Headlines & Launches
- OpenAI Launches ChatGPT for Clinicians
- ŌURA Acquires Galen AI to Pioneer Unified Health Companion, Merging Clinical Records with Continuous Biometrics
- Johnson & Johnson teams up with Viz.ai on hematoma detection
- The Casino Model: Why Medtech VCs Are Betting Billions on Unproven AI
- Almanac Health Launches with $10M to Scale Research-Validated Clinical AI for Point-of-Care Support
Deep Dives & In Depth Analysis
- Behaviorally Informed Text Messaging to Promote Colon Cancer Screening
- Prostate Cancer Incidence, Mortality, and Survival in Switzerland
- A Prediction Model for Risk of Death in Kidney Transplant Recipients
- Sequential Surveillance of RSVpreF Vaccination Early in the Postapproval Period
- Adaptive Mobile Health Intervention to Reduce Excess Gestational Weight Gain
New Research
- Programmable RNA translation through deep learning-driven IRES discovery and de novo generation
- DeepDrugDiscovery identifies blood–brain barrier permeable autophagy enhancers for Alzheimer’s disease
- Skill-Based Virtual Reality Therapy in High-Impact Chronic Pain: 2-Year Follow-Up Results From a Secondary Analysis of a Randomized Controlled Trial
- Comprehensive profiling of clinically approved kinase inhibitors reveals mutation-specific inhibitors and opportunities for drug repurposing
- Genome editing of phylogenetically distinct bacteria using cross-species retron-mediated recombineering
Market Outlook
| | Price (USD) | 1D | YTD | | :— | :— | :— | :— | | Company | | | | | Eli Lilly and Company (LLY) | 934.60 | +9.80% | | Johnson & Johnson (JNJ) | 229.85 | +1.10% | | Novo Nordisk A/S (NVO) | 42.22 | +4.79% | | Pfizer Inc. (PFE) | 26.70 | +1.68% | | Sector Index | | | | | iShares Biotechnology ETF (IBB) | 168.69 | +2.00% | | State Street SPDR S&P Biotech ETF (XBI) | 131.33 | +1.89% | | State Street SPDR S&P Pharmaceuticals ETF (XPH) | 57.61 | +1.89% | | Vanguard Health Care Index Fund ETF Shares (VHT) | 272.46 | +2.12% | | General Market Index | | | | | Russell 2000 Index (^RUT) | 2799.91 | +2.21% | | Invesco QQQ Trust (QQQ) | 667.74 | +0.93% | | State Street SPDR S&P 500 ETF Trust (SPY) | 718.66 | +0.99% | —
Headlines & Launches
1. OpenAI Launches ChatGPT for Clinicians: Free AI Documentation and Research Tool for Verified Physicians
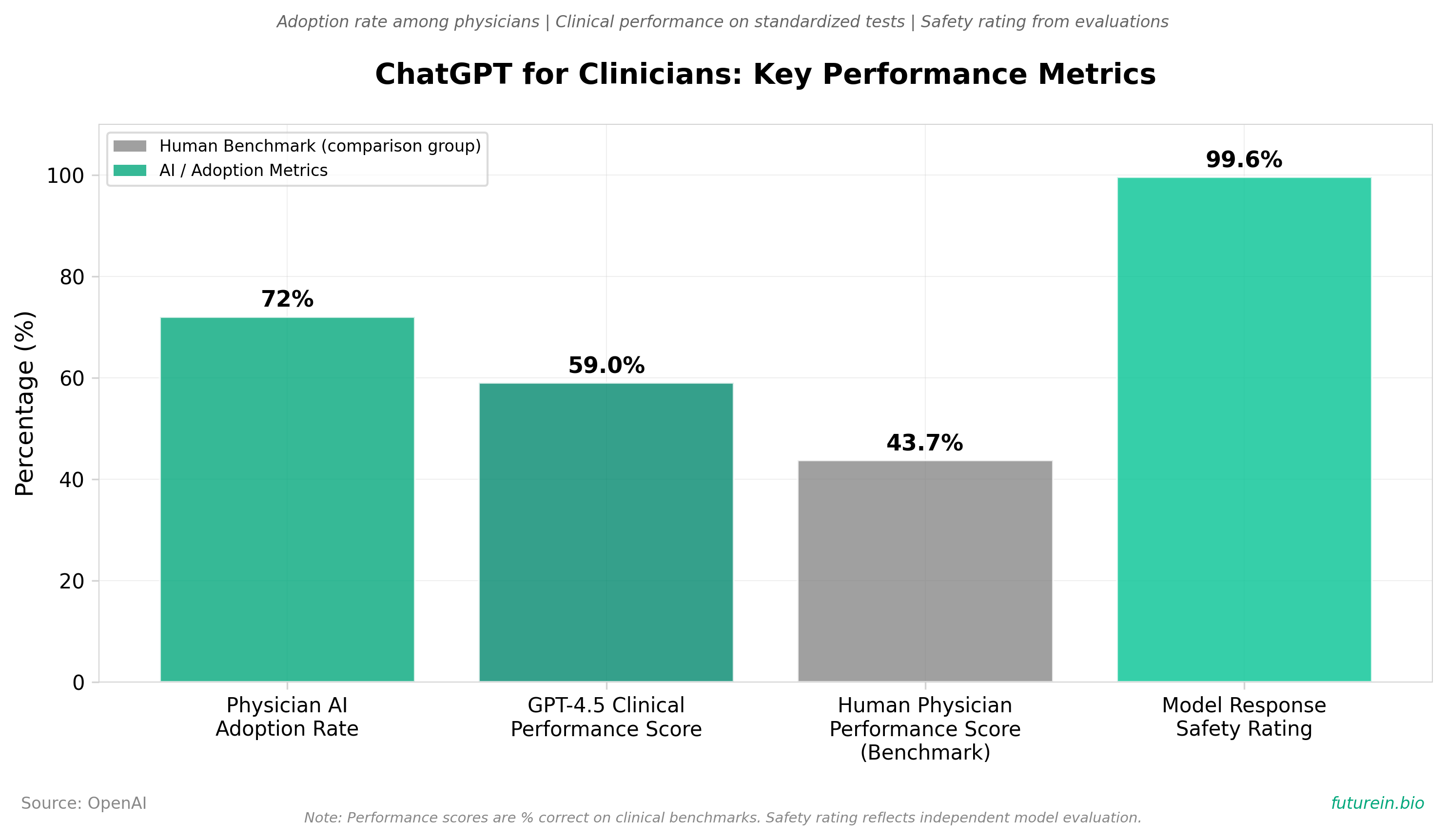
• Physician AI adoption has reached critical mass at 72% (up from 48% YoY), creating immediate market opportunity for healthcare systems to implement clinician-facing AI tools. The 50% year-over-year growth rate indicates AI integration in clinical workflows is now mainstream rather than experimental, making adoption delays a competitive disadvantage for health systems addressing physician burnout.
• GPT-5.4 in the Clinicians workspace scores 59.0 on HealthBench Professional—35% higher than human physicians (43.7)—establishing quantifiable clinical utility that justifies institutional investment and removes performance uncertainty from adoption decisions. The model’s superiority on care consults, documentation, and research tasks directly addresses documented sources of administrative burden.
• Pre-launch validation of 700,000+ model responses with 99.6% safety/accuracy rating and 6,924 real-world clinician conversations provides sufficient evidence for regulatory confidence and risk mitigation. Organizations can now justify BAA-compliant deployment with documented clinical safety data rather than relying on general-purpose AI benchmarks.
2. ŌURA Acquires Galen AI to Pioneer Unified Health Companion, Merging Clinical Records with Continuous Biometrics
• ŌURA acquired Galen AI, a Stanford-founded startup that unifies fragmented health data from over 10,000 healthcare systems, to merge clinical records, lab results, and medications with continuous biometric data from their smart ring platform. This follows ŌURA’s recent $875-900M Series E funding at $11B valuation (doubled from $5B) and builds on their 80% market share of the global smart ring market, with 5.5+ million units sold and 2024 revenue of $500M representing 100% YoY growth.
• The acquisition enables ŌURA to transition from biometric tracking to a comprehensive AI-powered health companion that contextualizes daily habits and biometric trends with official medical history from 800+ healthcare institutions across 12,000+ locations in the US, UK, and Canada. This integration of EMR and lab data positions ŌURA’s ring as a diagnostic and monitoring partner that doctors can actually use, moving beyond the typical hardware metrics focus of competitors.
• This represents a strategic inflection point in the convergence of consumer wearables and clinical healthcare, as smart rings now capture 75% of all US fitness tracker revenue (up from 46% one year ago) with 195% growth in sales over nine months. ŌURA’s systematic acquisition pattern (including Doublepoint, Veri, and Sparta Science) demonstrates the emerging trend of wearable companies building comprehensive health AI platforms that bridge the gap between continuous consumer biometrics and fragmented clinical data systems.
