Headlines & Launches
- OpenAI launches biotech-specific AI model dubbed GPT-Rosalind
- Novartis’ Vas Narasimhan heads to Anthropic’s board
- Scribing startup Abridge adds NEJM, JAMA as it moves into medical AI search
- FDA will reclassify at least a dozen peptides, teeing up potential telehealth win
- Walmart Expands Better Care Services Platform with GLP-1 Weight Management Offerings
Deep Dives & In Depth Analysis
- Machine Learning Model to Predict Postmastectomy Breast Reconstruction Complications
- AI Will Accelerate Drug Discovery by Accelerating Clinical Evidence
- Conversational AI Agent for Psychiatric Symptoms and Digital Therapeutic Alliance
- Validation of the Very Favorable IMDC Risk Group in Metastatic Renal Cell Carcinoma
New Research
- Steering semi-flexible molecular diffusion model for structure-based drug design with reinforcement learning
- Astrocyte-based CAR immunotherapy against Alzheimer’s disease
- Phenotypic prediction of missense variants via deep contrastive learning
- AI Augmented Confocal Laser Endomicroscopy for Rapid Intraoperative Diagnosis of Brain Tumors
- Predictive models for the occurrence and lymph node metastasis of papillary thyroid carcinoma with regional risk heterogeneity
Market Indicators
| | Price (USD) | 1D | YTD | | :— | :— | :— | :— | | Company | | | | | Eli Lilly and Company (LLY) | 934.60 | +9.80% | | Novartis AG (NVS) | 147.85 | +3.44% | | Novo Nordisk A/S (NVO) | 42.22 | +4.79% | | Walmart Inc. (WMT) | 131.93 | +3.06% | | Sector Index | | | | | iShares Biotechnology ETF (IBB) | 168.69 | +2.00% | | State Street SPDR S&P Biotech ETF (XBI) | 131.33 | +1.89% | | State Street SPDR S&P Pharmaceuticals ETF (XPH) | 57.61 | +1.89% | | Vanguard Health Care Index Fund ETF Shares (VHT) | 272.46 | +2.12% | | General Market Index | | | | | Russell 2000 Index (^RUT) | 2799.91 | +2.21% | | Invesco QQQ Trust (QQQ) | 667.74 | +0.93% | | State Street SPDR S&P 500 ETF Trust (SPY) | 718.66 | +0.99% | —
Headlines & Launches
1. OpenAI launches biotech-specific AI model dubbed GPT-Rosalind
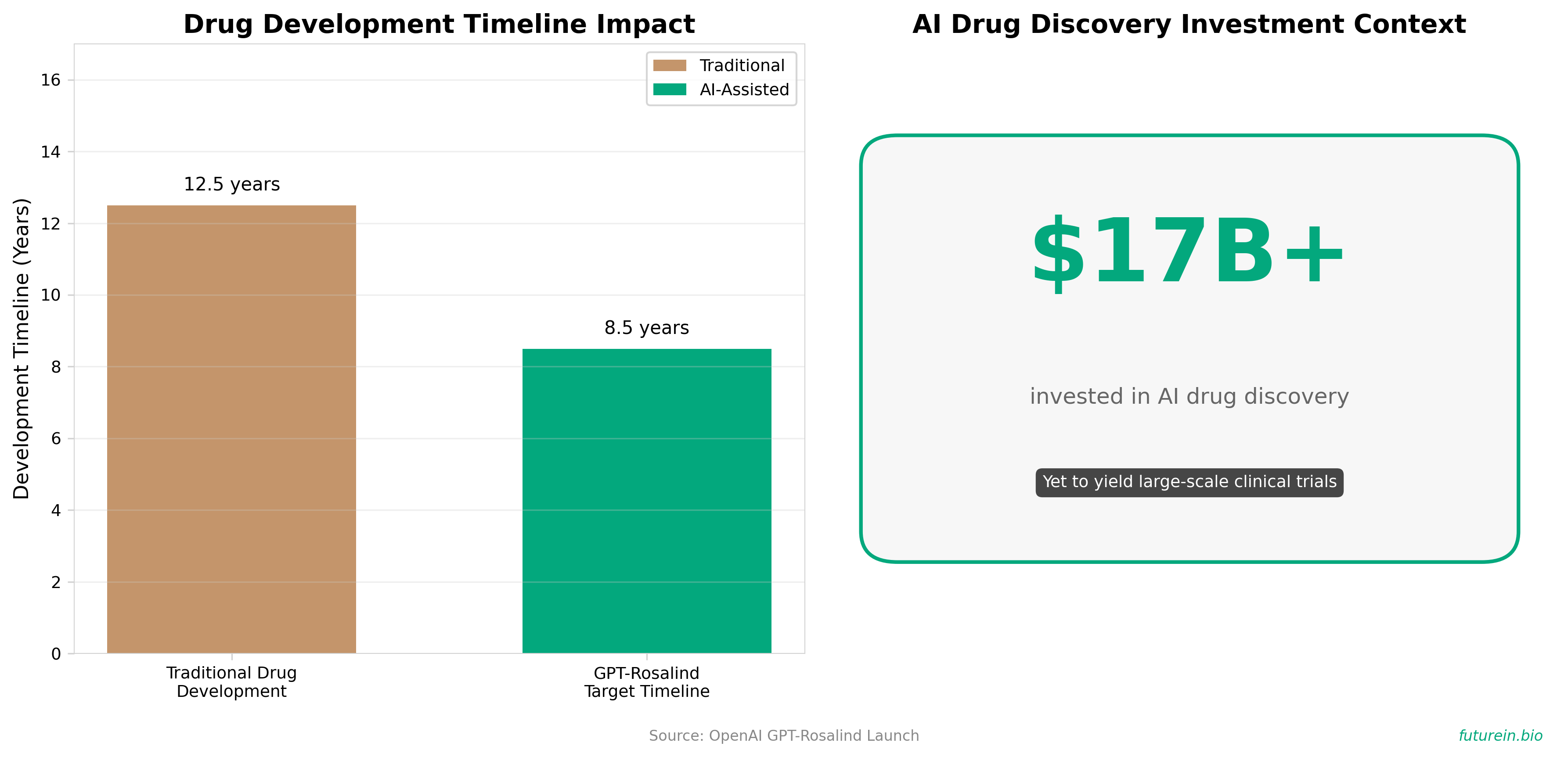
• Biotech-specific AI model enters competitive drug discovery landscape with major pharma adoption. GPT-Rosalind is designed to compress the 10-15 year drug development timeline by accelerating target discovery and literature synthesis, with early adoption from Amgen, Moderna, and Eli Lilly signaling industry confidence—though $17B+ in AI drug discovery investments have yet to yield large-scale clinical trials.
• Trusted access program limits immediate availability but establishes foundation for enterprise integration. The model is accessible only through OpenAI’s controlled research preview and API, meaning organizations must qualify for access; this differs from general ChatGPT availability and suggests potential premium pricing or partnership requirements for production-scale deployment.
• OpenAI positions this as first in an expanding life sciences suite, not a standalone tool. The company explicitly states plans to develop additional models with enhanced biochemical reasoning, indicating a strategic shift toward vertical-specific AI products rather than generalist models—creating opportunities for early adopters to shape requirements but also risk of tool proliferation across R&D workflows.
2. Novartis’ Vas Narasimhan heads to Anthropic’s board; Metsera alum joins Structure as COO
• AI integration in drug development is becoming institutionalized: Anthropic’s appointment of Novartis’ CEO to its board signals major pharma’s commitment to embedding AI capabilities at the executive level, indicating this is moving beyond pilot programs to core strategy.
• Strategic consolidation in AI-biotech is accelerating: Anthropic’s acquisition of Coefficient Bio demonstrates larger AI platforms are directly acquiring specialized biotech capabilities rather than partnering, suggesting companies need to either build internal AI capacity or prepare for acquisition as a competitive necessity.
