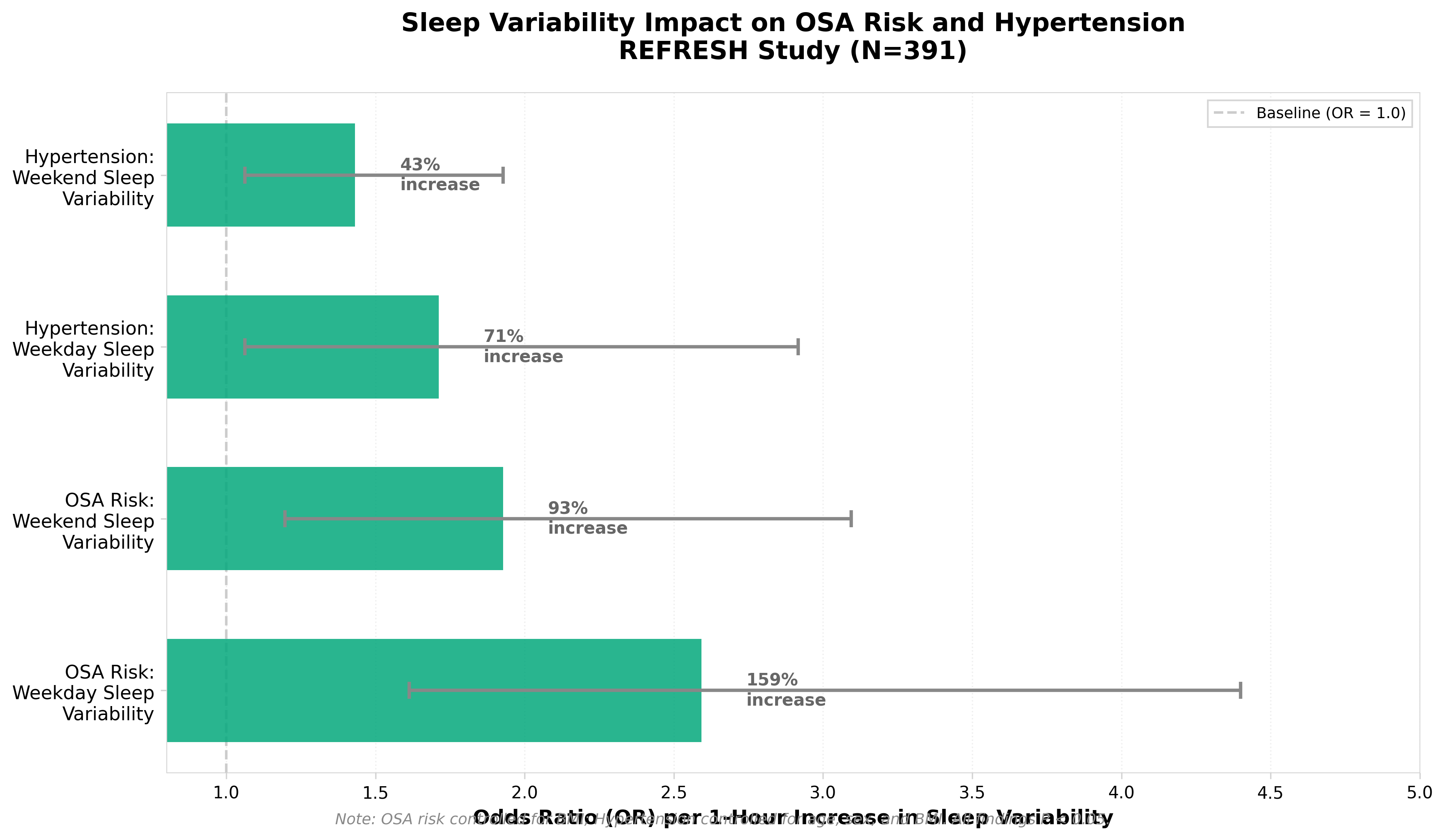
• Sleep variability is a stronger independent predictor of OSA and hypertension risk than sleep duration alone. A 1-hour increase in weekday sleep variability increased OSA risk odds by 159% and hypertension odds by 71%, independent of average sleep duration—suggesting clinical screening protocols should shift focus from duration metrics to consistency metrics when using wearable data for risk stratification.
• Decentralized digital studies can generate clinically actionable biomarkers at scale without formal diagnosis. The REFRESH study validated activity tracker data from 391 participants over ~3 years of longitudinal sleep tracking, demonstrating that consumer wearables can identify modifiable disease risk factors comparable to clinical assessments, with post hoc power of 99% for OSA prediction—establishing a replicable model for real-world evidence generation in chronic disease monitoring.
• Market conditions and consumer adoption rates now align with clinical implementation feasibility. Over 580 million wearables shipped globally in 2024 with 27% of users already leveraging devices for health monitoring, and the market is projected to reach $352 billion by 2033—creating infrastructure for rapid translation of biomarker discoveries like sleep variability into scalable preventive care interventions.