3. Johnson & Johnson teams up with Viz.ai on hematoma detection
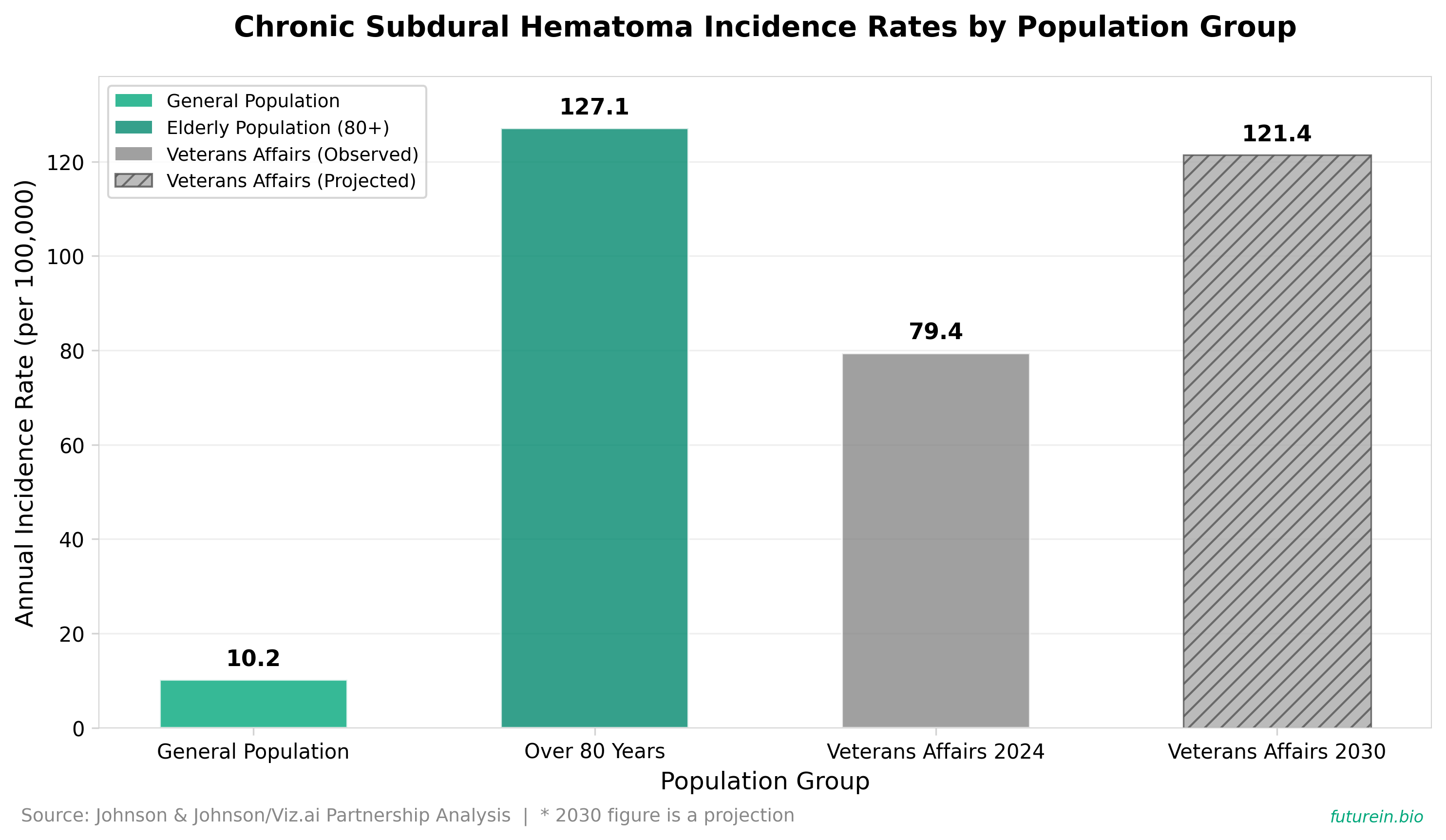
• Johnson & Johnson partnered with Viz.ai to integrate AI-powered Subdural Hemorrhage detection software with J&J’s FDA-approved TRUFILL n‑BCA Liquid Embolic System for automated detection and treatment of chronic subdural hematoma (cSDH), targeting a condition with incidence rates of 1.7-20.6 per 100,000 people annually that jumps to 127.1 per 100,000 in patients over 80.
• The collaboration enables automated patient identification for J&J’s new endovascular technology across Viz.ai’s network of nearly 2,000 U.S. hospitals covering 230+ million lives, potentially increasing device uptake in a market where cSDH has a 20.1% recurrence rate and Veterans Affairs population incidence is projected to rise from 79.4 to 121.4 per 100,000 by 2030.
• This partnership reflects the growing integration of AI diagnostics with targeted therapeutics, as Viz.ai achieved a $1.2 billion valuation with $48.8M revenue in June 2024 and ~90% click-through rates on clinical alerts, while expanding its life sciences partnerships from 7 to 13 companies over 18 months in the rapidly aging healthcare market where the median cSDH patient age is 76 years.
4. The Casino Model: Why Medtech VCs Are Betting Billions on Unproven AI
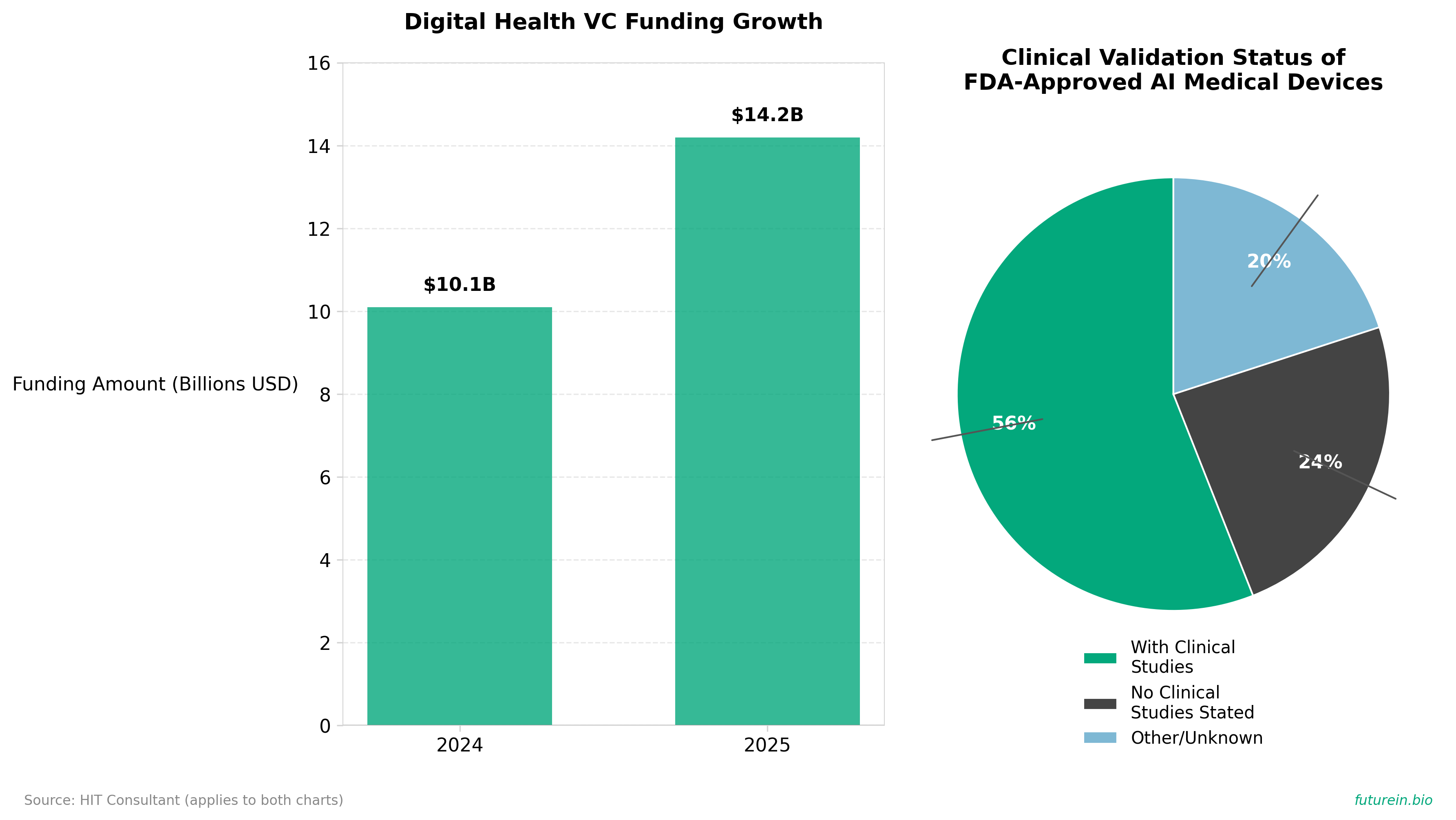
• The article critiques how medtech VCs are investing billions in AI-powered medical devices with minimal clinical validation, operating on a “casino model” where 90%+ failure rates are acceptable if a few investments deliver 10x-50x returns. U.S. digital health venture funding reached $10.1 billion across 497 deals in 2024 and climbed to $14.2 billion in 2025, while only 56% of 903 FDA-approved AI devices had clinical performance studies and 24% explicitly stated none had been conducted.
• This investment approach creates immediate market distortions where capital chases compelling narratives over clinical proof, with 96.4% of AI medical devices cleared via the streamlined FDA 510(k) pathway requiring only “substantial equivalence” rather than fresh clinical benefit data. The flood of overhyped failures creates market noise that slows adoption for genuinely strong innovations, while talent and capital scatter across too many unproven long shots.