3. Scribing startup Abridge adds NEJM, JAMA as it moves into medical AI search
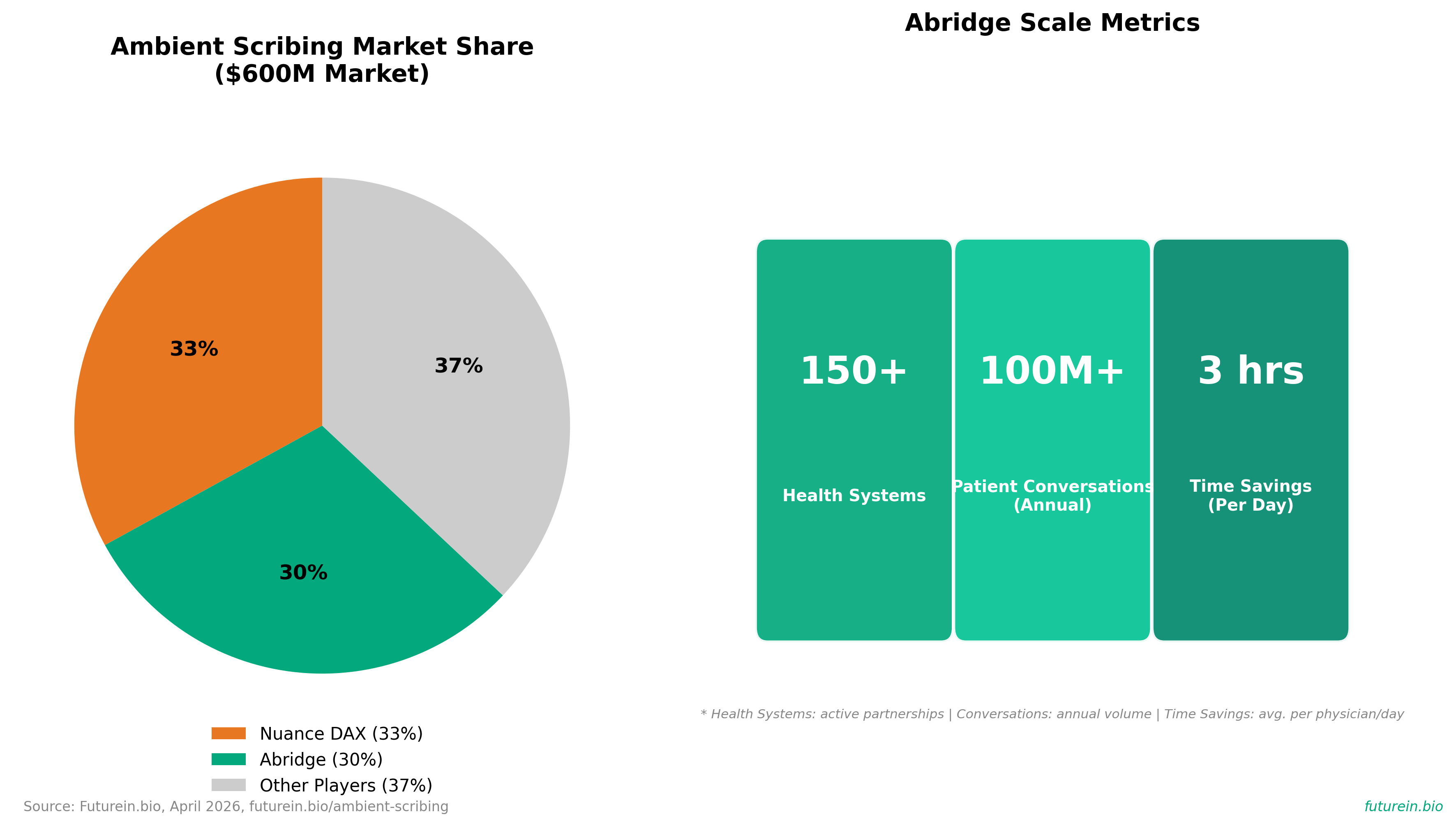
• Medical AI market consolidation around ambient scribing: Abridge commands 30% market share in a $600M ambient documentation category (up 2.4x YoY), positioning itself as a dominant player competing against incumbent Nuance DAX (33% share). This indicates the market is concentrating among 2-3 major players, raising barriers to entry for new competitors and suggesting consolidation risk for smaller vendors.
• Rapid clinical adoption at scale: Abridge deployed across 150+ health systems (50% growth in 4 months) and is contracted to support 100M+ patient conversations annually, with reported 3-hour daily documentation time savings per physician. This demonstrates validated product-market fit and quantifiable ROI that directly improves clinician workflows—critical for enterprise healthcare adoption.
• Journal partnerships signal shift toward integrated clinical decision support: Adding NEJM and JAMA as content partners moves Abridge from documentation-only toward an all-in-one platform combining ambient scribing, research access, and AI search. This expansion targets the competitive threat from OpenEvidence and reflects industry movement toward consolidating disparate clinical tools into unified physician platforms.
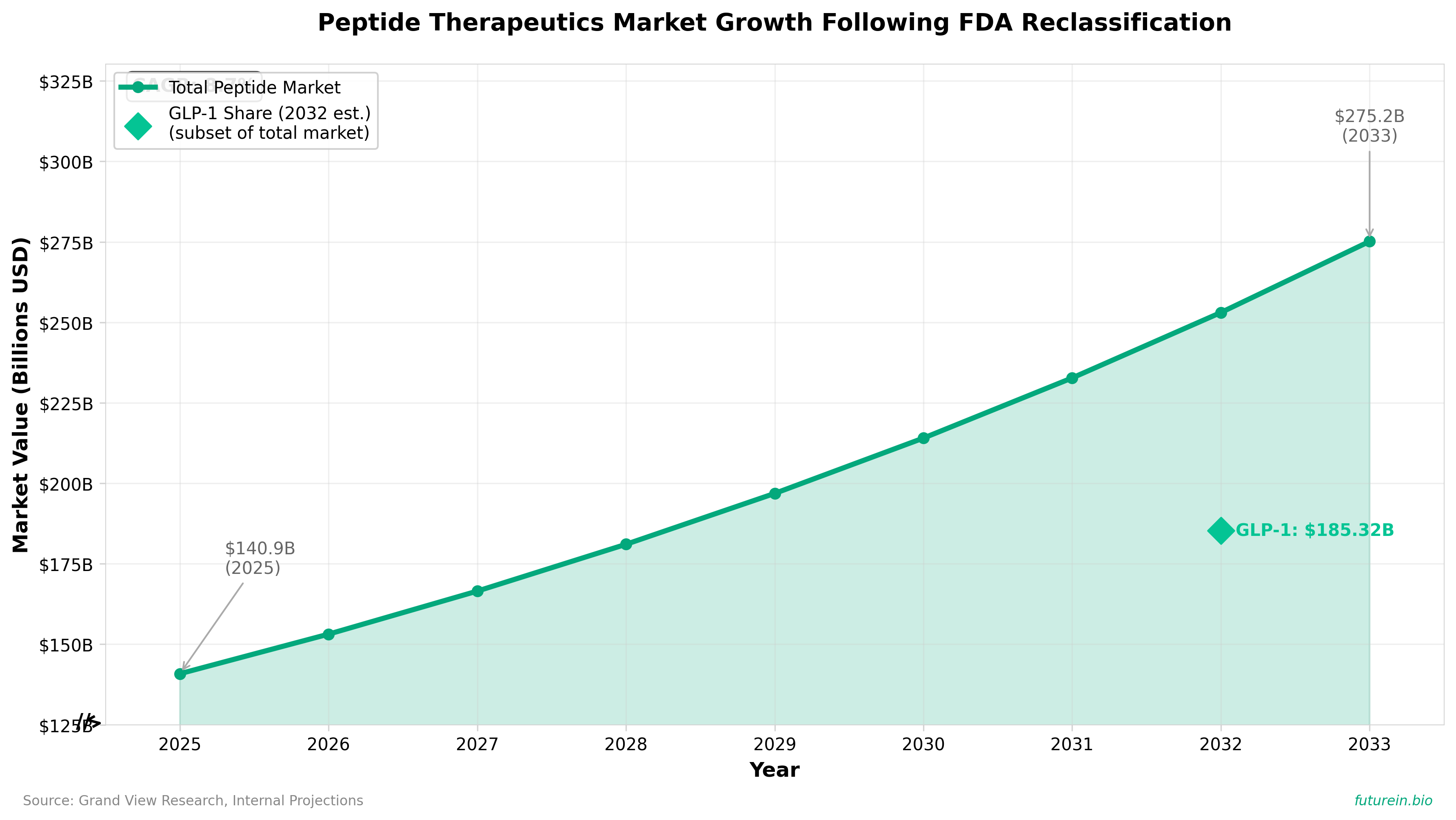
4. Updated: FDA will reclassify at least a dozen peptides, teeing up potential telehealth win
• FDA reclassification of ~14 peptides from Category 2 to Category 1 status removes compounding restrictions and unlocks a $294.58B market opportunity by 2033 (growing from $140.86B in 2025 at 8.73% CAGR), with GLP-1 compounds alone projected to reach $185.32B by 2032—directly enabling telehealth distribution models for previously opaque, cash-pay peptide therapies.