2. With Phase 3 Duchenne success, Capricor plots path forward after FDA rejection
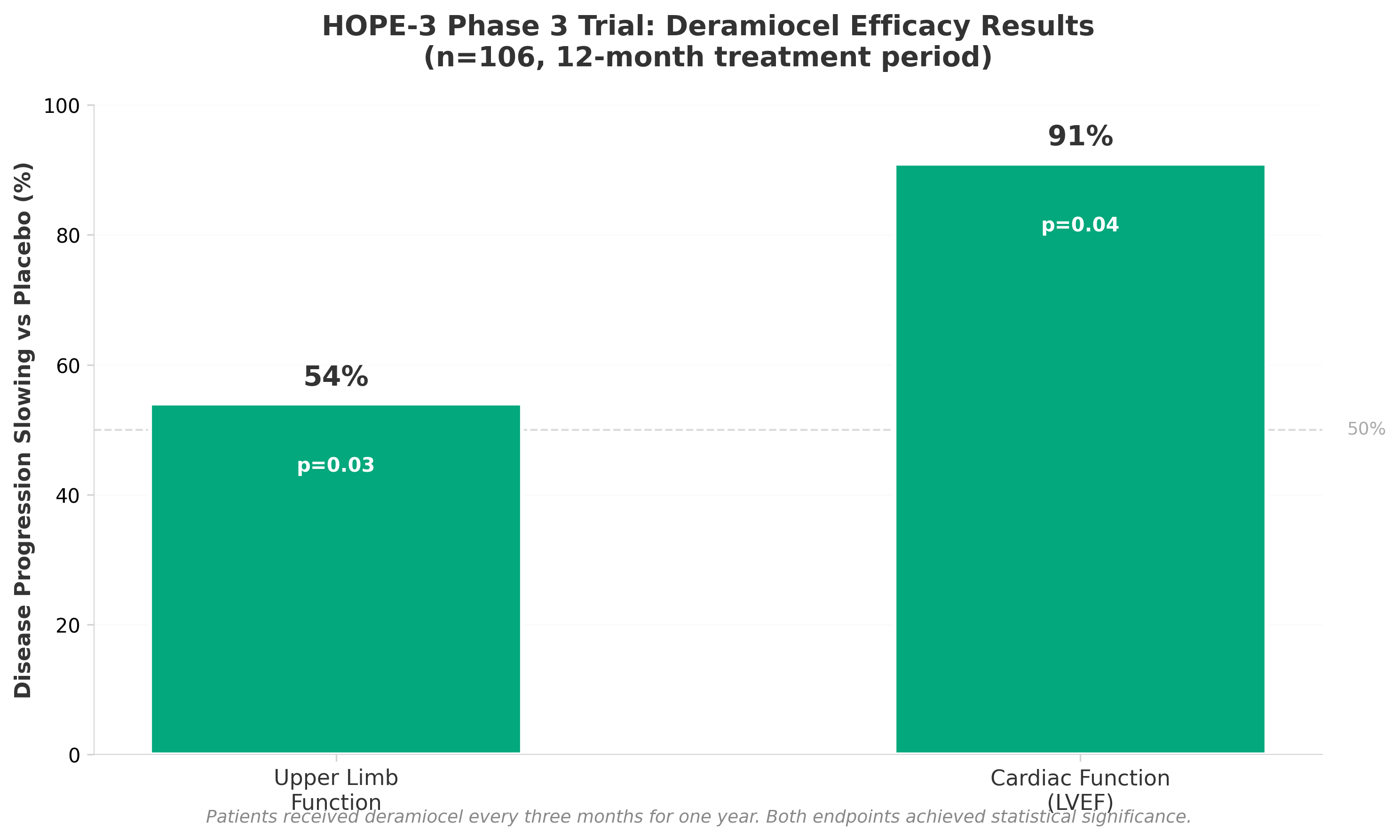
• Cardiac protection efficacy significantly outperforms skeletal muscle preservation: Deramiocel achieved 91% slowing of cardiac disease progression versus 54% for upper limb function in the HOPE-3 Phase 3 trial (p=0.04 and p=0.03 respectively), positioning cardiac preservation as the primary value proposition for resubmission to the FDA after the previous rejection.
• Market expansion window aligns with approval timeline: The DMD drugs market is projected to grow 185% from $3.47B (2023) to $9.91B (2030) at 16.8% CAGR, with ~2,000 annual diagnoses globally, providing commercial runway for first-mover advantage if Capricor gains approval in the next 12-18 months.
• Multi-year durability data strengthens regulatory case: Four+ years of continuous treatment data from HOPE-2 extension studies demonstrate sustained clinical benefits and consistent dosing tolerance, directly addressing likely FDA scrutiny around long-term safety and durability—a critical requirement for cell therapy approval in pediatric populations.
5. COVID-19 mRNA Vaccination and 4-Year All-Cause Mortality Among Adults in France