• The trend reflects a fundamental shift toward speed-over-rigor in healthcare innovation that could reshape how medical technologies reach market, with digital health operating in largely unregulated spaces where engagement metrics substitute for outcome evidence. A 2022 analysis found average “clinical robustness” scores for venture-backed digital health companies near the bottom of measurement scales, with no meaningful correlation between evidence strength and funding raised, suggesting this pattern will likely persist as the dominant funding model.
5. Almanac Health Launches with $10M to Scale Research-Validated Clinical AI for Point-of-Care Support
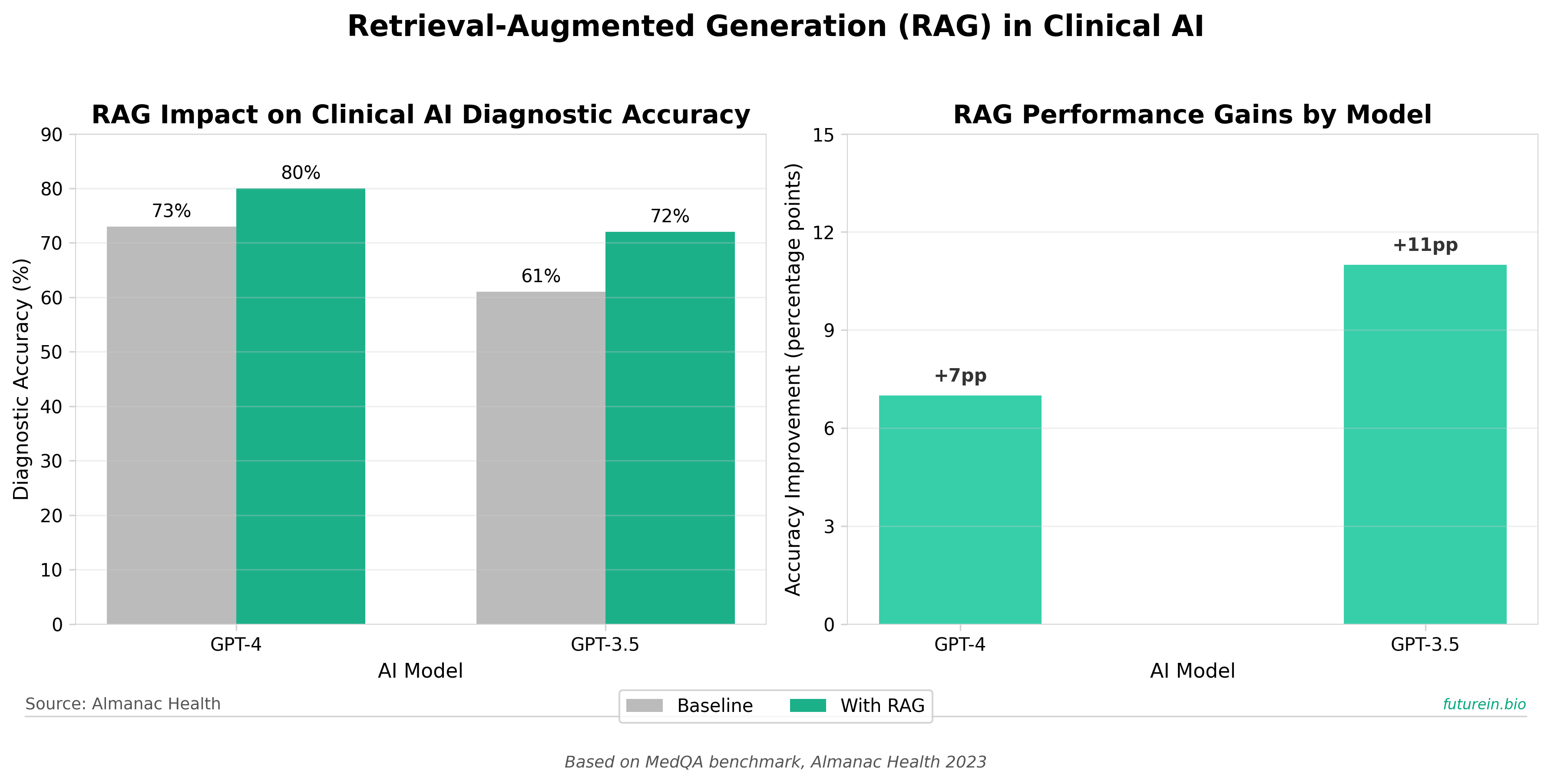
• RAG-Based Clinical AI Shows Measurable Performance Gains: Retrieval-augmented generation (RAG) improved diagnostic accuracy across multiple LLMs (GPT-4: 73% → 80%; GPT-3.5: 61% → 72%), establishing a research-validated technical standard that competitors must match to credibly enter the clinical decision support market.
• Clinician Burnout Reduction Drives Healthcare System Adoption: AI-integrated workflows reduced clinician burnout by 77% (OR 1.77, p<0.00001), directly addressing the physician fatigue crisis—making Almanac’s point-of-care integration strategy a compelling ROI argument for health systems over standalone tools.
• Institutional Control & Evidence Requirements Define Market Maturation: Almanac’s emphasis on continuous validation in academic medical centers and institutional governance reflects a fundamental market shift away from black-box deployment; health systems now demand accountability mechanisms, favoring startups that build governance into product design rather than bolting it on post-hoc.
Deep Dives & In Depth Analysis
1. Behaviorally Informed Text Messaging to Promote Colon Cancer Screening
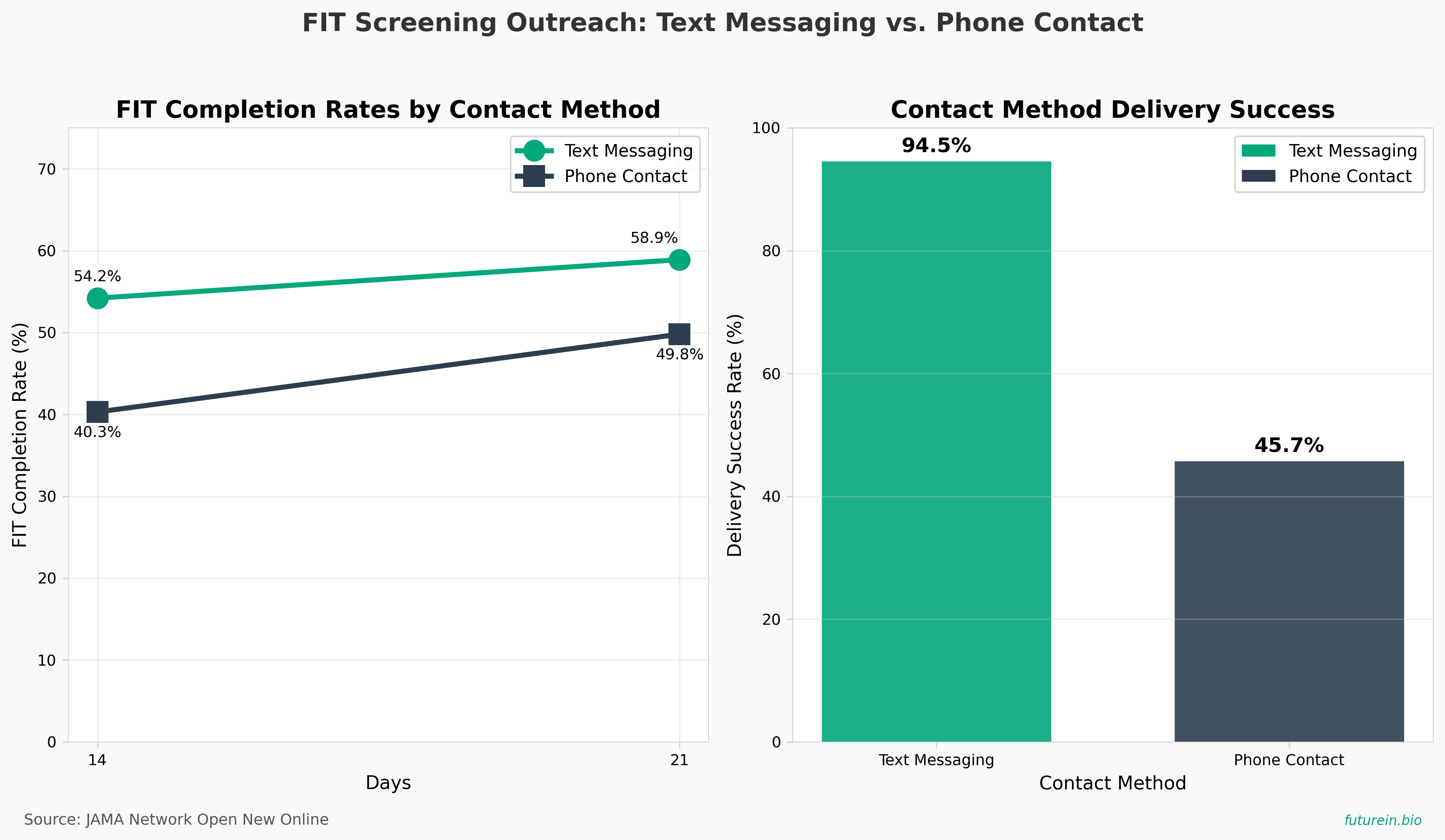
• Text messaging achieves 94.5% delivery success vs. 45.7% phone contact completion, making automated outreach dramatically more scalable for resource-constrained safety-net settings; the approach freed up 18 nursing hours monthly while improving FIT completion by 9 percentage points (58.9% vs. 49.8% at 21 days).