• North America controls 61.99% of the global peptide therapeutics market with the U.S. segment valued at $21.24B (2025) and projected to reach $34.98B by 2035, establishing a massive addressable market for telehealth platforms operating in a region with established regulatory clarity and high commercial demand.
• Major pharmaceutical manufacturers are deploying $1.1B+ in capital investments (CordenPharma, Amneal) to expand peptide synthesis capacity, signaling supply-side confidence that FDA reclassification will drive sustained demand and lower manufacturing costs—critical infrastructure for scaling telehealth-distributed peptide therapies at competitive pricing.
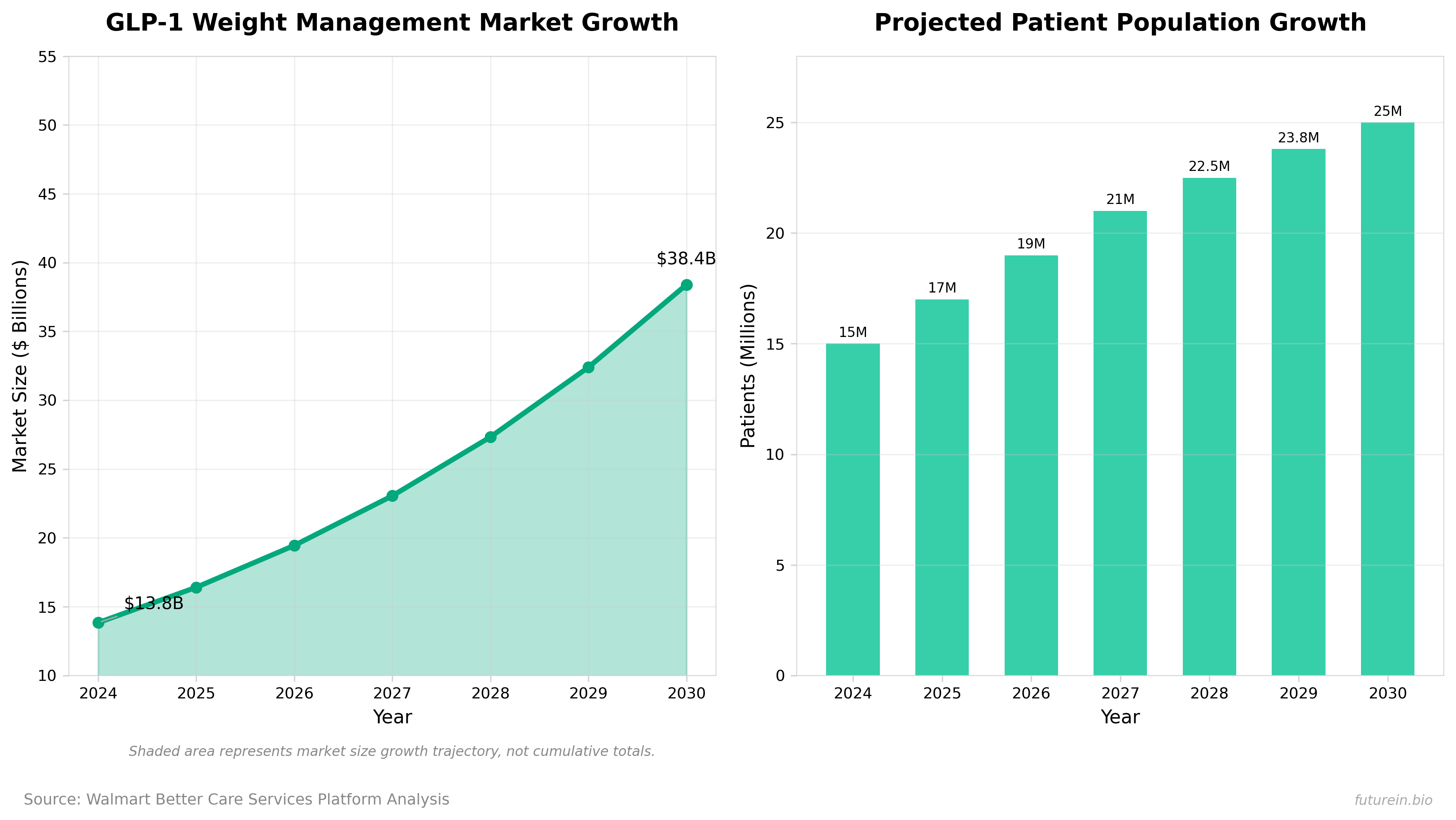
5. Walmart Expands Better Care Services Platform with GLP-1 Weight Management Offerings
• Market timing advantage: The GLP-1 weight-loss market is projected to grow from $13.84B (2024) to $48.84B by 2030 at 18.54% CAGR, with patient population expected to reach 25 million Americans by 2030—Walmart’s integrated platform positions it to capture significant share as the market scales and adoption accelerates beyond early adopters.
• Ecosystem bundling as competitive moat: By consolidating medication fulfillment, nutrition guidance (via Walmart Everyday Health Signals), fitness (Aaptiv), and clinical support in one platform, Walmart addresses a critical market gap where GLP-1 medication alone achieves only 17.5% of patients at ≥10% weight loss—the bundled approach increases stickiness and lifetime customer value in a market where adherence and outcomes drive retention.
• Pharmacy network leverage: Walmart’s 4,600 pharmacies represent 60% of retail pharmacy market share, creating a direct distribution and patient touchpoint advantage that competitors like telehealth-only GLP-1 providers cannot replicate, enabling data integration across medication dispensing, clinical outcomes, and consumer behavior to refine personalized recommendations.