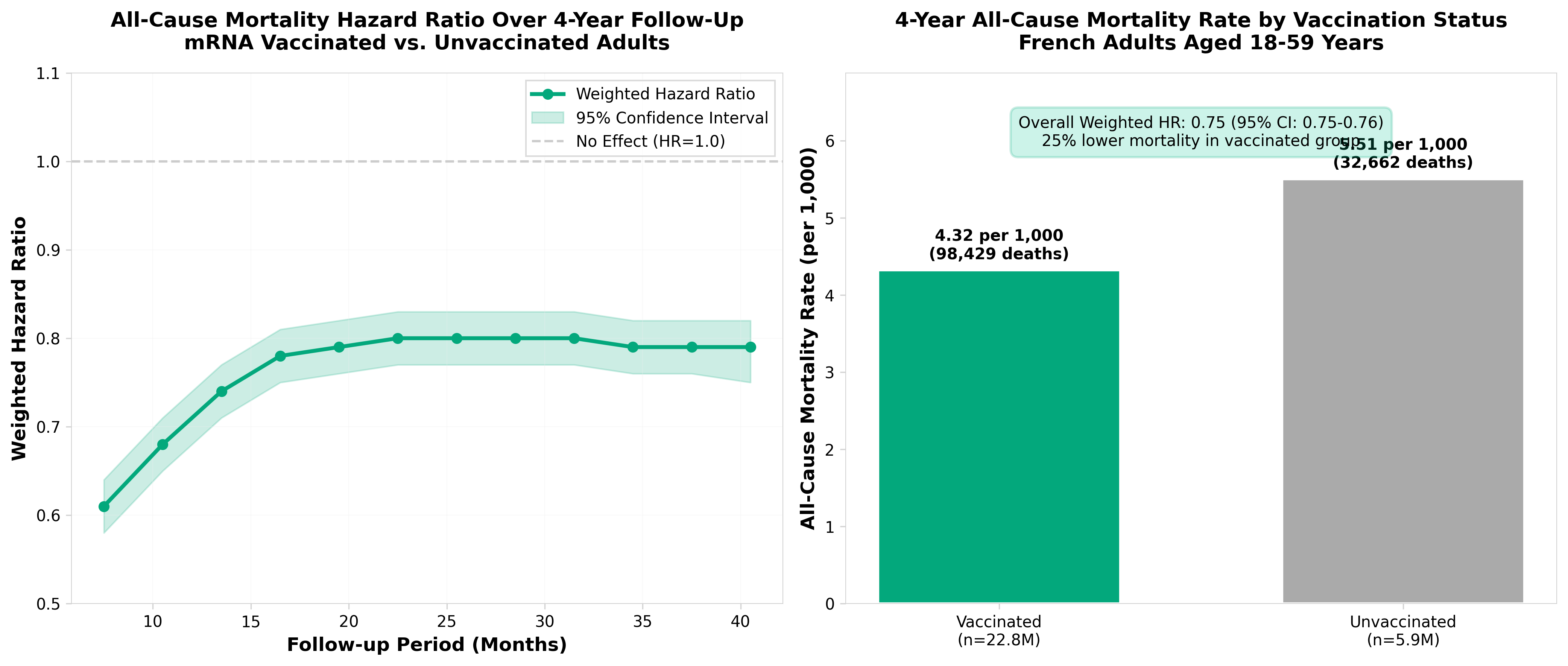
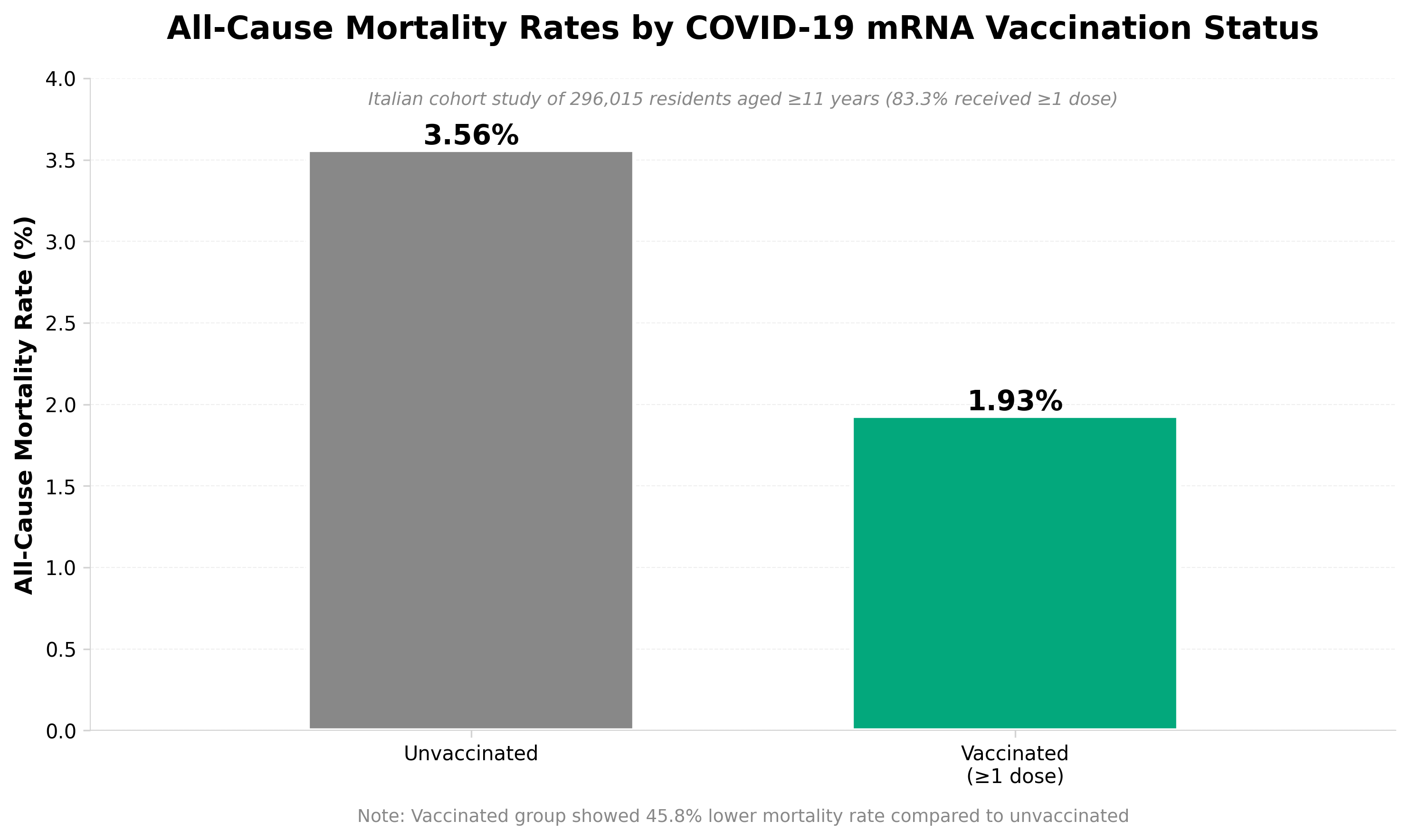
• Long-term safety profile established over 4-year follow-up: Analysis of 28 million French adults shows vaccinated individuals (18-59 years) had 25% lower all-cause mortality versus unvaccinated, with no increased mortality risk despite higher baseline comorbidities in the vaccinated group—critical data for addressing persistent safety concerns in regulatory submissions and public health communications.
• Differential vaccine effectiveness by product and time period: BNT162b2 demonstrated 27% mortality reduction versus 12% for mRNA-1273 in the same cohort; protective effect declined from 61% at 6-9 months follow-up to 20% by 39-42 months—informs real-world effectiveness expectations and booster timing strategies for clinical development programs.
• COVID-19 mortality dominates early benefits, non-COVID mortality reduction warrants investigation: Vaccinated cohort showed 74% lower risk of COVID-related death but also 25% lower all-cause mortality including cancer, circulatory, and external causes; this persistent benefit across non-COVID outcomes suggests potential protection mechanisms (reduced long COVID sequelae) requiring mechanistic investigation in future trials.