• Multi-touch messaging outperforms single-contact interventions: Three strategically timed text reminders (days 2, 5, 8) with behavioral nudges (deadlines, social proof, gain-framing) produced faster engagement, with day 14 completion reaching 54.2% vs. 40.3% for phone-only—a 14-point gap that translates to actionable early-screening indicators for program management.
• Intervention effectiveness is consistent across demographic subgroups (race, ethnicity, sex, insurance type, digital literacy), except younger adults (18-44 years), suggesting text-based screening outreach can address health equity gaps in underserved populations without requiring population-specific customization or higher costs.
2. Prostate Cancer Incidence, Mortality, and Survival in Switzerland
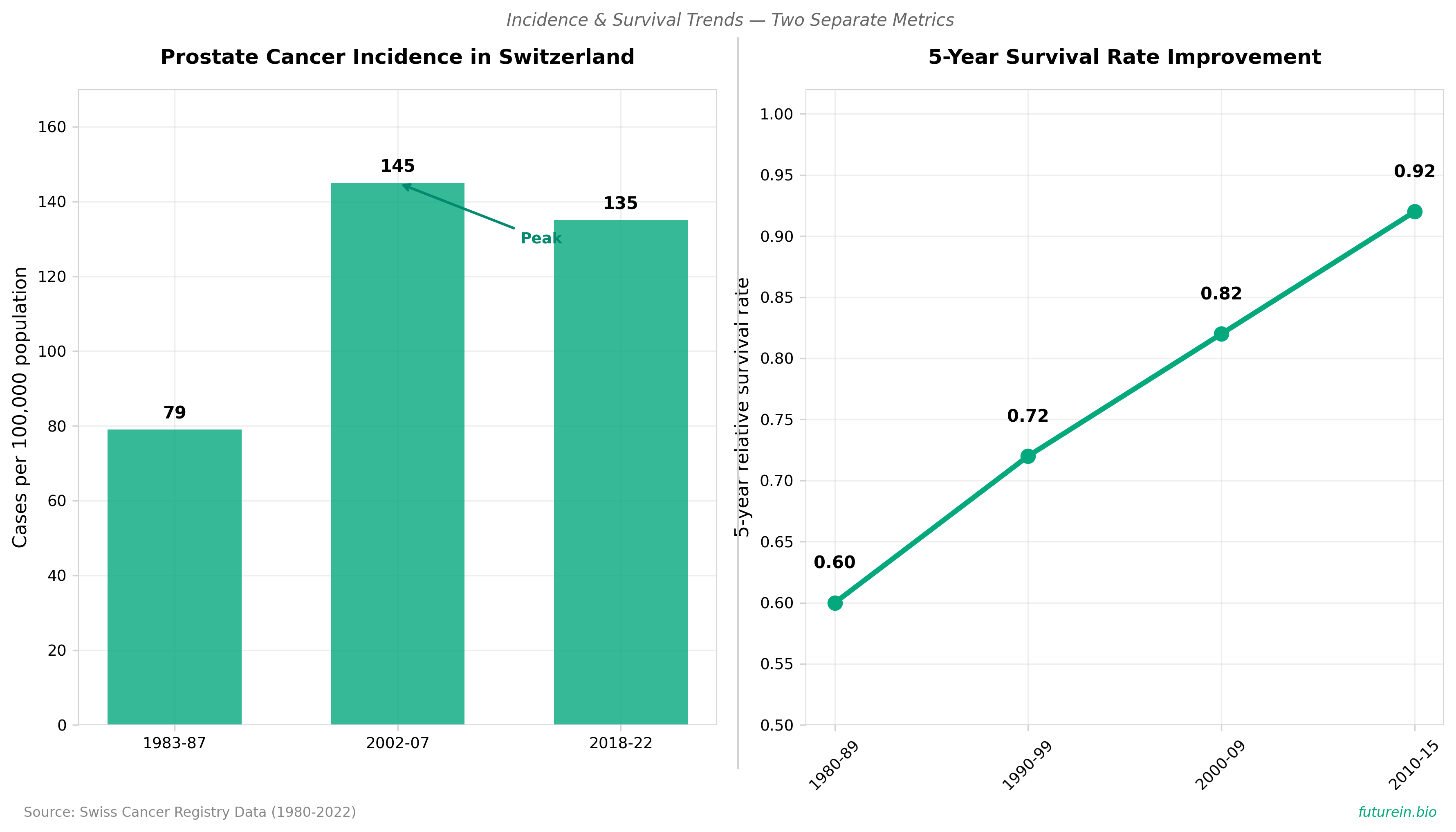
• This Swiss cohort study analyzed 41 years (1980-2021) of prostate cancer data, revealing prostate cancer incidence increased from 79 to 135 cases per 100,000 population between 1983-87 and 2018-2022, with incidence peaking around 2002-2007 before declining due to PSA screening impacts.
• The study demonstrates a significant survival paradox: while Switzerland historically had among the world’s highest prostate cancer mortality rates at 20.3 per 100,000 (30% higher than US rates), 5-year relative survival dramatically improved from 0.60 to 0.92 between 1980-1989 and 2010-2015, with mortality decreasing 13% between 2013-2017 and overall 5-year survival now exceeding 80%.
• These findings highlight critical trends for precision oncology development, showing 10-year survival rates varying dramatically by Gleason score (≤6: 98.4%, 3+4: 92.1%, 4+3: 76.5%, 8-10: 69.9%) and age stratification (5-year survival 0.85 for patients up to age 69 versus 0.70 for those 80+), providing quantitative frameworks for AI-driven prognostic tools and personalized treatment algorithms in digital health platforms.
3. A Prediction Model for Risk of Death in Kidney Transplant Recipients
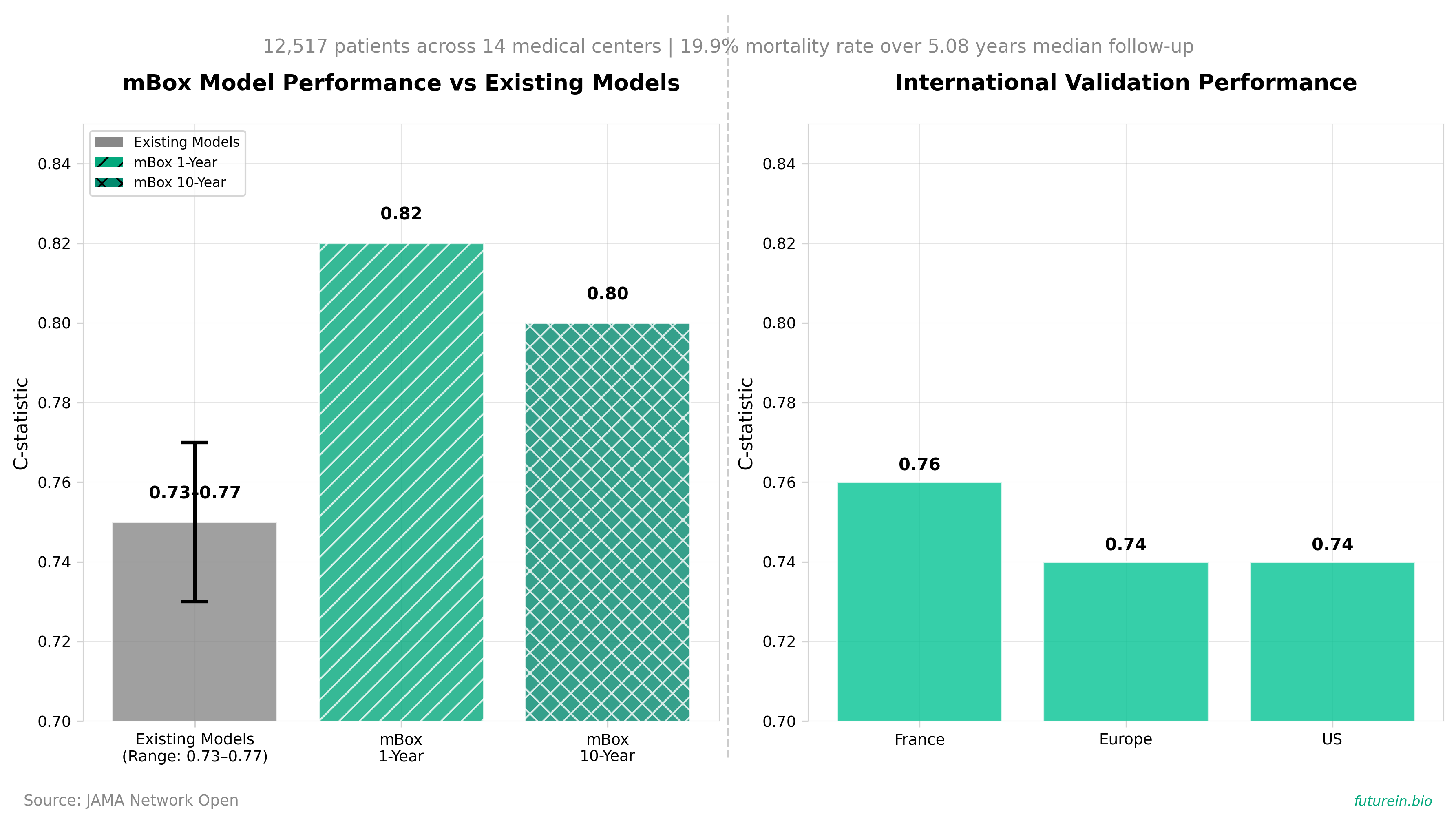
• Researchers developed and validated a mortality prediction model (mBox) for kidney transplant recipients using data from 12,517 patients across 14 medical centers, identifying 14 key risk factors that achieved C-statistics of 0.82 at 1 year and 0.80 at 10 years post-transplant. The model demonstrated superior performance compared to existing prediction scores (C-statistic 0.80 vs 0.73-0.77 for previous models) and was validated across multiple countries with consistent accuracy: France (0.76), Europe (0.74), and US (0.74).