Deep Dives & In Depth Analysis
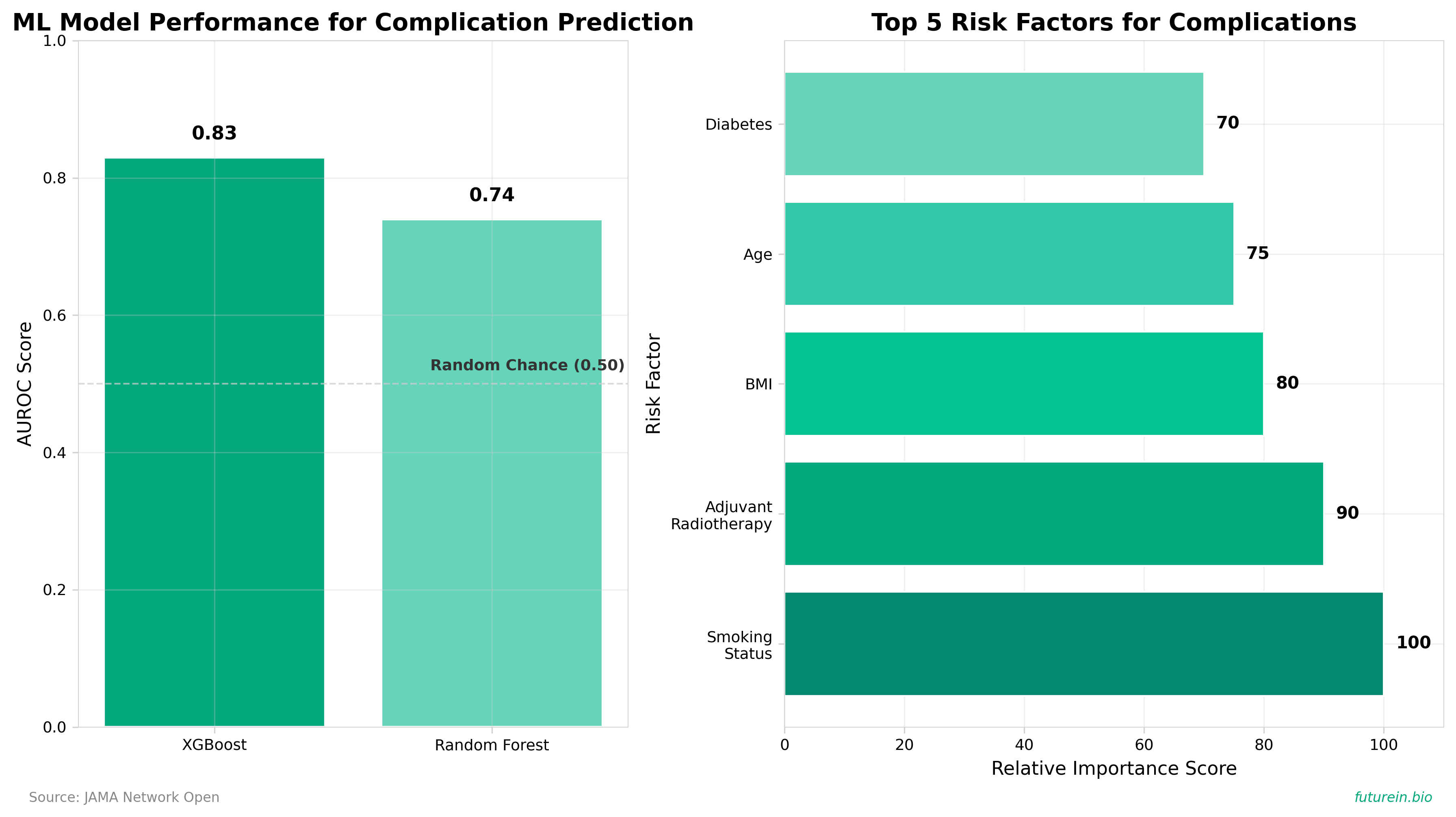
1. Machine Learning Model to Predict Postmastectomy Breast Reconstruction Complications
• XGBoost ML models achieved 0.83 AUROC for predicting major complications (25.8% baseline rate) across both implant and autologous reconstruction types, outperforming random forest (0.74 AUROC). This consistency across reconstruction modalities creates a pathway for integrated clinical decision-support tools that don’t require separate models by procedure type, reducing implementation complexity.
• Five modifiable and non-modifiable risk factors emerged as primary complication predictors: smoking status, adjuvant radiotherapy, BMI, age, and diabetes. The top 2-3 factors were consistent across both algorithms, enabling targeted preoperative interventions (smoking cessation, weight optimization) and risk-stratified counseling that directly addresses patient information gaps identified in the literature.
• Manual abstraction of unstructured clinical notes proved critical for accurate outcome attribution (distinguishing index reconstruction complications from unrelated events), but limits scalability to 411 patients from 4000+ eligible. Adopting automated text extraction methods (large language models, NER systems) is essential for external validation studies and broader implementation, as current labor-intensive processes prevent deployment across diverse clinical settings.
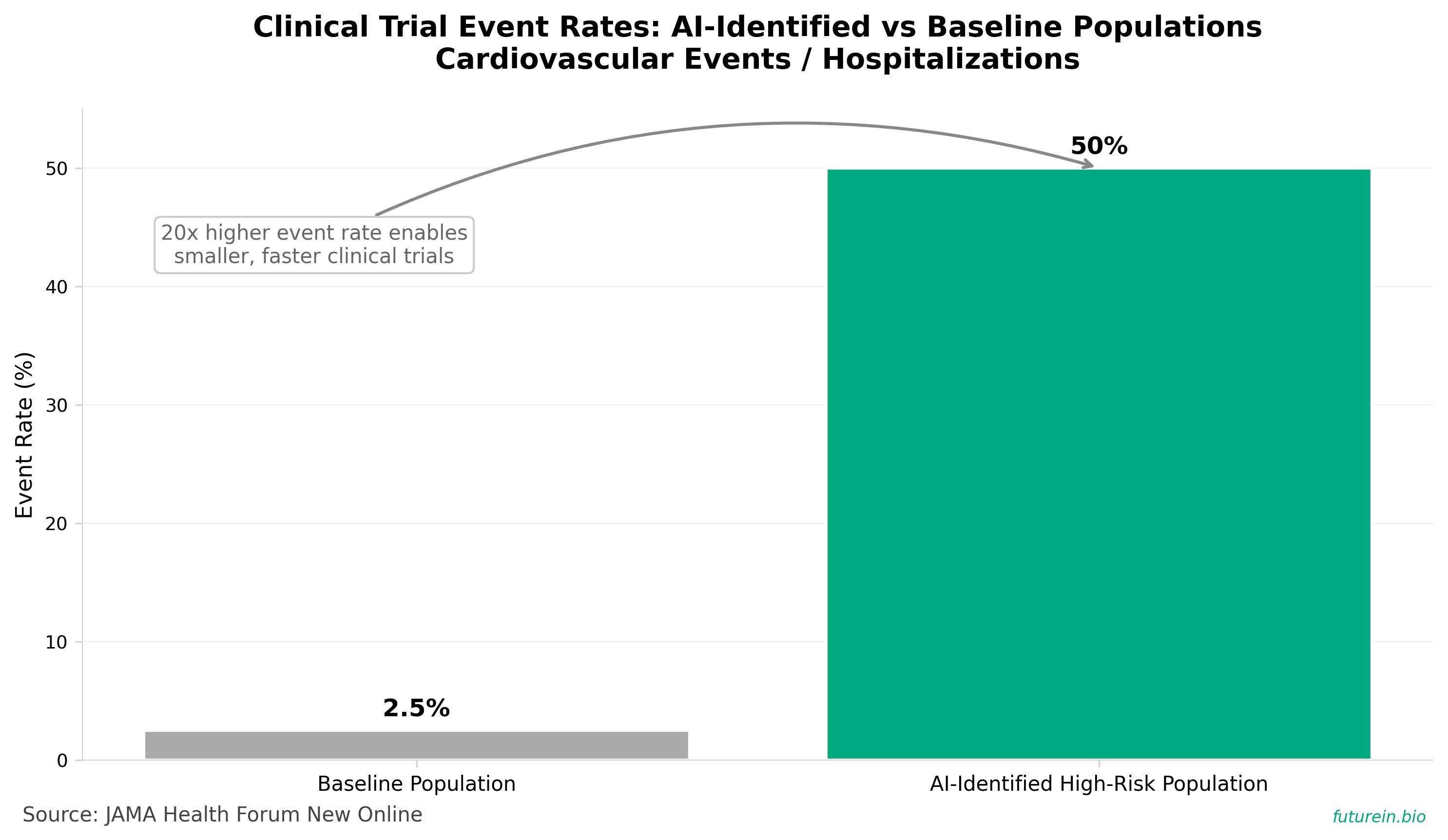
2. AI Will Accelerate Drug Discovery by Accelerating Clinical Evidence
• AI-derived surrogate endpoints can dramatically reduce clinical trial size and duration: AI predictions from existing physiological data (ECGs, imaging, lab results) can identify high-risk populations with 40-60% event rates versus 2-3% baseline rates, enabling smaller, faster, cheaper trials—critical for prevention and early-intervention indications that sponsors currently avoid due to enrollment costs.