New Research
1. Deriving novel atrial fibrillation phenotypes using a tree-based artificial intelligence-enhanced electrocardiography approach
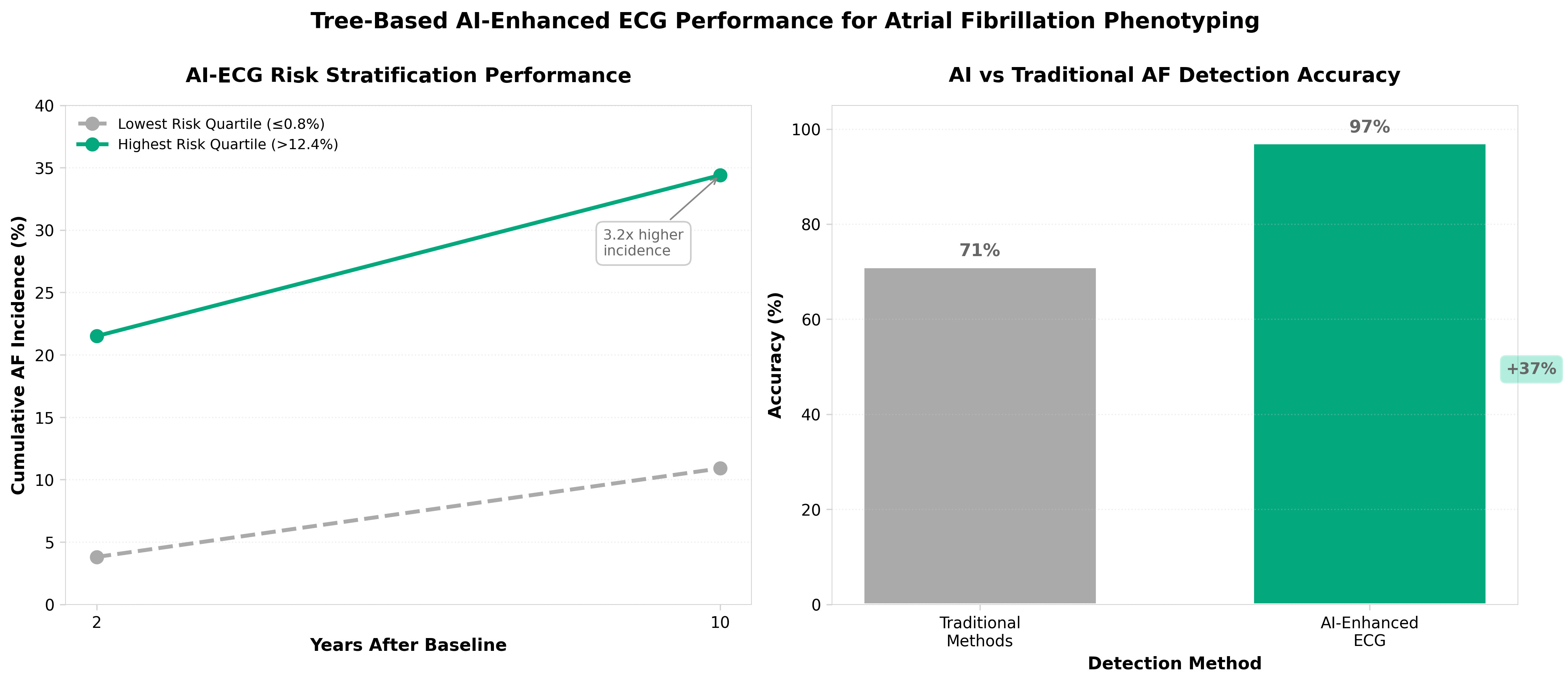
• Risk-Based AF Stratification Beyond Traditional Classification: The study identifies five distinct phenogroups with measurable prognostic differences, including a high-risk paroxysmal subgroup previously masked by duration-based classification. This enables clinicians to move beyond arrhythmia duration alone for treatment decisions, potentially improving outcomes through targeted early intervention strategies for high-risk patients currently classified as “low-risk paroxysmal.”
• Scalable AI-ECG Framework with Proven Explainability: Trained on >1.1M ECGs with tree-based clustering that preserves interpretability, this approach achieved superior discrimination compared to traditional ECG methods (40% improvement in detection accuracy per recent benchmarks). The open-source code and published methodology enable health systems to operationalize this phenotyping tool across existing ECG infrastructure without proprietary limitations.
• Clinical Heterogeneity Quantification for Personalized Management: The framework specifically identifies AF with heart failure as a distinct highest-risk phenotype with greater mortality burden, supporting evidence-based triage for early rhythm-control therapy. This addresses current gaps in AF management guidelines that lack mechanistic phenotyping, directly enabling precision medicine implementation in cardiology practices and health systems.