• This validated prediction tool enables clinicians to stratify patient mortality risk at the time of transplant, potentially improving organ allocation decisions and post-transplant care strategies, addressing a critical gap given that 19.9% of the 12,517 study patients died during median follow-up of 5.08 years. The model’s ability to predict both short-term and long-term outcomes (1-10 years) with high accuracy across diverse patient populations could significantly enhance transplant clinical practice and resource allocation.
• The study represents a major advancement in precision transplant medicine, demonstrating how comprehensive data collection (121 candidate variables) and international validation can create robust predictive models that outperform traditional approaches. With kidney transplant demand exceeding supply globally and over 7 million patients with end-stage kidney disease worldwide as of 2020, such AI-enhanced prediction tools will become increasingly critical for optimizing transplant outcomes and healthcare resource management.
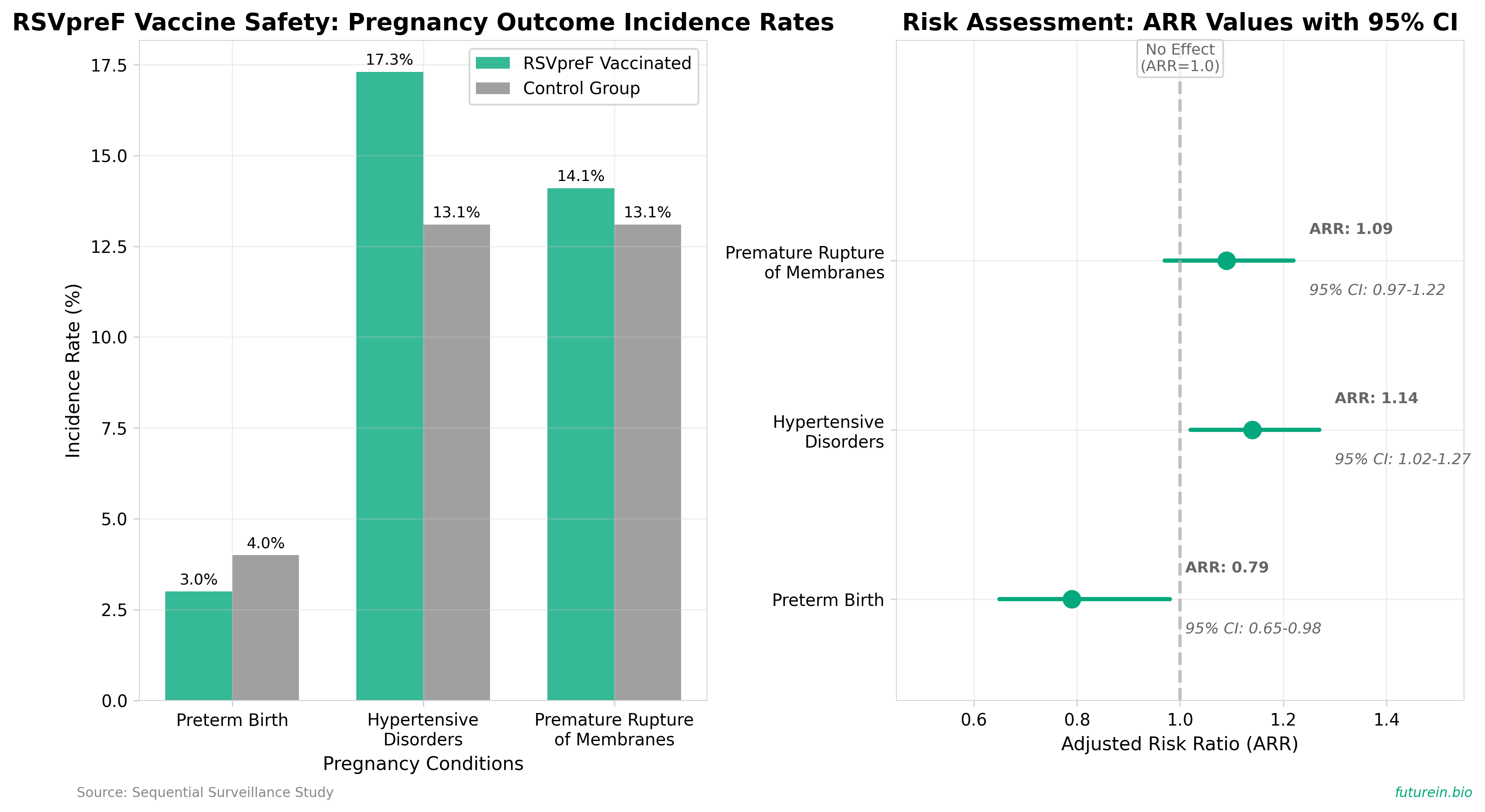
4. Sequential Surveillance of RSVpreF Vaccination Early in the Postapproval Period
• This study analyzed 13,619 RSVpreF-exposed pregnancies during the first vaccination season in the US, finding no increased risk of preterm birth (3.0% incidence vs 4.0% in concurrent comparators, ARR: 0.79, 95% CI: 0.65-0.98) but detecting potential elevated risks for pregnancy-associated hypertensive disorders (17.3% vs 13.1%, ARR: 1.14, 95% CI: 1.02-1.27) and premature rupture of membranes (14.1% vs 13.1%, ARR: 1.09, 95% CI: 0.97-1.22).
• The sequential surveillance analysis ruled out preterm birth safety concerns by the third monitoring period while identifying new safety signals that require further investigation, potentially impacting clinical recommendations for the 56.8% vaccine efficacy against infant RSV hospitalization and the CDC’s seasonal administration guidelines (September-January) for pregnant women at 32-36 weeks gestation.
• These real-world safety findings complement the growing RSV vaccine market projected to reach $28.39 billion by 2030 (42.17% CAGR from $3.51 billion in 2024), with Pfizer’s Abrysvo generating $755 million in 2024 sales, demonstrating how post-market surveillance data increasingly shapes vaccine adoption patterns and regulatory frameworks in the expanding maternal immunization sector.
5. Adaptive Mobile Health Intervention to Reduce Excess Gestational Weight Gain
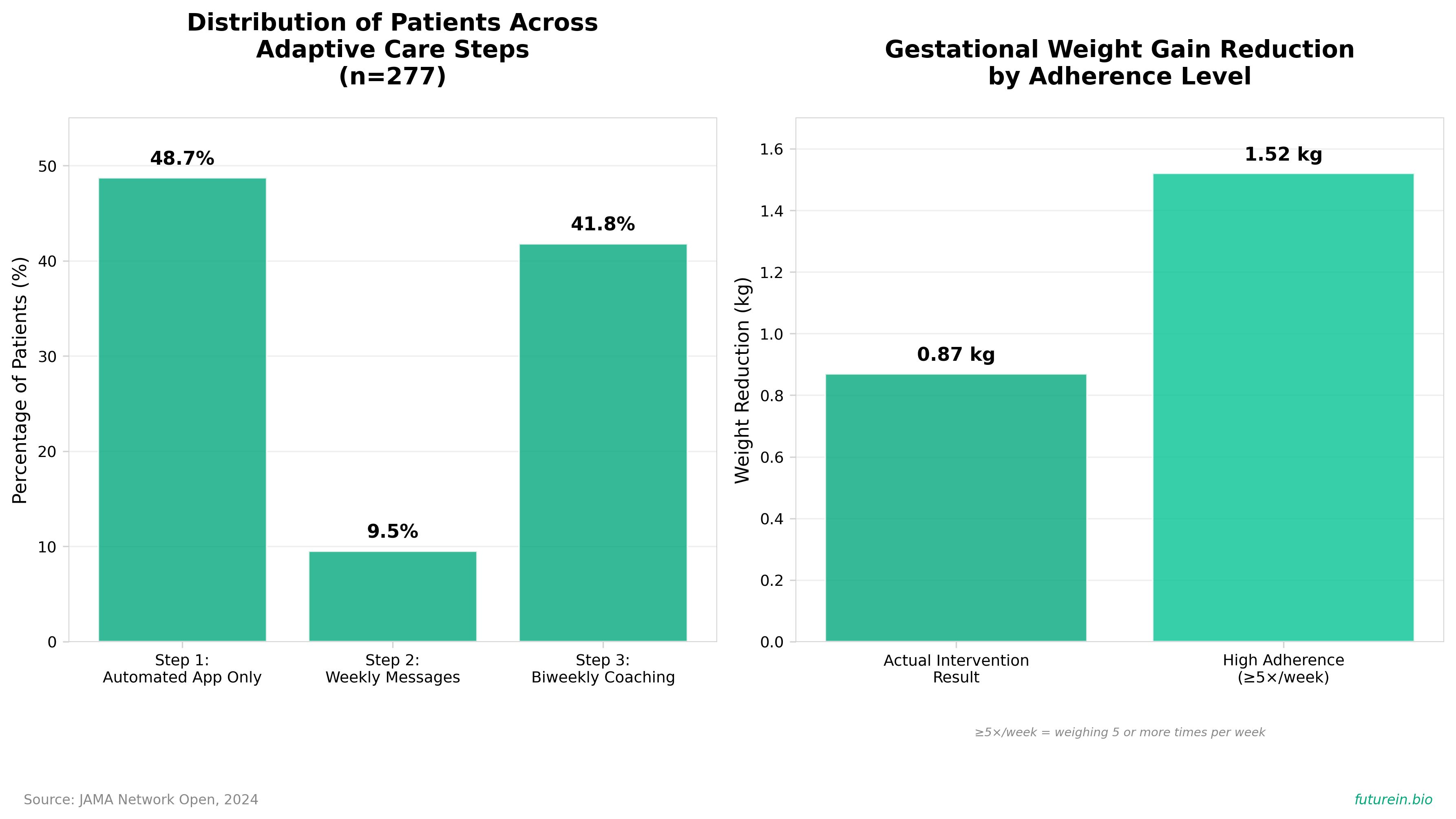
• Modest but scalable efficacy with real-world implementation: The intervention reduced total gestational weight gain by 0.87 kg and weekly gain by 0.03 kg/week in routine clinical practice—smaller than controlled trials but meaningful at population scale given that 50% of pregnant patients with overweight/obesity exceed guidelines; instrumental variable analysis showed effects could triple (1.52 kg reduction) with higher self-weighing adherence (>5x/week).