• Data-driven surrogate outcomes bypass failed mechanistic approaches but require rigorous validation: Unlike traditional surrogates that rely on causal understanding (many of which have caused patient harm), AI surrogates leverage comprehensive physiological measurement to predict outcomes empirically; however, they remain surrogates and must still be validated against true clinical endpoints to avoid reproducing training data biases across patient subgroups.
• Clinical trial acceleration is now the primary bottleneck for drug delivery, not laboratory discovery: While AI gains headlines for protein folding and molecular design, the actual barrier to patient access is the enrollment and evaluation phase; addressing this through AI-optimized trial design and outcome measurement directly unlocks treatment options for high-burden, previously “undrugged” conditions.
3. Conversational AI Agent for Psychiatric Symptoms and Digital Therapeutic Alliance
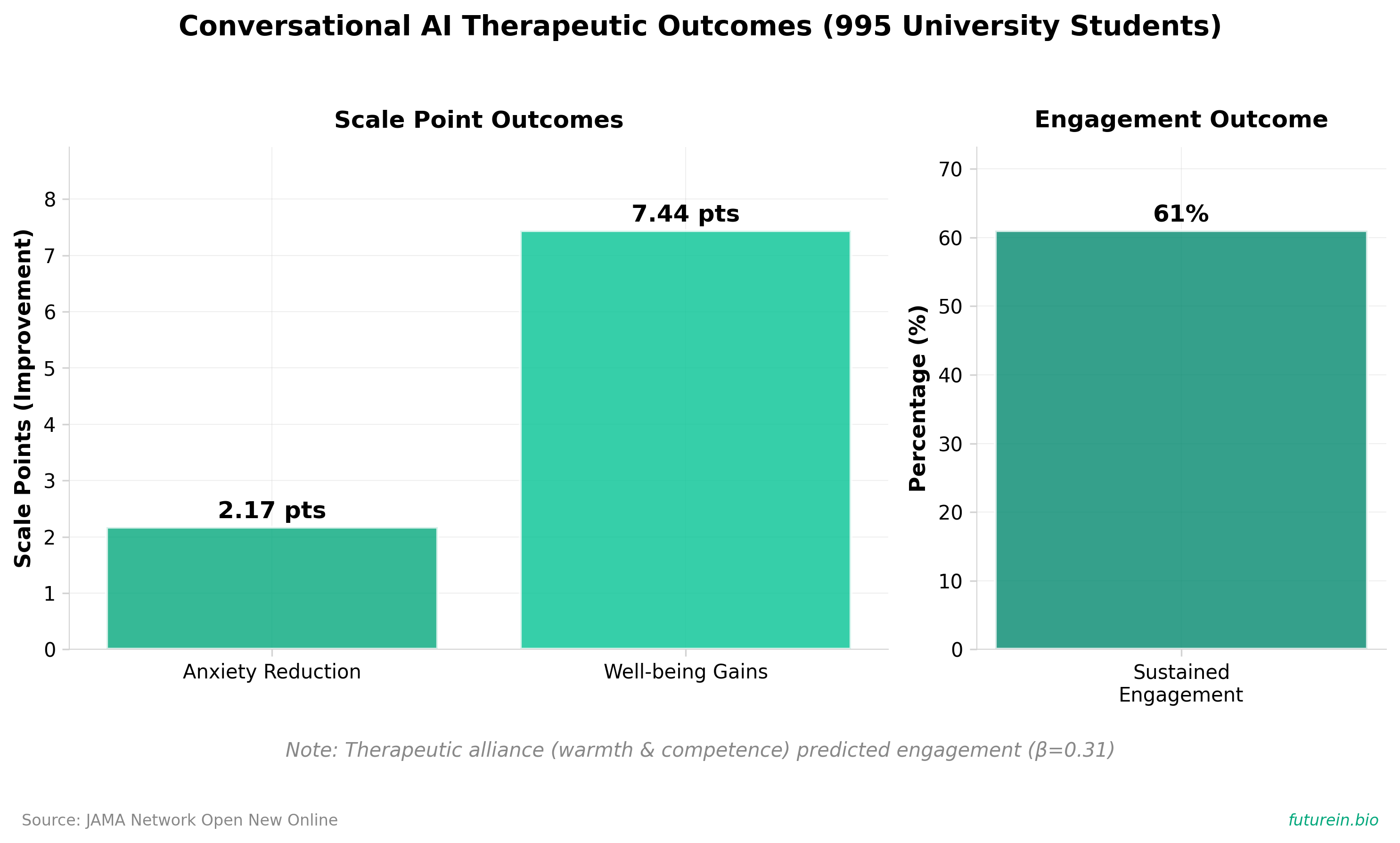
• AI-driven digital therapeutics demonstrate scalable efficacy for anxiety and well-being: Conversational AI outperformed face-to-face group therapy in anxiety reduction (2.17-point improvement) and well-being gains (5.72-9.16 points) among 995 university students, with 61% sustained engagement through 12 weeks, positioning AI as a viable adjunct for addressing mental health resource gaps in clinical settings.
• Therapeutic alliance is the critical mechanistic driver of AI platform engagement and outcomes: Structural equation modeling revealed perceived warmth and competence predicted engagement (β=0.31), which directly drove symptom improvement (β=-0.58), indicating that replicating human therapeutic qualities in AI design is essential for clinical efficacy rather than a secondary feature.
• PTSD non-responsiveness and declining therapy-seeking intentions signal implementation guardrails needed: The platform showed no benefit for trauma symptoms and reduced post-intervention therapy intentions in the AI group, requiring developers to establish trauma-specific modules, referral protocols, and monitoring systems to prevent inappropriate substitution of AI for specialized human care.
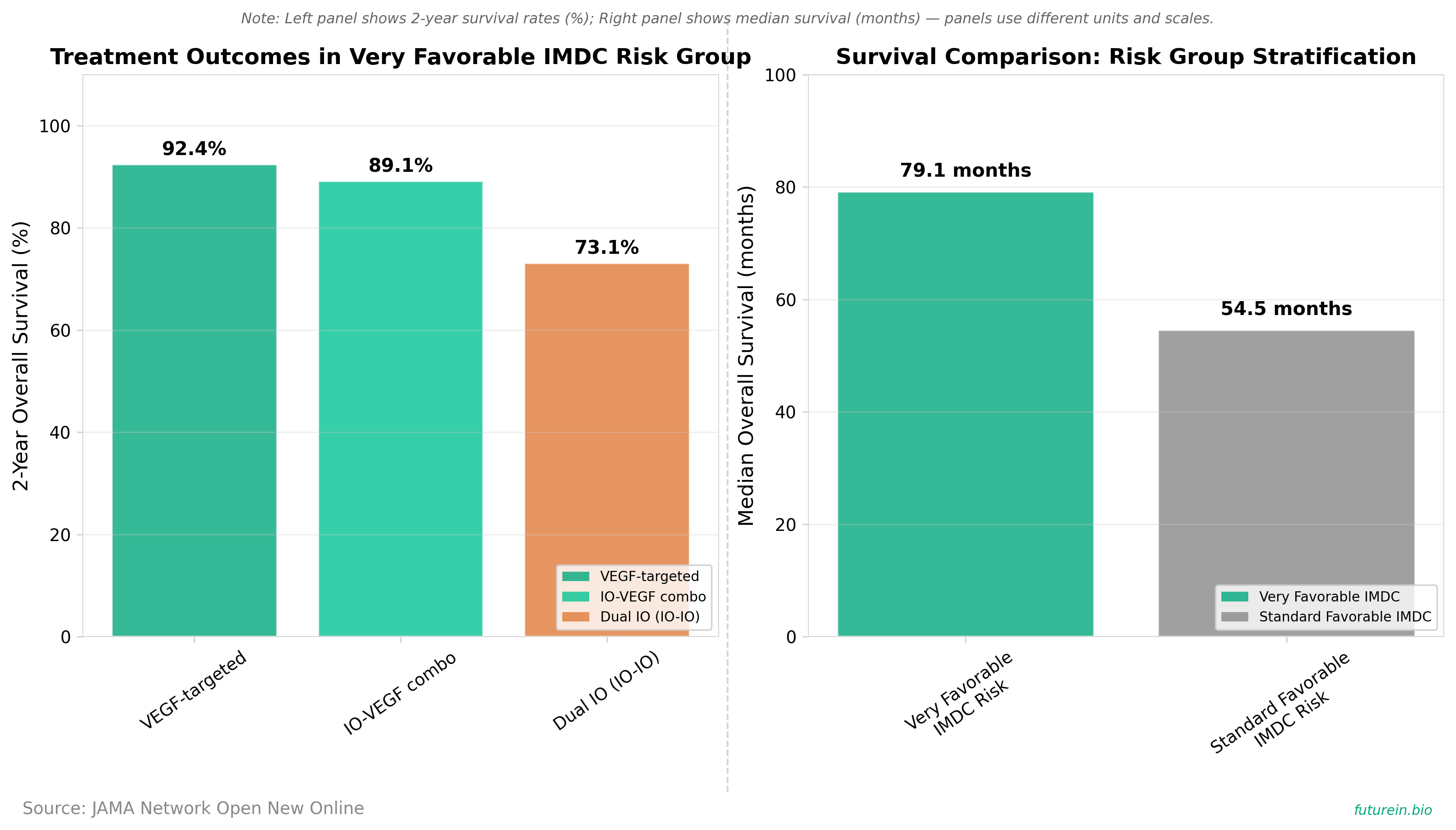
4. Validation of the Very Favorable IMDC Risk Group in Metastatic Renal Cell Carcinoma
• This study validates a refined “very favorable” IMDC risk subgroup within metastatic renal cell carcinoma patients, analyzing 641 favorable-risk patients where 176 (27.5%) met very favorable criteria and demonstrated superior median overall survival of 79.1 months versus 54.5 months for standard favorable-risk patients. The very favorable subgroup showed 92.4% 2-year overall survival with VEGF-targeted therapy, 89.1% with IO-VEGF combinations, but only 73.1% with dual immunotherapy (IO-IO), representing a 3.64-fold higher risk of death with IO-IO treatment.