2. Predicting 1-year successful clinical use of an arteriovenous access for hemodialysis using machine learning
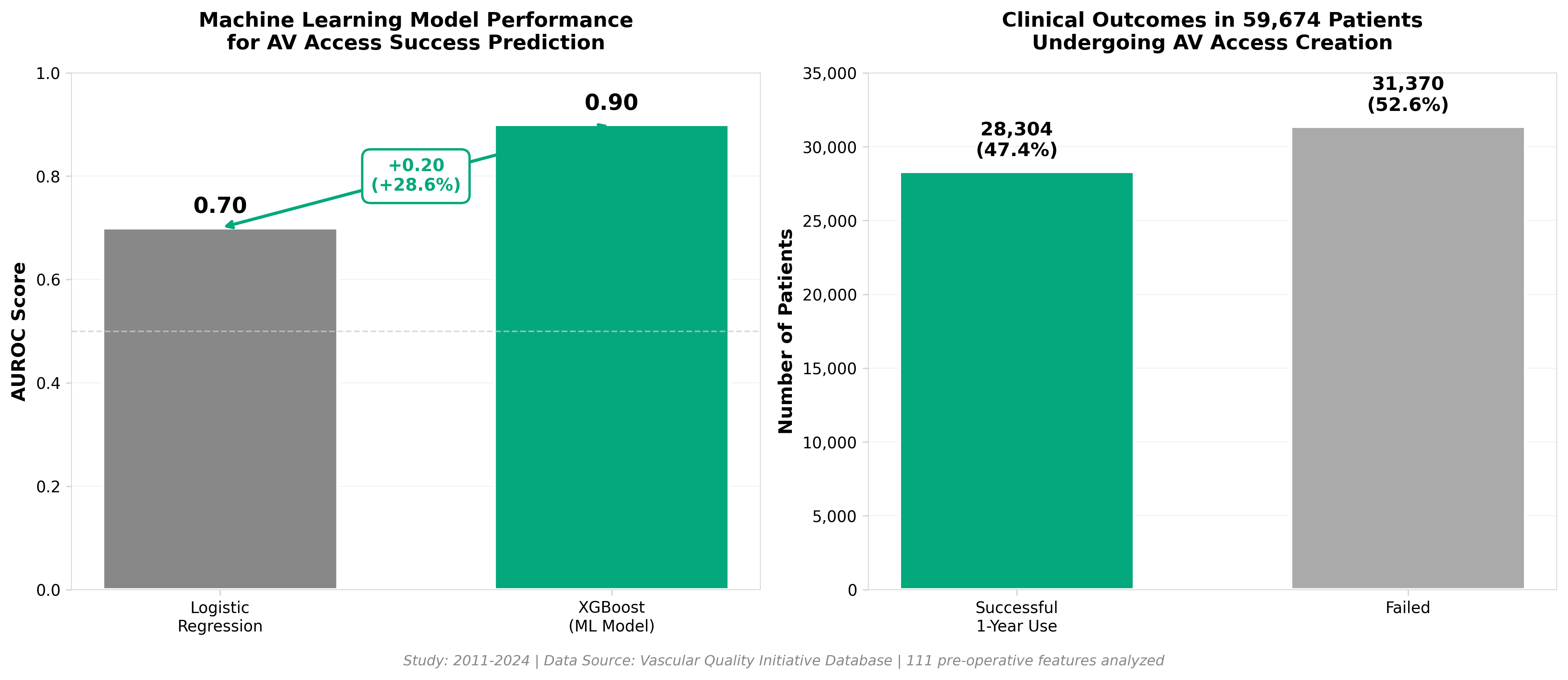
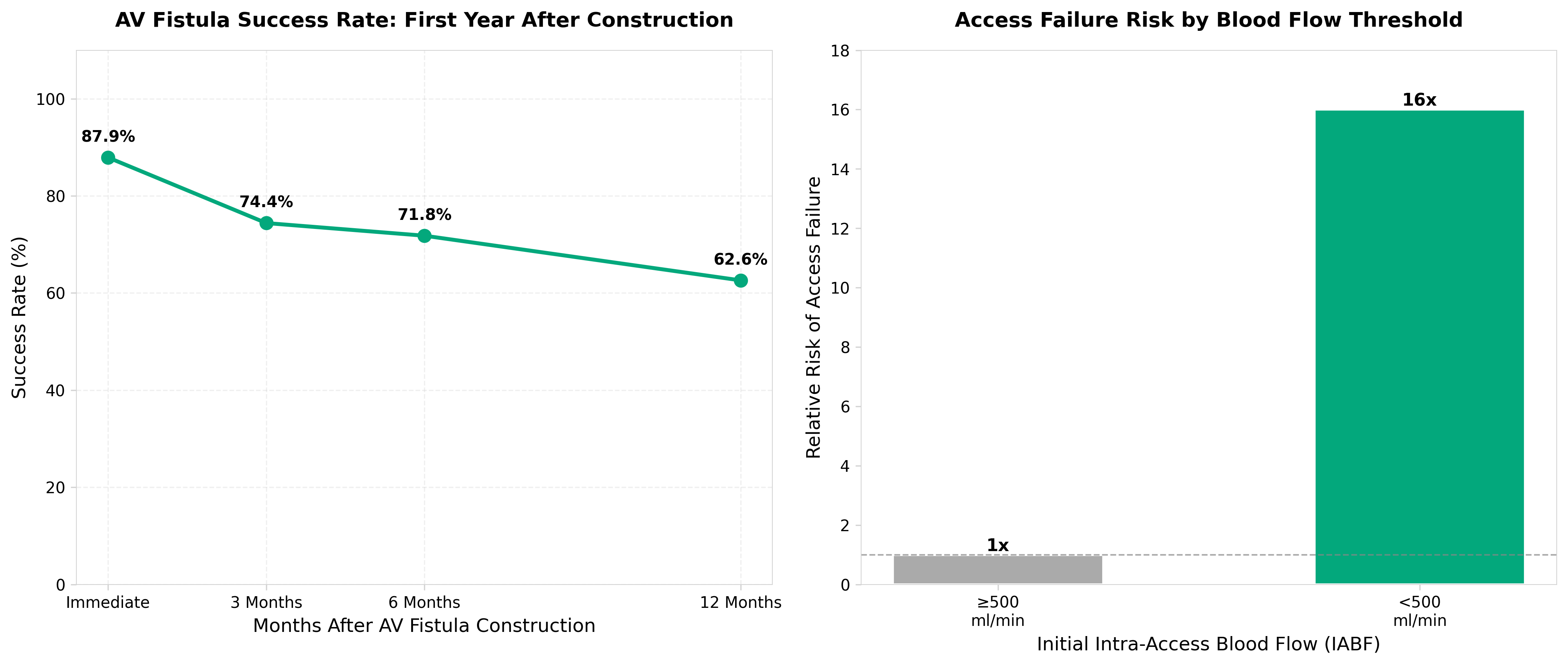
• XGBoost model achieves 0.90 AUROC vs. 0.70 for logistic regression in predicting 1-year AV access success from pre-operative data across 59,674 patients (28.3% success rate). This 29% improvement in predictive accuracy enables earlier identification of high-risk access cases during the critical 3-month maturation window when 25.6% of fistulae fail, allowing for proactive clinical interventions rather than reactive management.
• 111 pre-operative features now clinically actionable for patient stratification. The study validates that demographic, clinical, and anatomic data collected before surgery can reliably predict long-term access viability, shifting decision-making from post-operative monitoring to pre-surgical planning and patient selection—potentially reducing costly failed access attempts and associated complications.
• Real-world dataset from Vascular Quality Initiative (VQI) with public code availability accelerates adoption. Model trained on actual clinical data with open-source implementation (GitHub) and established data-access pathway through VQI enables rapid integration into institutional workflows and multi-center validation studies, reducing barriers to clinical deployment versus single-center pilot studies.