• Adaptive stepped-care model conserves resources while maintaining effectiveness: 41.8% of intervention patients progressed to step 3 (biweekly coaching) and 9.5% to step 2 (weekly messages), while 48.7% required only automated app feedback and education—this tiered approach reduces clinician burden and coaching costs while targeting intensive support only to patients with accelerated weight gain trajectories.
• Engagement and equity gaps require targeted solutions for implementation: Only 53.4% of eligible intervention patients downloaded the app and 58.5% of identified patients consented; non-Hispanic Black and Hispanic patients were less likely to enroll, and intervention participants were more educated than non-participants—successful scaled deployment depends on addressing language access (app was English-only), improving patient motivation for self-weighing, and designing engagement strategies for underrepresented populations.
New Research
1. Programmable RNA translation through deep learning-driven IRES discovery and de novo generation
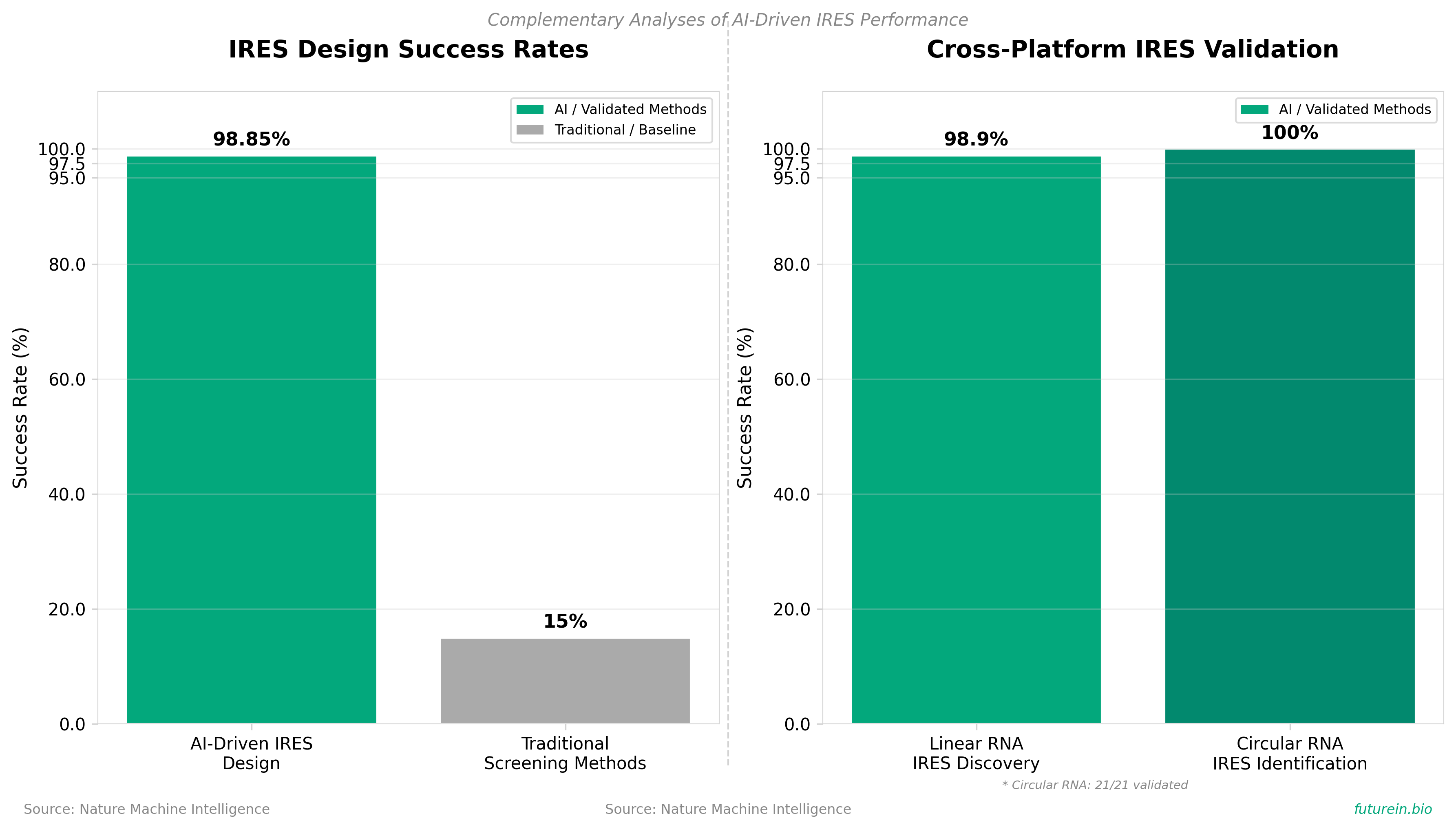
• Validated IRES design achieves 98.4-99.3% functional success rates through AI-driven discovery and de novo generation, dramatically reducing experimental screening burden compared to traditional methods; this enables rapid optimization of cap-independent translation for multi-payload RNA therapeutics where precise protein expression control is currently a development bottleneck.
• Cross-platform applicability demonstrated on both linear and circular RNAs with perfect identification of all 21 validated circular RNA IRESs; this expands the toolkit for emerging circular RNA therapeutics, a market segment growing within the broader RNA therapeutics sector (projected $28-45B by 2034) where manufacturing and delivery remain key constraints.
• Open-source framework with comprehensive datasets and code repositories provides immediate adoption pathway for synthetic biology and therapeutic development teams; combining language models (IRES-LM), evolutionary algorithms (IRES-EA), and diffusion models (IRES-DM) into a modular system allows researchers to accelerate construct optimization without proprietary tools or extensive computational infrastructure.
2. DeepDrugDiscovery identifies blood–brain barrier permeable autophagy enhancers for Alzheimer’s disease
• mTOR-independent mechanism addresses critical safety gap: Most autophagy enhancers work via broad mTOR inhibition, causing systemic side effects; DeepDrugDiscovery’s identified compounds operate through independent pathways (Pip4k2a, Mtmr1, Mapk8ip3, Ikbkb), enabling safer therapeutic profiles essential for chronic neurodegenerative disease treatment where patients tolerate long-term dosing.