• This refined risk stratification immediately impacts treatment selection for the 15-16% of mRCC patients classified as favorable risk, as it identifies a distinct 27.5% subgroup within this population that experiences significantly worse outcomes with dual immunotherapy approaches. The molecular validation showed 64.7% PBRM1 alterations, only 8.8% BAP1 alterations, and 21.9% PD-L1 positivity in very favorable patients, indicating angiogenic rather than immunogenic tumor biology that responds poorly to IO-IO combinations with response rates of only 26.3% versus 63.3% for IO-VEGF.
• This research supports the broader trend toward precision oncology by demonstrating how clinical criteria can serve as surrogates for molecular features, potentially reducing the need for expensive genomic testing while improving treatment outcomes. The findings reinforce the importance of VEGF inhibition in angiogenic tumors and may influence future clinical trial design by incorporating very favorable risk as a stratification factor, ultimately contributing to more personalized treatment algorithms in kidney cancer where median survival differences exceed 24 months between optimal and suboptimal therapy selection.
New Research
1. Steering semi-flexible molecular diffusion model for structure-based drug design with reinforcement learning
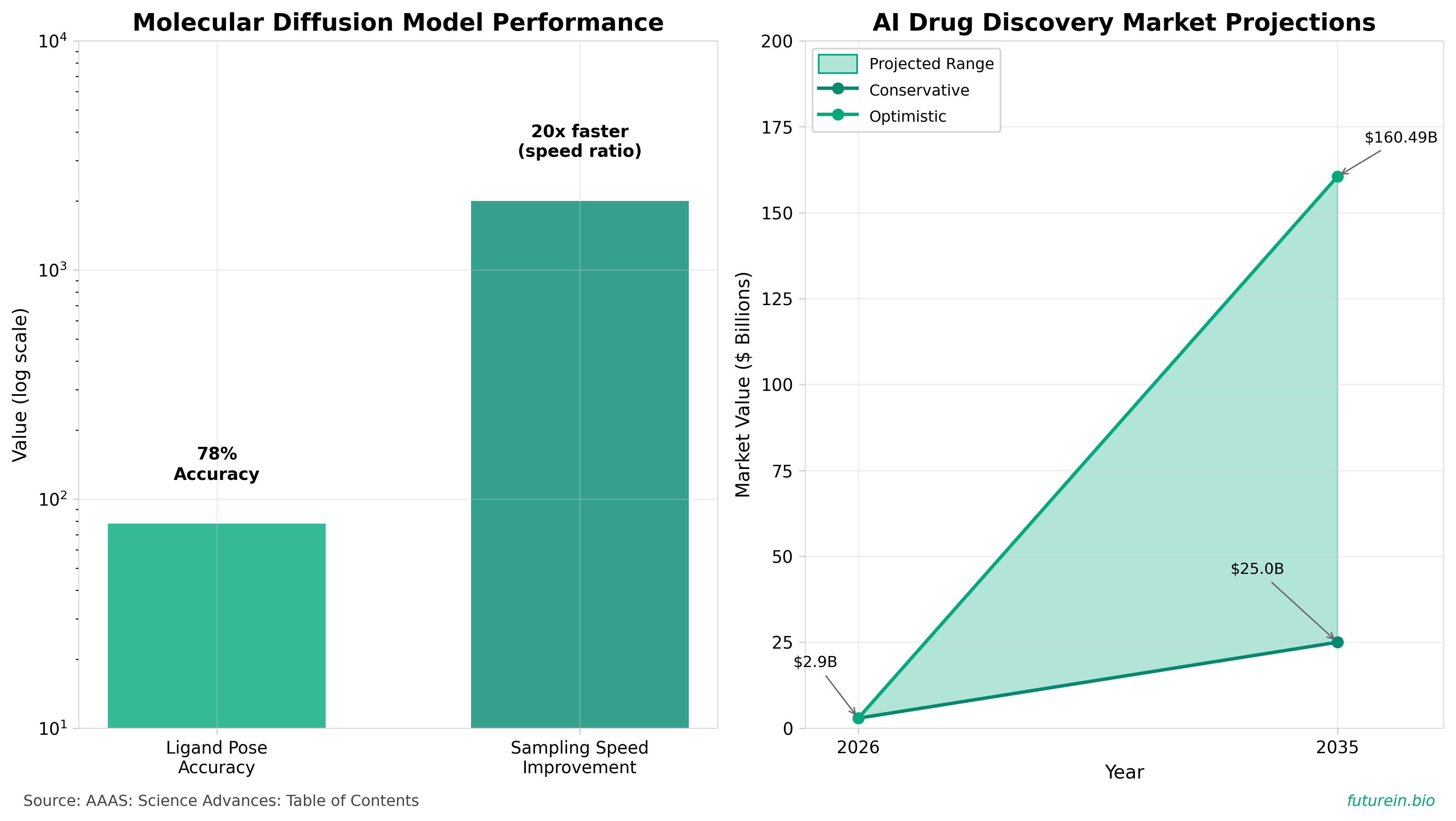
• This article presents a reinforcement learning approach to guide semi-flexible molecular diffusion models for structure-based drug design, building on technology that achieves 78% accuracy in predicting ligand poses within protein pockets (RMSD <2 Å) and delivers 20x acceleration in sampling speed (reducing from 1000 to 50 steps).
• The development addresses critical bottlenecks in computational drug discovery by significantly improving both training and inference efficiency, contributing to the AI drug discovery market’s explosive growth from $2.9 billion in 2026 to projected $25.0-160.49 billion by 2035 (CAGR of 12.6-23.22%), with machine learning applications currently holding 40% market share.
• This advancement reinforces the strategic importance of AI-driven drug design platforms, evidenced by massive investments including the $1 billion Eli Lilly-Nvidia partnership and Xaira Therapeutics’ $1+ billion funding round, while lead optimization maintains 50% market share and digital health funding surged to $7.4 billion in Q1 2026 with 60% concentrated in mega-rounds exceeding $100 million.