3. A bioorthogonal ligation system induces controlled proximity for cancer therapy
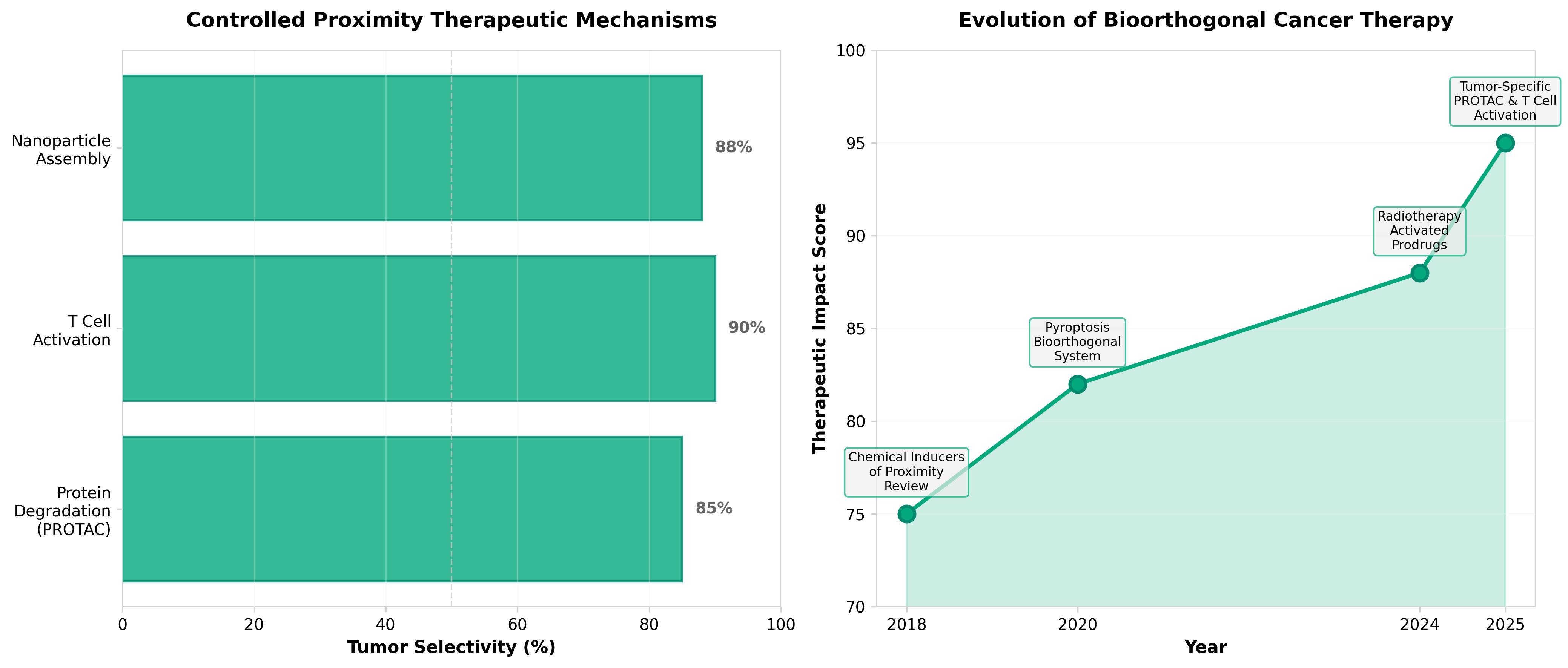
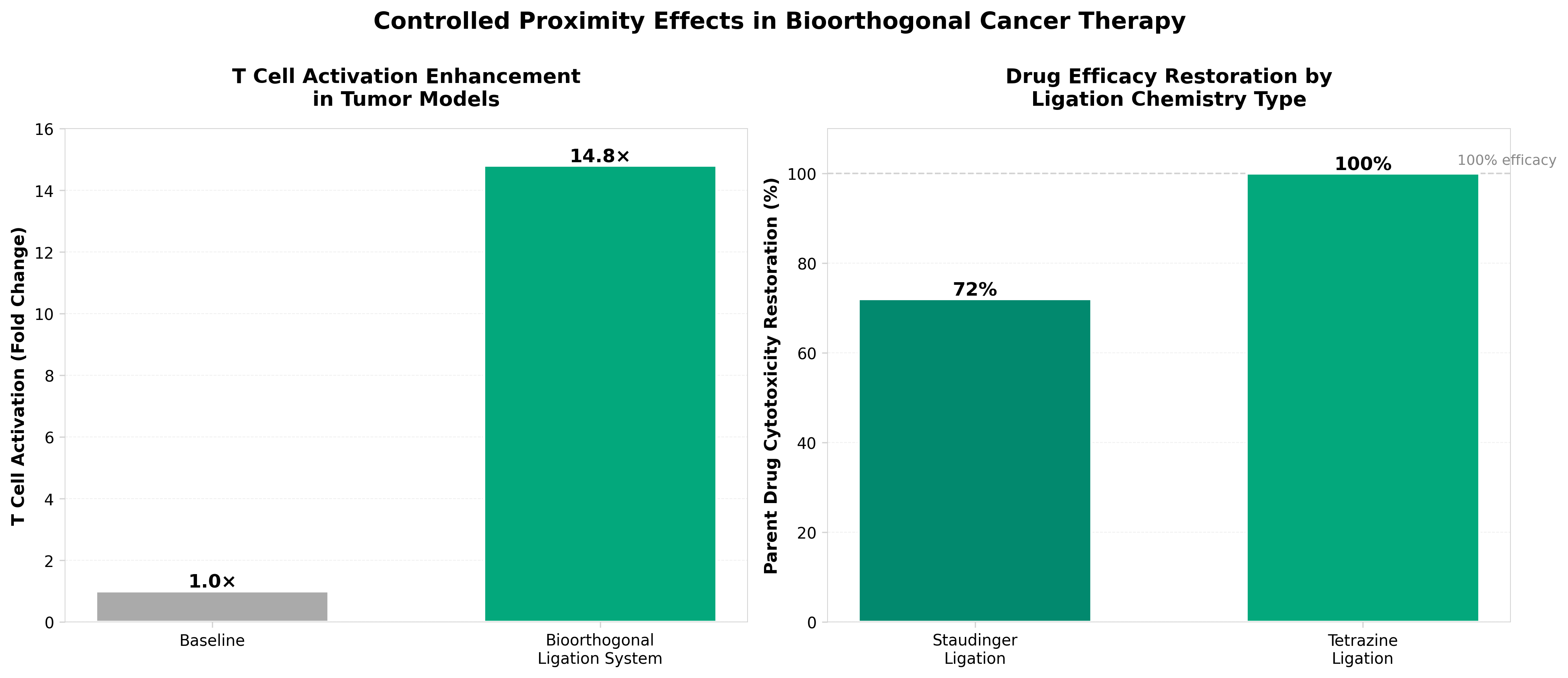
• Tumor-selective drug assembly reduces systemic toxicity while improving efficacy: Bioorthogonal ligation enables therapeutic molecules to assemble only at tumor sites, achieving 14.8-fold T cell activation enhancement and 60% tumor inhibition in preclinical models while minimizing off-target side effects—a critical advancement for addressing on-target, off-tumor toxicity that has limited CAR-T and bispecific antibody development.
• Tetrazine-based ligation chemistry achieves 100% parent drug cytotoxicity restoration: Comparative data shows tetrazine ligation outperforms Staudinger ligation (68-76% restoration), establishing a superior chemical platform for proximity-induced prodrug activation that could accelerate clinical translation of bioorthogonal therapeutics currently in early trials.
• 200+ bispecific antibodies in development signal near-term clinical validation pathway: With no approved bioorthogonal applications to date but multiple concepts already in clinical trials, this technology addresses a significant market gap in precision oncology and positions bioorthogonal chemistry as a versatile platform for controlled therapeutic assembly beyond current proteolysis-targeting chimera approaches.