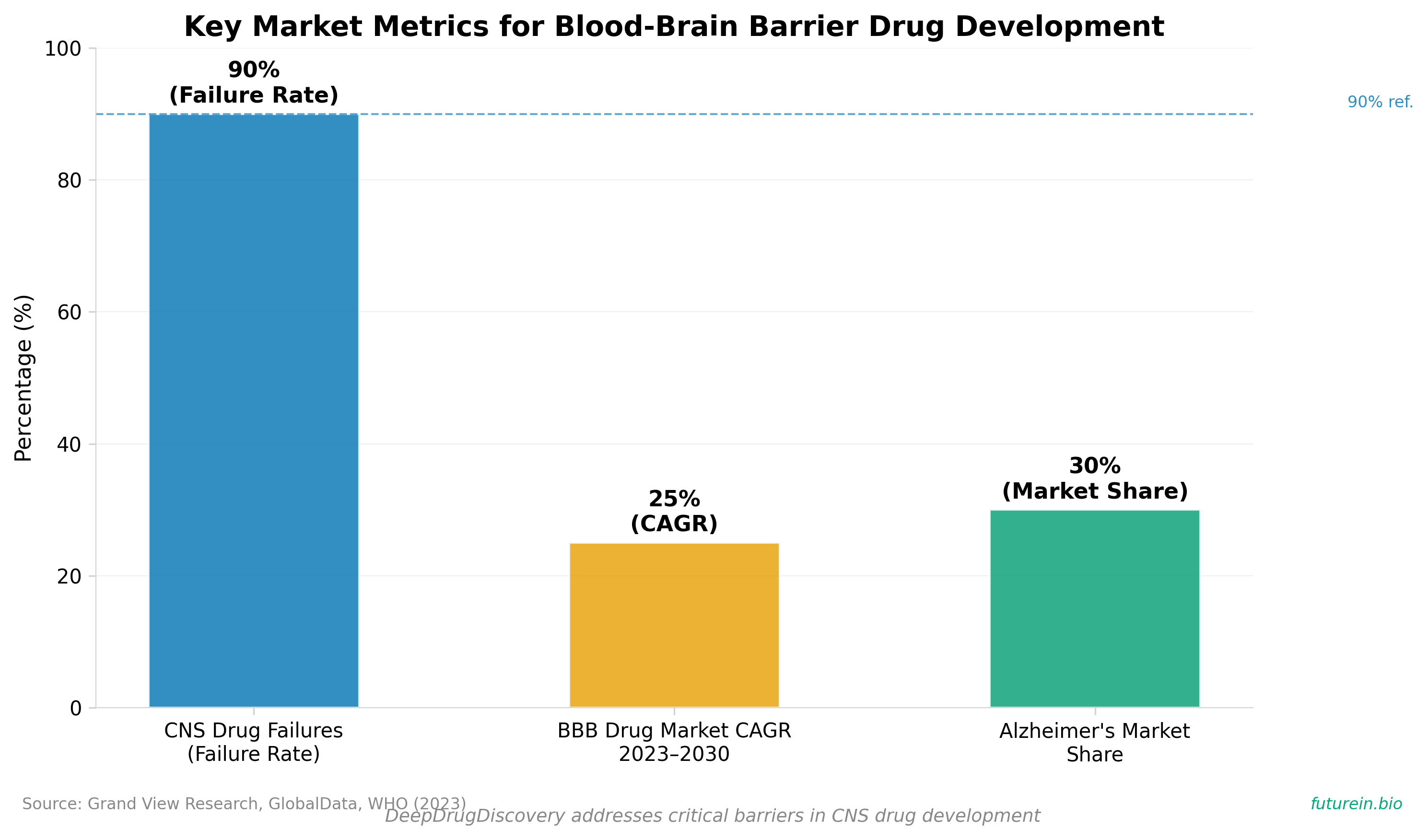
• Blood-brain barrier permeability solves a primary failure point: ~90% of potential CNS drugs fail to achieve therapeutic brain penetration; the two lead compounds demonstrated functional BBB crossing with restored memory in mouse AD models, directly addressing why most autophagy modulators historically failed in clinical translation despite promising in vitro efficacy.
• Open-source platform enables rapid competitive advantage in high-growth market: Blood-brain barrier drug market projects 25.22% CAGR through 2031 with Alzheimer’s commanding 30% share; releasing DeepDrugDiscovery as open-source with modular architecture positions early adopters (CROs, biotech companies) to accelerate proprietary compound screening at substantially reduced time-to-candidate costs compared to traditional HTS pipelines.
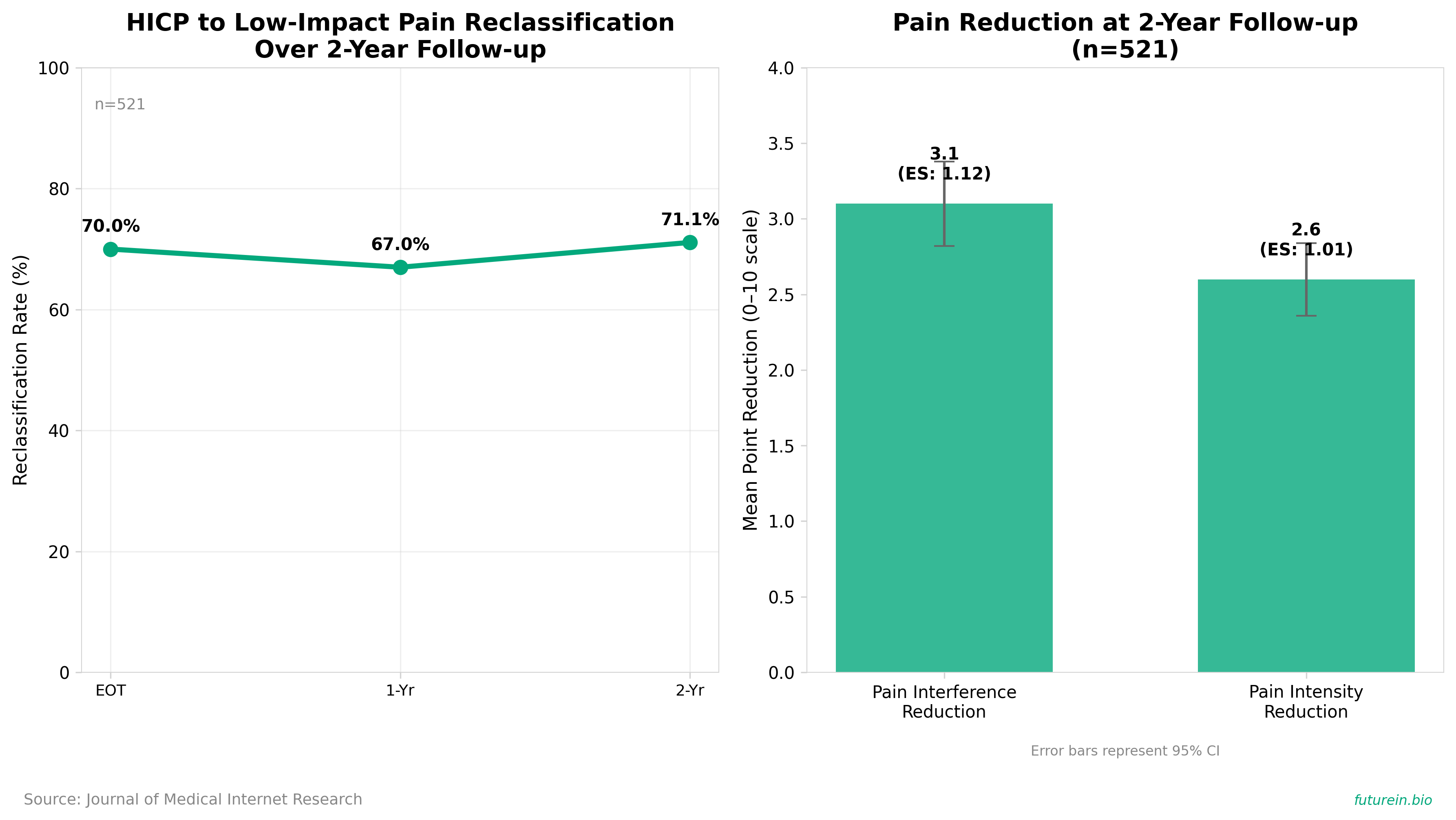
3. Skill-Based Virtual Reality Therapy in High-Impact Chronic Pain: 2-Year Follow-Up Results From a Secondary Analysis of a Randomized Controlled Trial
• This study presents 2-year follow-up results from a randomized controlled trial of skill-based virtual reality therapy for high-impact chronic pain (HICP), analyzing 536 participants with chronic lower back pain where 35.8% (192/536) had HICP. The VR therapy consisted of 56 sessions combining cognitive behavioral strategies, mindfulness, and pain education, with participants showing clinically meaningful reductions in pain interference (mean 3.1 points, effect size=1.12) and pain intensity (mean 2.6 points, effect size=1.01) at 2 years posttreatment.
• The therapy demonstrated durable effectiveness with 71.1% (106/149) of HICP participants reclassified as low-impact chronic pain at 2-year follow-up, maintaining the 70% reclassification rate observed at end-of-treatment and 67% rate at 1-year follow-up. This represents significant functional improvement in a population that affects 8.5% of adults, drives higher healthcare costs, and typically receives 5x higher opioid doses with 3x more emergency department visits compared to low-impact chronic pain patients.
• These findings support the scalability of home-based VR therapy for underserved chronic pain populations, addressing the limited access to behavioral interventions that affects patients who are “frequently overmedicalized and undertreated.” With 88.1% (420/477) participant retention at 2 years and no serious adverse events, this validates VR as a viable digital therapeutic platform for complex chronic conditions, potentially reducing healthcare utilization in a population with substantial functional disability (42% had severe or complete disability at baseline).