2. Astrocyte-based CAR immunotherapy against Alzheimer’s disease
• Novel cell-type engineering strategy overcomes prior therapeutic limitations: Shifting from microglial and myeloid cell-based approaches to astrocytes addresses technical challenges of previous CAR immunotherapies; astrocytes offer advantages as abundant, naturally phagocytic brain cells that are more readily engineerable, positioning this as a potentially more tractable manufacturing pathway for CNS-directed cell therapies.
• Modular CAR construct design enables targeting flexibility for Alzheimer’s pathology: Four distinct anti-Aβ CAR variants were engineered combining different antibody specificities (soluble vs. oligomeric Aβ targeting) with diverse phagocytic receptor domains (MEGF10, Dectin1), demonstrating proof-of-concept for customizable targeting strategies that could be optimized for different disease stages or patient subpopulations.
• One-time, disease-modifying therapy potential represents significant clinical and commercial advantage: Lead investigator Marco Colonna indicates this approach could enable a single-administration, preventive treatment for Alzheimer’s disease if preclinical 5xFAD mouse model results translate clinically—a substantial differentiation from current chronic dosing regimens in neurodegenerative disease management.
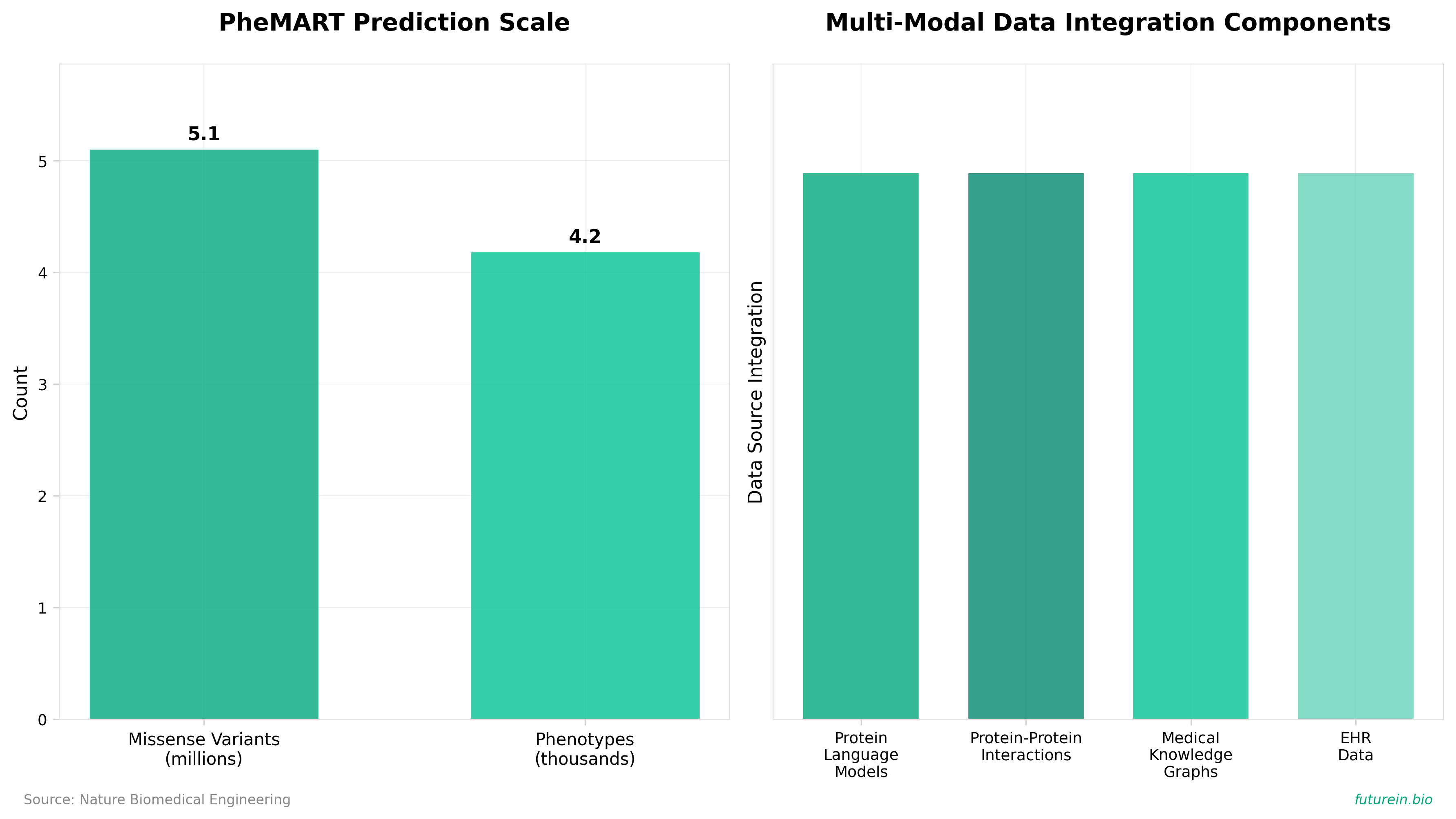
3. Phenotypic prediction of missense variants via deep contrastive learning
• Comprehensive phenotype prediction at scale: PheMART predicts clinical consequences for 5.1 million missense variants across 4,179 phenotypes using a unified metric space, directly addressing the gap between pathogenicity assessment and actual phenotypic heterogeneity—enabling faster differential diagnosis in clinical genomics workflows.
• Multi-modal integration outperforms single-approach models: The framework combines protein language models, protein-protein interactions, medical knowledge graphs, and EHR data through contrastive learning, substantially outperforming existing pathogenicity-only tools and demonstrating that variant interpretation requires integrated multi-source data rather than siloed computational approaches.
• Rare disease diagnosis acceleration: PheMART effectively pinpoints causative variants and clinical diagnoses in undiagnosed cases, with publicly available interactive database and code—providing diagnostic labs and rare disease centers immediate access to phenotype-variant associations that reduce diagnostic odyssey timelines.
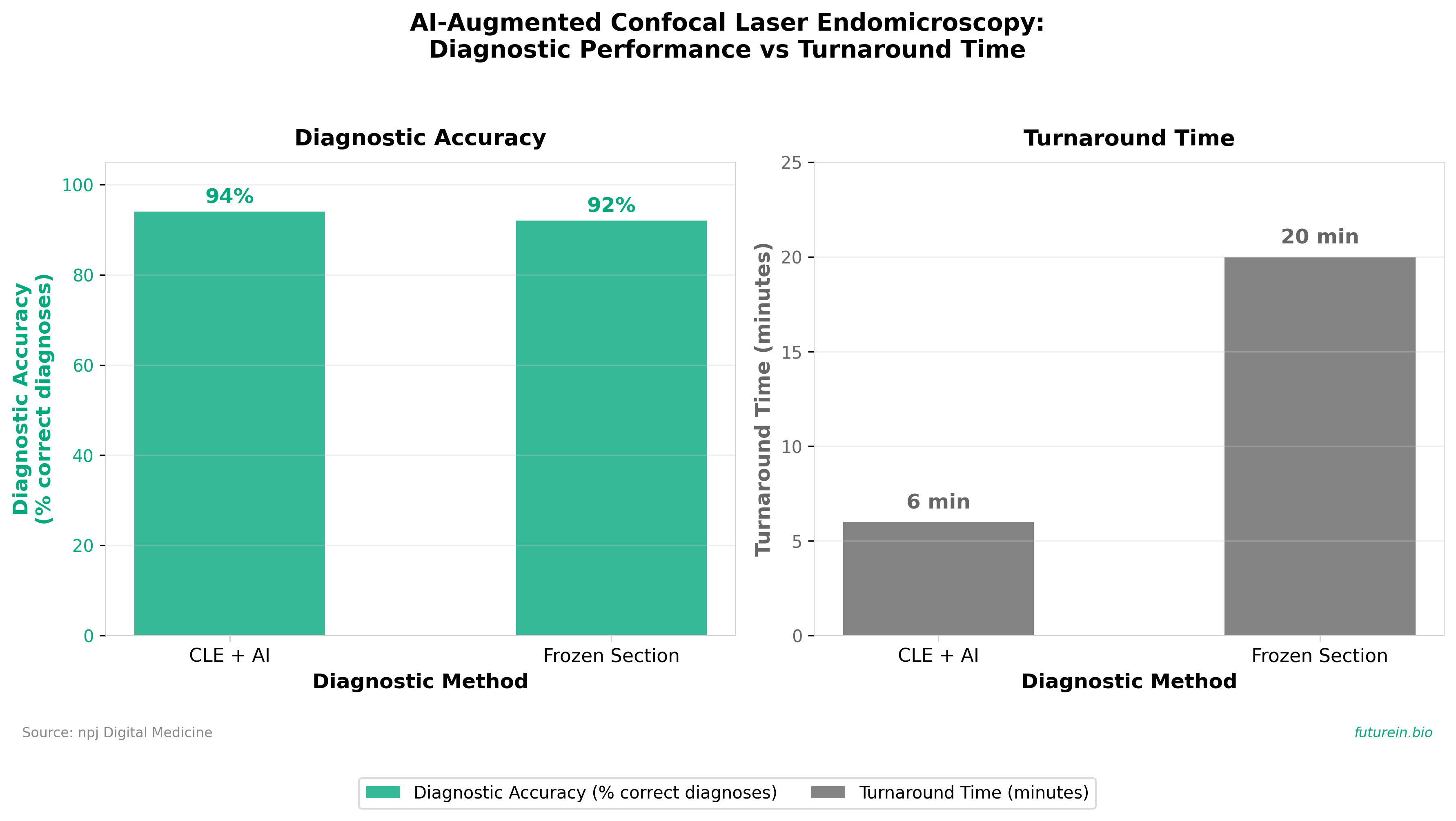
4. AI Augmented Confocal Laser Endomicroscopy for Rapid Intraoperative Diagnosis of Brain Tumors
• Clinical Workflow Acceleration with Non-Inferior Diagnostics: Confocal laser endomicroscopy (CLE) achieved diagnostic accuracy parity with frozen section (0.94 vs 0.92) while reducing turnaround time from 20 minutes to ~6 minutes, directly enabling real-time surgical decision-making and potentially reducing operative time in brain tumor resection procedures.