4. Unlocking the potential of X-rays to scale up tissue ultrastructure mapping
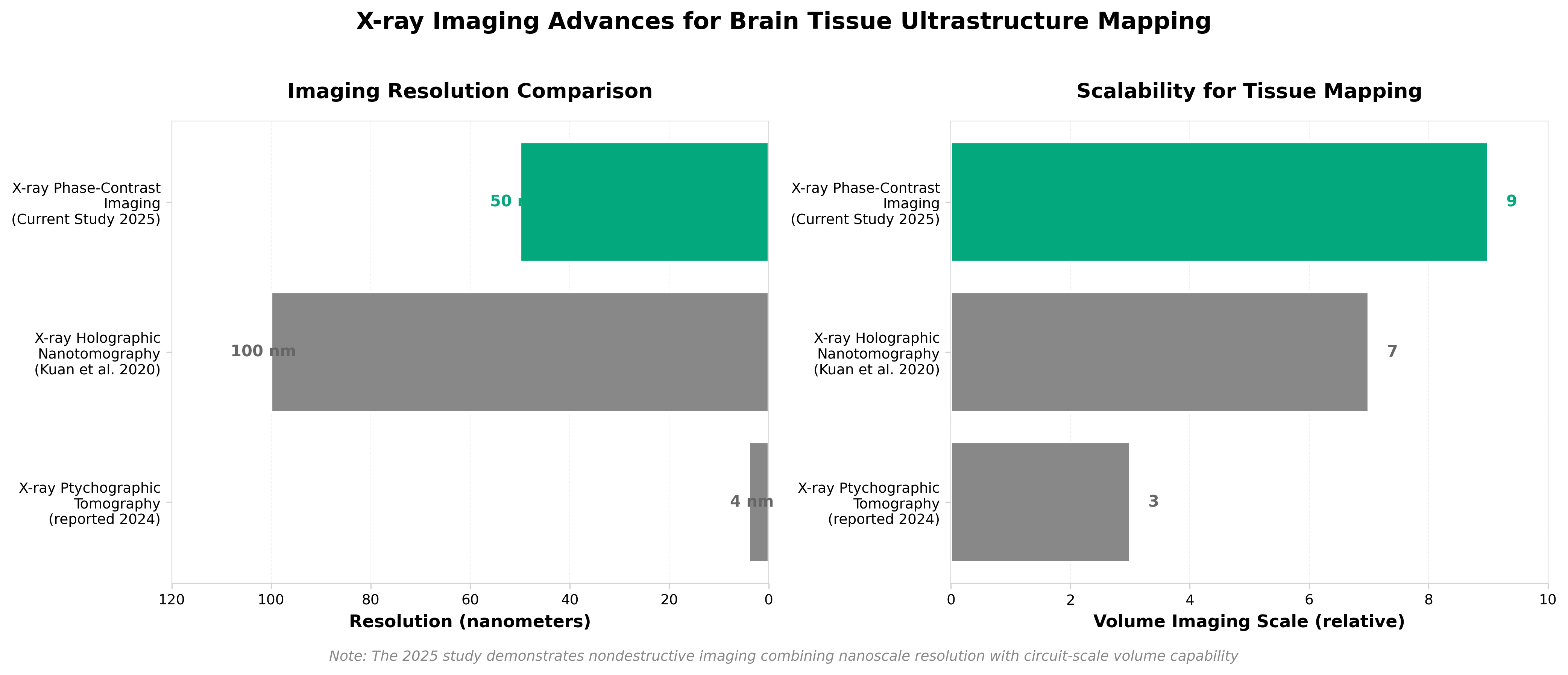
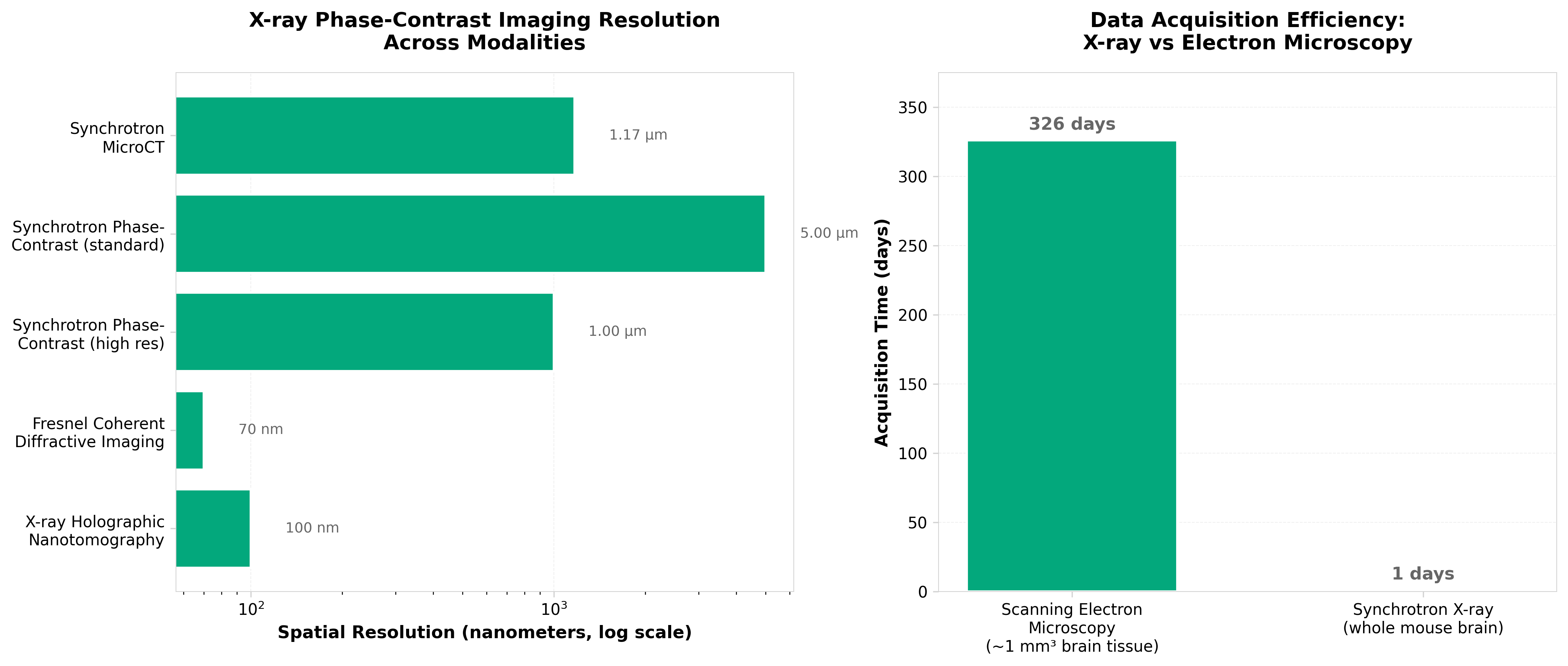
• Nondestructive 3D imaging at nanoscale resolution (sub-100nm) enables whole-tissue circuit mapping in days rather than months—coherent X-ray phase-contrast tomography eliminates sample destruction inherent to electron microscopy while delivering ~4nm voxel resolution, directly addressing the throughput bottleneck in connectomics and neuronal circuit studies where 326-day acquisition timelines currently constrain research scope.