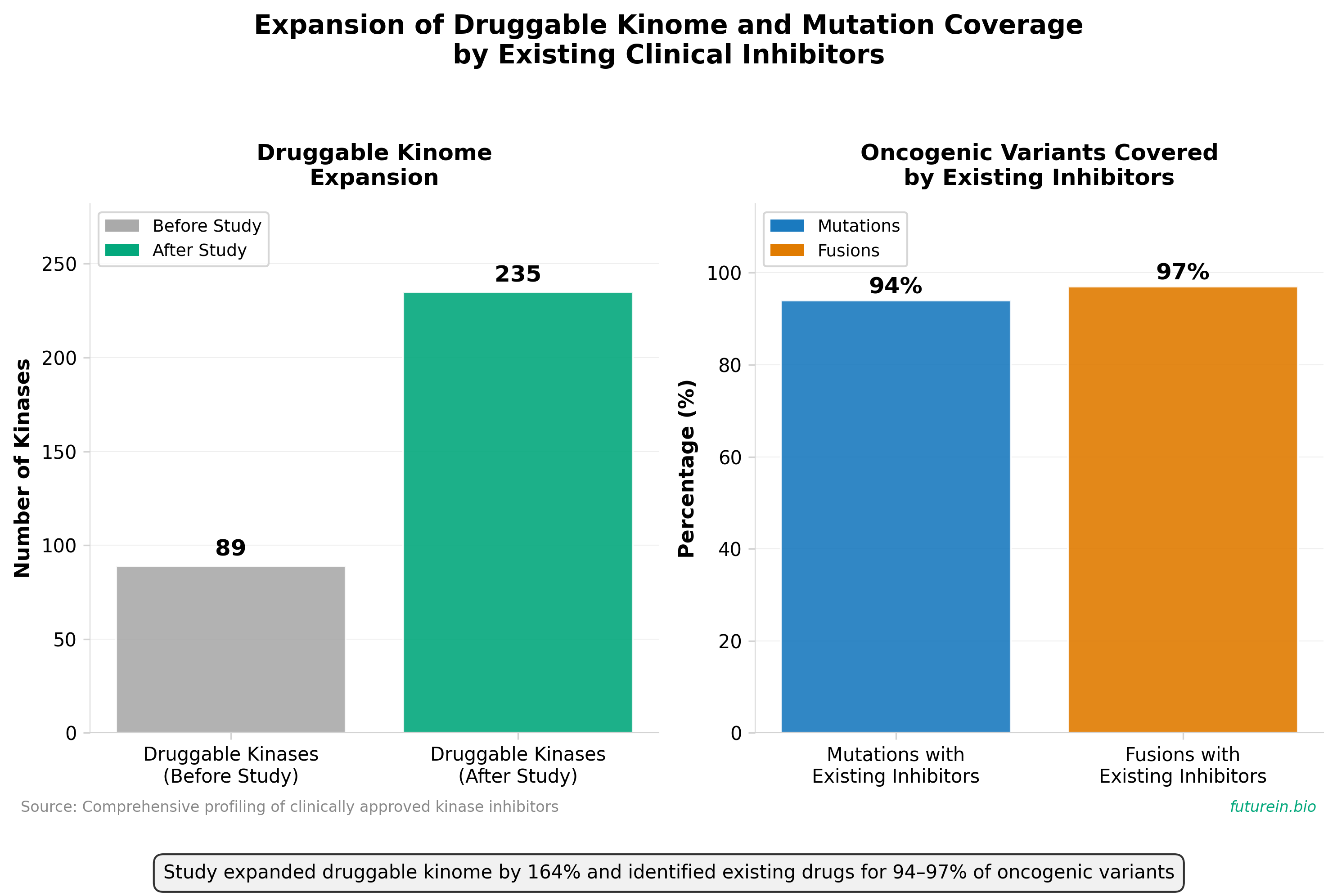
4. Comprehensive profiling of clinically approved kinase inhibitors reveals mutation-specific inhibitors and opportunities for drug repurposing
• This comprehensive study profiled 92 clinical kinase inhibitors against 758 kinases (409 wild-type and 349 oncogenic variants), generating ~290,000 kinase–drug interaction measurements using biochemical assays. The research expanded the druggable kinome from 89 to 235 kinases, revealing that 94% of mutations and 97% of fusions are inhibited by at least one existing drug, with mutation-specific selectivity patterns particularly in FGFR and MET tyrosine kinases.
• The findings enable immediate drug repurposing opportunities by identifying FDA-approved inhibitors for new indications, with validated examples including tepotinib for IRAK1/4-driven glioblastoma (showing 72-97% inhibition), brigatinib for MARK2/3-targeting pancreatic cancer, and gilteritinib for overcoming MET mutation-driven resistance. The study provides KIRHub, a web-based tool allowing clinicians to identify existing inhibitors for specific wild-type and mutated kinases to guide precision oncology treatment decisions.
• This work represents the largest functional kinase inhibitor dataset to date and establishes a systematic framework for leveraging polypharmacology in cancer treatment, moving beyond single-target approaches to multi-kinase strategies. The comprehensive mapping of kinase-inhibitor interactions supports the growing trend toward personalized medicine and drug repositioning, potentially accelerating therapeutic development timelines and reducing costs by identifying new applications for the ~100 approved kinase inhibitors currently available.
5. Genome editing of phylogenetically distinct bacteria using cross-species retron-mediated recombineering
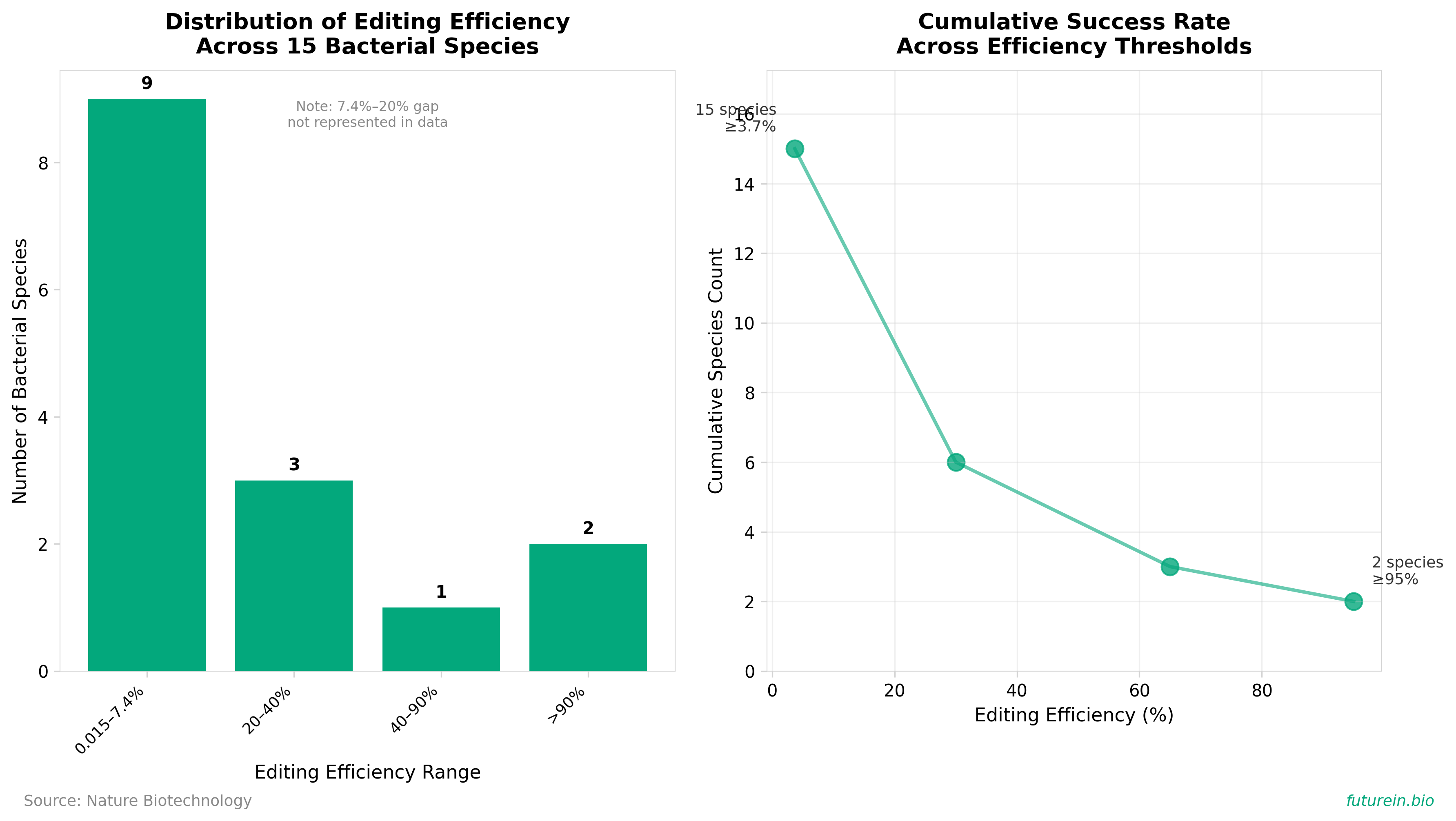
• Researchers demonstrated cross-species retron-mediated recombineering across 15 bacterial species spanning three phyla (Proteobacteria, Bacillota, Actinomycetota), achieving editing efficiencies >20% in six species, >40% in three species, and >90% in two species, with remaining nine species showing 0.015% to 7.4% efficiency rates.
• This breakthrough extends precise genome editing capabilities beyond E. coli to medically and industrially relevant bacteria including antibiotic-resistant pathogens like Klebsiella pneumoniae and Pseudomonas aeruginosa, biotechnology workhorses like Vibrio natriegens, and probiotic species like Lactobacillus reuteri, enabling broader applications in drug development and synthetic biology.
• The technology addresses a critical gap in the microbial genome editing market (valued at $241 million in 2024, projected to reach $325 million by 2034 with 4.4% CAGR), potentially reducing project costs currently ranging $5,000-$50,000 per editing project while expanding accessible bacterial hosts for the broader genome editing market projected to grow from $12.84 billion in 2026 to $52.56 billion by 2035 at 16.95% CAGR.