• AI Model Validates Platform Integration Readiness: The Swin Transformer-based AI achieved 0.94 accuracy for tumor detection and 0.88 for subtype diagnosis, demonstrating sufficient performance to support clinical deployment; code availability and multi-institutional validation (461 biopsies, 376 patients) position this for regulatory pathway advancement and market differentiation.
• Regulatory and Commercialization Pathway Emerging: Multi-center prospective trial design with published code, defined performance metrics against the clinical standard, and international institutional collaboration (Korea, Canada) create a clear foundation for CE mark/FDA submissions; the 0.79 specificity gap versus frozen section (vs. 0.68) warrants addressing before replacement strategy repositioning.
5. Predictive models for the occurrence and lymph node metastasis of papillary thyroid carcinoma with regional risk heterogeneity
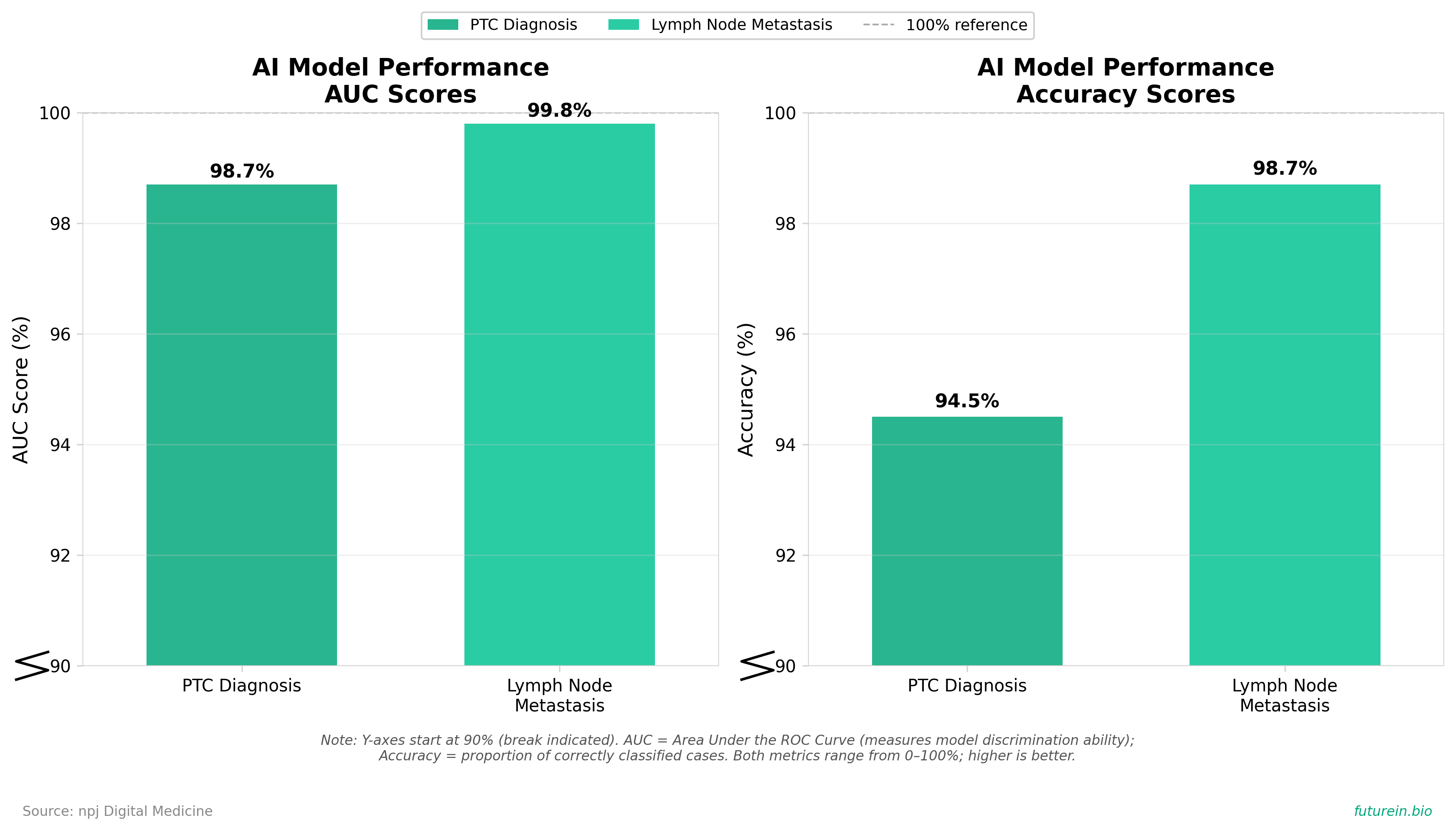
• This study developed AI models using deep neural networks to predict papillary thyroid carcinoma (PTC) occurrence and lymph node metastasis from 419 samples across Asia, Europe, and America, achieving 98.7% AUC with 94.5% accuracy for PTC diagnosis and 99.8% AUC with 98.7% accuracy for metastasis prediction, while identifying key genetic markers (SYT1, REN, CNTN5, ADAM12 for diagnosis; COL9A1, CYP4F3, GAD1 for metastasis).
• The high-accuracy predictive models (94.5% diagnostic accuracy, 98.7% metastasis prediction accuracy) could significantly improve early detection and treatment planning for PTC, which affects 68 per million people annually in the US with lymph node metastasis occurring in up to 30% of patients at diagnosis, potentially reducing the 31.5% recurrence rate associated with lymph node involvement.
• This demonstrates the growing precision of AI-driven cancer diagnostics using transcriptomic data, with models achieving near-perfect accuracy (99.8% AUC) that could accelerate personalized medicine adoption in oncology, particularly as PTC incidence has increased from 9.9 to 16.1 per 100,000 between 2003-2017 before plateauing, creating demand for more sophisticated diagnostic tools that can handle regional risk variations identified in this multi-continental dataset.