• Synchrotron-based X-ray phase-contrast imaging shifts the technology bottleneck from resolution to accessibility and standardization—with established 4-5µm synchrotron capabilities already validated for whole-brain imaging in single-day sessions, the near-term commercial opportunity lies in making beamline access routine for life sciences rather than developing new detection capabilities, creating demand for protocol standardization and computational infrastructure for petabyte-scale datasets.
• Market expansion potential of 7.8% CAGR (USD 14.6B to USD 24.7B by 2032) in X-ray imaging positions phase-contrast tomography as a high-growth specialty segment, with immediate applications in brain connectomics, tissue regeneration mapping, and disease pathology studies that currently lack scalable nondestructive alternatives—institutional adoption hinges on beamline democratization and standardized image reconstruction pipelines rather than fundamental technology
5. C-COMPASS: a user-friendly neural network tool profiles cell compartments at protein and lipid levels
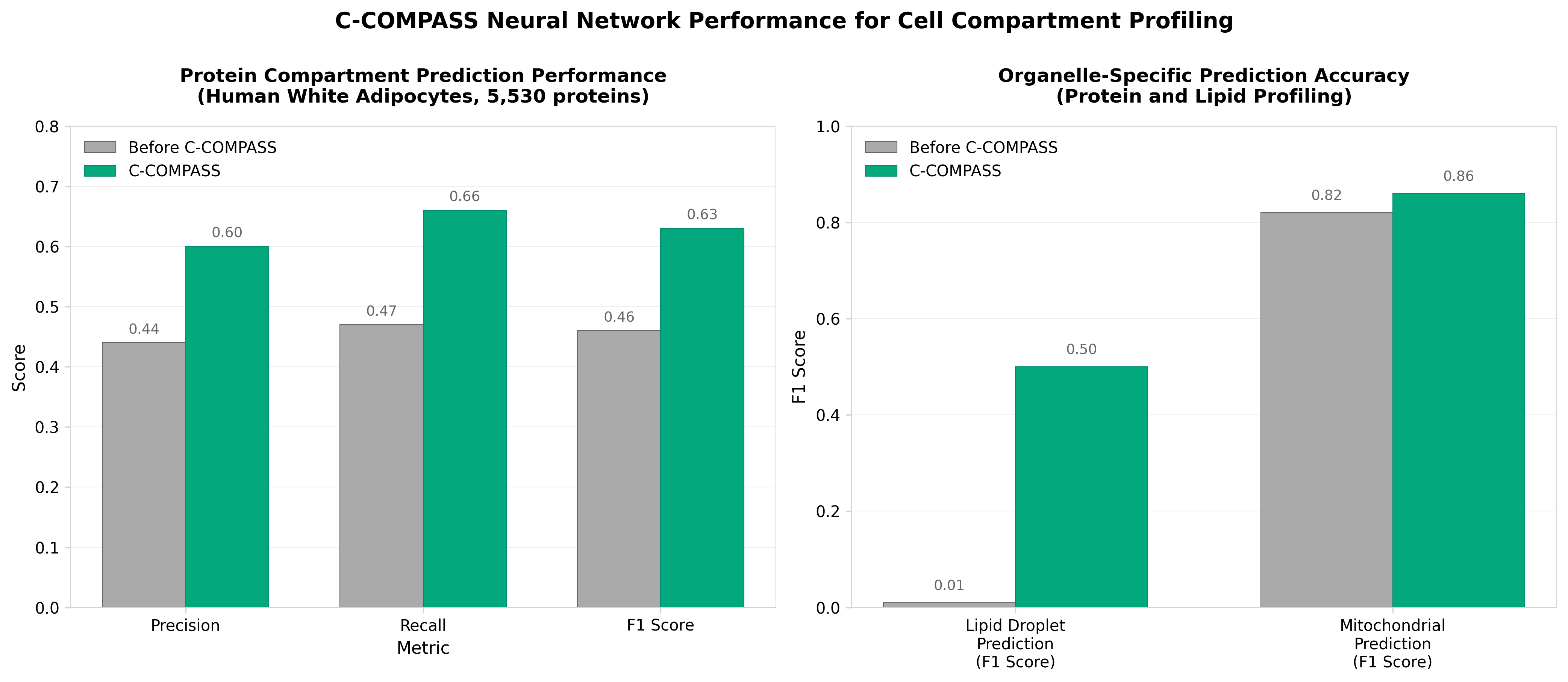
• Quantitative multilocalization mapping addresses a critical gap in spatial proteomics: C-COMPASS is the first tool to provide quantitative (rather than probabilistic) predictions of protein distribution across multiple organelles, with ~50-60% of cellular proteins exhibiting multiple localizations—a complexity that existing SVM-based methods struggle to capture accurately.
• Superior performance on underrepresented organelles improves data quality across cell types: The neural network approach with upsampling increased prediction accuracy for lipid droplet proteins from F1 < 0.01 to 0.50 and outperformed established methods (SVMs, BANDLE, DOM-ABC) in dual and triple-localization scenarios, enabling more reliable organellar profiling in diverse experimental contexts.
• Integrated proteomics-lipidomics capability enables comprehensive organellar characterization without established lipid markers: C-COMPASS extends neural networks trained on protein markers to predict lipid localization across 411 lipid species, revealing organelle-specific fatty acid chain compositions (e.g., longer, more unsaturated PG species in lysosomes vs. shorter, saturated versions in mitochondria) and identifying previously uncharacterized lipid-organelle associations.
